Abstract
The spray roasting process is recently applied for production of catalysts and single metal oxides. In our study, it was adapted for large-scale manufacturing of a more complex mixed oxide system, in particular symmetric lithium nickel manganese cobalt oxide (LiNi1/3Co1/3Mn1/3O2—NMC), which is already used as cathode material in lithium-ion batteries. An additional lithiation step was coupled with the main process in order to obtain the desired layered structure. Thermogravimetric analysis and high-temperature X-ray diffractometry built the basis for determining suitable synthesis temperature regions for the used chloride precursors and the post-treatment step. The optimized process was proven on an industrial pilot line where a setup for minimum production capacity of 12 kg h−1 was possible. The powder obtained directly after roasting had a very striking morphology compared to the final lithiated product. Hollow aggregates (≥250 μm) with overall 10.926 m2 g−1 surface area and a pore diameter of 3.396 nm were observed. Their well-faceted primary particles were converted into nanosized spheres after lithiation, building a few micrometer big high-porous agglomerates. Actual composition was verified by inductively coupled plasma atomic emission spectroscopy analysis, and the crystal structure and corresponding unit cell parameters were identified and confirmed by Rietveld fit of the derived X-ray diffraction pattern. The initial electrochemical measurements show a 149-mAh g−1discharge capacity, as determined from cyclic voltammetry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The electrification of the transport sector is a necessary and very important step for the reduction of CO2 emissions. Electric vehicles on the road are still very limited in number. The main reasons for the low acceptance are their high costs and limited driving ranges. Improvement in both categories can only be achieved with high-performance batteries at reasonable prices. To meet these market requirements, not only new materials are needed but also sustainable and efficient processes for their production. The total energy storage costs depend mainly on the cathode material, since the cathode makes up for about 42% of the battery. Moreover, according to “Avicenne Energy’s” market forecast, a long-term reduction of cost per kilowatt-hour by 40% is possible if lithium nickel manganese cobalt oxide (NMC) cathode materials are used [1]. A future robust and flexible production process of the material will therefore have great impact on the TCO (total cost of ownership).
Due to its layered structure, NMC provides optimal diffusion pathways for fast lithium intercalation and deintercalation during charging and discharging [2]. Nowadays, this already commercialized electrode active material is synthesized in different ways, such as solid state [3,4,5], sol-gel routes [6,7,8], the co-precipitation method [9,10,11,12], and combinations of them. All these multi-step syntheses have in common that they are very expensive in terms of time and energy consumption.
There are several advantages of spray techniques over these methods: the high throughput, easy-to-handle, scalable, continuous one-step process, a broad spectrum of precursor’s solutions to choose from, etc. [13, 14]. Additionally, the process of flame-assisted spray pyrolysis is well known in industry [15], due to its wide application in production of metals, oxides, nitrides, borides, and battery materials for Li-S batteries [16]. It was therefore chosen for further adaptation for battery material production using mixed metal precursors in a single solution.
According to the literature, some mixed metal compounds have been successfully synthesized via spray pyrolysis experiments in different laboratories [13], mostly using nitrates [17,18,19] or acetates [20] as precursors, and additional chelating agents [21, 22]. In every instance, a post-treatment of the initial spray pyrolysis product was necessary. The post-treatment typically includes calcination of either the pure spray pyrolysis product or its mixture with a lithium precursor (mostly LiOH), depending on whether or not lithium is introduced at the spray pyrolysis step. In contrast to the reported research activities in this field, chloride precursors have not been used yet, and the information about the resulting battery material quality, in particular NMC, produced through this method in one large-scale batch is missing as well. This publication provides information about these matters.
Nickel, cobalt, and manganese chlorides can be completely oxidized under normal spray roasting conditions [23]. At the same time, pyrohydrolysis of monovalent chlorides, such as LiCl, does not proceed below 2000 °C, whereas the temperature in the middle part of spray roasters usually varies between 600 and 900 °C [24]. A one-step production of the layered NMC material by decomposition of their precursor chlorides therefore does not seem straightforward, and the post-lithiation step seems indispensable. Therefore, post-treatment for lithiation was applied in the presented process.
In our research, thermogravimetric data and preliminary experiments built the basis for the parameter setup. The cathode material NMC was synthesized on an industrial scale via spray roasting and post-treatment for lithiation from a chloride precursor solution. The composition was verified by inductively coupled plasma atomic emission spectroscopy (ICP-AES) analysis and the specific discharge capacities were determined from cyclic voltammetry measurements.
Experimental
Thermogravimetric measurements
A thermogravimetric (TG) measurement of the mixed metal chloride precursors NiCl2·CoCl2·MnCl2·nH2O (Sigma-Aldrich, ≥98%) was conducted in order to estimate the temperature required for complete decomposition. Nickel, cobalt, and manganese chlorides in a molar ratio of 1:1:1 were dissolved in a small amount of deionized water. The solution was dried at 60 °C in air atmosphere to form a solid film. The film was carefully scraped off and ground in a mortar. For the measurement, a precise quantity of sample (60 mg) was placed into a microsized alundum crucible and heated up to 950 °С at a rate of 5 °С min−1 in synthetic air flow (80 vol% N2, 20 vol% O2).
Synthesis
Metal chlorides were chosen as precursors due to the well-known process of metal oxide synthesis from chloride solutions and further hydrochloric acid recovery from the outlet gases. An aqueous solution of Mn2+, Ni2+, and Co2+ was prepared from MnCl2·4H2O (Sigma-Aldrich, 98%), NiCl2·6H2O (Sigma-Aldrich, 98%), and CoCl2 (Sigma-Aldrich, 97%) in a stoichiometric ratio of 1:1:1. The solution was pre-concentrated to achieve a density of 1.15 g cm−3. The solution was sprayed through a nozzle at the top into the roasting reactor chamber, which was heated by a burner (burning gases: natural gas and air) continuously so that two temperature areas were achieved (500–700 °C). The droplets descended in a swirling flow and dried up, and the solutes were pyrohydrolyzed.
The process lasted 4 hours until the pilot line was shut down and the product was removed from the outlet of the reaction chamber. Meanwhile, the hydrochloric acid was recovered as a side product from the gas phase through the exhaust outlet. The intermediate product was further lithiated with LiOH·H2O (Alfa Aesar, 98%) at higher temperature to achieve the required crystallinity and desired layered α-NaFeO2 structure. For this purpose, the intermediate product was mixed with LiOH·H2O for 3 hours in a stainless steel planetary mill. The powder mixture was then heated for 10 hours in a muffle oven under ambient conditions.
Powder characterization
For powder characterization of the obtained intermediate and final products, different measurements were performed. The techniques and corresponding equipment are summarized in Table 1.
To determine the optimal synthesis temperature of the lithiation step, a high-temperature chamber was installed into the X-ray diffractometer. The temperature chamber was set to a heating rate of 5 °C min−1 under air atmosphere. X-ray diffraction patterns of the mixture of the intermediate product and the lithium precursor were recorded at every 100 °C step during the heating process.
For structural analysis using Rietvield fit, a polynomial background, scale factor, zero point, absorption correction, coefficients for the peak shape function, and cell parameters were refined. In the starting model, 3.34% of cation mixing between Ni and Li sites was assumed (which is likely due to their similar ionic radii of 0.69 and 0.76 Å, respectively) and the basic structural parameters proposed by Nazar et al. were taken into account [27, 28] (see Table 2).
Electrochemical characterization
To prove electrochemical activity, slurries were prepared from the corresponding active material (NMC), carbon black (Super P, Timcal), and binder (polyvinylidene fluoride, PVDF, MTI) with a ratio of 80:10:10 (w/w). The active material was ball-milled in a planetary mill with the carbon black for 3 hours and added to the PVDF, which had been dissolved before in N-methyl-2-pyrrolidone (NMP, Sigma-Aldrich, 99.5%). The slurry was mixed several hours at 600 rpm in order to achieve good homogeneity. The electrode coating was performed using the doctor-blade technique. The current collector was aluminum foil (15 μm, MTI) and wet tapes had a thickness of 120 μm. The coated electrodes were further dried and hot-pressed to a final thickness of 40 μm. Electrodes with a diameter of 15 mm were cut, dried again at 120 °C for several hours under a vacuum, and transferred into an argon-filled glove box (MBraun). Half-cell measurements were performed using lithium metal as counter electrode and 1 M LiPF6 in EC/DMC 1:1 (w/w) (BASF, battery grade) as electrolyte. Glass fiber separators were used after drying (Whatman). Cyclic voltammetry measurements were performed at a scan rate of 100 μV s−1 in the potential range between 3 and 4.2 V using a BioLogic Potentiostat SP300, and the specific capacities were calculated from the derived currents of the third cycle. Type 2032 coin cells were used for cycle ability tests at 0.1 C for 45 cycles using a Maccor Series 4000 between 3 and 4.2 V.
Results and discussion
Thermogravimetric measurements
The obtained TG and differential thermal analysis (DTA) curves are presented in Fig. 1.
Three steps can clearly be distinguished on the TG curve. The first two steps correspond to the elimination of crystallized water and the last one is the oxidation of the resulting anhydrous mixed chloride. The latter process begins at 440 °C and is completed at 725 °C. Table 3 summarizes the processed thermogravimetric data.
Oxygen content x in the decomposition product NiCoMnO x equals 3.9, as determined from thermogravimetric data. Interestingly, the decomposition product NiCoMnO3.9 undergoes some structural change at 839 °C, which appears as a jump on the DTA curve; it is not, however, seen on the TG curve. The anomaly might correspond to spinel dissociation as described for other Mn-rich spinels [29].These measurements show that temperatures of at least 700 °C would be necessary for complete decomposition of Ni, Co, and Mn chlorides in a spray roasting process. The heating process during the TGA measurement is quite fast, so that lower temperatures with longer residence time might be applicable as well and reduce the energy consumption of the industrial equipment.
Synthesis
As lithium chlorides are so stable that hydrolysis into metal oxide does not occur below 2000 °C, and such temperatures are not practical for large-scale production, no lithium precursor was added in the synthesis at the pilot line. Therefore, a mixture of spinel type M3O4 (M = Ni, Mn, Co) oxides could be prepared in a first step in the reaction chamber of the pilot line. Scanning electron microscope (SEM) images of the intermediate product (see Fig. 2) show large hollow particles with an outer diameter of around 250 μm. The particles are partly cracked and might be easily destroyed through the process of post-lithiation and electrode preparation. The inner sides of the particles show cuboid-shaped crystallites with edge lengths below 10 μm.
ICP-AES measurements were performed to verify the actual composition of the intermediate product. Three measurements were performed and the average values (AV) together with the standard deviation (SDEV) are presented in Table 4.
Taking into account the standard deviations, the overall mixture seems to be homogeneous by elemental composition and there is no clear indication for enrichment of any of the transition metals in the pilot line.
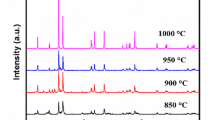
X-ray diffraction patterns from high-temperature chamber experiments were recorded at different temperatures to estimate the optimal lithiation temperature. Selected X-ray diffraction patterns are shown in Fig. 3.
It can be seen that the characteristic peaks starts to arise at around 500 °C, but the well-crystallized structure becomes visible only at temperatures around 900 °C. Therefore, this temperature was chosen for lithiation to achieve good crystallinity. Due to the evaporation of lithium around this temperature, a lithium precursor was used in excess to achieve the desired stoichiometry.
Powder characterization
SEM images of the final product are presented in Fig. 4.
The final powder has a completely different morphology compared to the intermediate product; small round particles on a nanometer scale were found instead of hollow spheres. The primary particle size seems well distributed from optical observation, having an overall similar radius. The concrete results of physico-chemical characterization of both the intermediate and the final product are presented in Table 5.
The specific surface area as well as the particle size decreased with lithiation. This is related to the breakup of the spheres which had been created within the spray roasting process. During ball milling, the particles broke, building into smaller spheres which then formed nanosized particles during lithiation.
X-ray diffraction patterns of the final product were recorded for Rietveld refinement. All diffraction peaks can be indexed on the hexagonal R-3m model. The refinement of the NMC structure gave excellent fits, as shown in Table 6, with the corresponding fitted diffraction profile in Fig. 5.
Electrochemical characterization
Cyclic voltammetry measurements of fresh electrodes showed reversible reaction with an oxidation peak at 3.8 V and a reduction peak at 3.6 V (see Fig. 6), which correspond to the Ni2+/4+ redox reactions. The specific discharge capacity was calculated with 149 mAh g−1 from the third cycle.
The cycling tests after formation showed efficiency of >99.9% within the first 45 cycles and a specific discharge capacity of 124.4 mAh g−1 in the last cycle. The specific capacities and cycle efficiencies over 45 cycles are presented in Fig. 7.
Taking into account the fact that no adjustment of the slurry preparation procedure or formula was performed, the real specific discharge capacity shows encouraging values for further attempts regarding this production route.
Conclusions
Overall battery price is strongly influenced not only by the cell chemistry but also by the industrial production route of the components. As the cathode material accounts for nearly half of the overall battery costs, the synthesis route and conditions are crucial. Therefore, a new large-scale production route was chosen for NMC synthesis in our research. The well-known process of spray pyrolysis techniques for metal oxide production and additional hydrochloric acid recovery was adopted for battery material production.
From nickel, cobalt, and manganese chloride solutions, spinel type “M3O4” was synthesized in a single step and successfully lithiated and transformed into layered NMC in a second step at 900 °C for 10 h. Cyclic voltammetry measurements showed a specific discharge capacity of 149 mAh g−1 at the third cycle. The cells showed good reversibility and long-term tests are ongoing and showed high efficiency >99.9% until the 45th cycle. At this point, it should be noticed that slurry preparation was not adjusted for the specific active material but that a generic formula was used for fast electrochemical screening. The lithiation step might be improved through lower temperatures or shorter sintering time but was not adjusted for now. The aim was to prove the feasibility of the whole process, which is currently being improved further through ongoing experiments.
Overall, the feasibility of the presented spray pyrolysis process with post-lithiation of mixed metal oxides at industrial scale was proven. The process can be performed continuously, which is a great improvement compared to conventional synthesis procedures. There are no individual batches, which reduces both processing time, energy consumption, and the number of necessary quality controls. In sum, this is expected to drive down cathode cost significantly; since cathodes account for about half of a battery’s entire cost, noticeable savings are certainly to be expected. The virtually complete recovery of hydrochloric acid, which closes the loop, is another major positive aspect of the environmental impact of the entire production process.
The electrochemical activity as well as the physico-chemical parameters is comparable to those of commercial NMC cathode materials. Improvement of the overall process through further experiments is ongoing.
References
Pillot C (2012-2025) The worldwide rechargeable battery market, 22nd edn. Presented at the BATTERIES conference in 2012 in Nice, France
Dou S (2013) Review and prospect of layered lithium nickel manganese oxide as cathode materials for Li-ion batteries. J Solid State Electrochem 17:911–926
Hsieh C-T, Hsu H-H, Mo C-Y, Chen Y-F, Pai C-T (2015) Medium-frequency induction sintering of lithium nickel cobalt manganese oxide cathode materials for lithium ion batteries. Solid State Ionics 270:39–46
Wang Z, Yin Y, Ren Y, Wang Z, Gao M, Ma T, Zhuang W, Lu S, Fan A, Amine K, Chen Z (2016) High performance lithium-manganese-rich cathode material with reduced impurities. Nano Energy 31:247–257
Zhao C, Shen Q (2014) Organic acid assisted solid-state synthesis of Li1.2Ni0.16Co0.08Mn0.56O2 nanoparticles as lithium ion battery cathodes. Curr Appl Phys 14:1849–1853
Hernández A, Fabela S, Torres-González LC, Sánchez E (2008) Preparation and electrochemical behavior of sol–gel LiNi0.3Co0.70−xMxO2 (M = Mn, Al). Ceram Int 34:225–229
Zhang W, Liu H, Hu C, Zhu X, Li Y (2008) Preparation of layered oxide Li(Co1/3Ni1/3Mn1/3)O2 via the sol-gel process. Rare Metals 27:158–164
Kiziltas-Yavuz H, Herklotz M, Hashem AM, Abuzeid HM, Schwarz B, Ehrenberg H, Mauger A, Julien CM (2013) Synthesis, structural, magnetic and electrochemical properties of LiNi1/3Mn1/3Co1/3O2 prepared by a sol–gel method using table sugar as chelating agent. Electrochim Acta 113:313–321
Ma S, Hou X, Lin Z, Huang Y, Gao Y, Hu S, Shen J (2015) One-pot facile co-precipitation synthesis of the layered Li1+x(Mn0.6Ni0.2Co0.2)1 − x O2 as cathode materials with outstanding performance for lithium-ion batteries. J Solid State Electrochem 20(1):95–103
Xiang Y, Yin Z, Li X (2014) Synthesis and characterization of manganese-, nickel-, and cobalt-containing carbonate precursors for high capacity Li-ion battery cathodes. J Solid State Electrochem 18:2123–2129
Sun H-H, Choi W, Lee JK, Oh I-H, Jung H-G (2015) Control of electrochemical properties of nickel-rich layered cathode materials for lithium ion batteries by variation of the manganese to cobalt ratio. J Power Sources 275:877–883
Yang Z, Lu J, Bian D, Zhang w, Yang X, Xia J, Chen G, Gu H, Ma G (2014) Stepwise co-precipitation to synthesize LiNi1/3Co1/3Mn1/3O2 one-dimensional hierarchical structure for lithium ion batteries. J Power Sources 272:144–151
Li S, Ren Y, Biswas P, Tse SD (2016) Flame aerosol synthesis of nanostructured materials and functional devices: Processing, modeling, and diagnostics. Prog Energy Combust Sci 55:1–59
Konstantinov K, Ng SH, Wang JZ, Wang GX, Wexler D, Liu HK (2006) Nanostructured PbO materials obtained in situ by spray solution technique for Li-ion batteries. J Power Sources 159/1:241–244
Lebl A, Andritz AG (2009) US Patent 7,597,873 B2
Zhang K, Li J, Li Q, Fang J, Zhang Z, Lai Y, Tian Y (2013) Synthesis of spherical porous carbon by spray pyrolysis and its application in Li-S batteries. J Solid State Electrochem 17:3169–3175
Lengyel M, Atlas G, Elhassid D, Luo P, Zhang X, Belharouak I, Axelbaum RL (2014) Effects of synthesis conditions on the physical and electrochemical properties of Li1.2Mn0.54Ni0.13Co0.13O2 prepared by spray pyrolysis. J Power Sources 262:286–296
Taniguchi I (2005) Powder properties of partially substituted LiMxMn2−xO4 (M = Al, Cr, Fe and Co) synthesized by ultrasonic spray pyrolysis. Mater Chem Phys 92/1:172–179
Oljaca M, Blizanac B, Pasquier AD, Sun Y, Bontchev R, Suszko A, Wall R, Koehlert K (2014) Novel Li(Ni1/3Co1/3Mn1/3)O2 cathode morphologies for high power Li-ion batteries. J Power Sources 248:729–738
Shui M, Gao S, Zheng W, Xu D, Chen L, Feng L, Ren Y (2013) LiNi1/3Co1/3Mn1/3O2 cathode materials for LIB prepared by spray pyrolysis I: the spectral, structural, and electro-chemical properties. Ionics 19/1:41–46
Kim MG, Shin HJ, Kim J-H, Park S-H, Sun Y-K (2005) XAS Investigation of Inhomogeneous Metal-Oxygen Bond Covalency in Bulk and Surface for Charge Compensation in Li-Ion Battery Cathode Li [ Ni1 ∕ 3Co1 ∕ 3Mn1 ∕ 3 ] O2 Material Batteries, Fuel Cells, and Energy Conversion. J Electrochem Soc 152/7:A1320–A1328
Park SH, Yoon CS, Kang SG, Kim H-S, Moon S-I, Sun Y-K (2004) Synthesis and structural characterization of layered Li[Ni1/3Co1/3Mn1/3]O2 cathode materials by ultrasonic spray pyrolysis method. Electrochim Acta 49/4:557–563
Harris LJF (1994) Introduction to spray roasting process for hydrochloric acid regeneration and its application to mineral processing. Hydrometallurgy 923–937. doi:10.1007/978-94-011-1214-7_62
Peek EML (1995) Chloride pyrohydrolysis, lixiviant regeneration and metal separation. Doctoral thesis, Delft University of Technology
Larson AC, Von Dreele RB (1987) Los Alamos National Laboratory report, no. LAUR-86-748.
Toby BH (2001) EXPGUI, a graphical user interface for GSAS. J Appl Crystallogr 34:210
Nazar LF, Swainson I, Rho Y-H, Yin S-C (2006) X-ray/Neutron Diffraction and Electrochemical Studies of Lithium De/Re-Intercalation in Li1-xCo1/3Ni1/3Mn1/3O2 (x = 0 → 1). Chem Mater 18:1901
Myung ST, Komaba S, Kurihara K, Hosaya K, Kumagai N, Sun Y-K, Nakai I, Yonemura M, Kamiyama T (2006) Synthesis of Li[(Ni0.5Mn0.5)1-xLix]O2 by emulsion drying method and impact of excess Li on structural and electrochemical properties. Chem Mater 18:1658–1666
Balakirev VF, Barkhatov VP, Golikov YV, Maizel SG (1999) Manganites: equilibrium and unstable states. UD RAS, Yekaterinburg in Russian
Acknowledgements
This work was financially supported by the Austrian Federal Ministry for Transport, Innovation and Technology (bmvit) and the Austrian Research Promotion Agency (FFG) through the project PyroBat, GA no. 848603.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Fröhlich, K., Legotin, E., Bärhold, F. et al. New large-scale production route for synthesis of lithium nickel manganese cobalt oxide. J Solid State Electrochem 21, 3403–3410 (2017). https://doi.org/10.1007/s10008-017-3644-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-017-3644-x