Abstract
To develop a European guideline on pharmacologic treatment of Tourette syndrome (TS) the available literature was thoroughly screened and extensively discussed by a working group of the European Society for the Study of Tourette syndrome (ESSTS). Although there are many more studies on pharmacotherapy of TS than on behavioral treatment options, only a limited number of studies meets rigorous quality criteria. Therefore, we have devised a two-stage approach. First, we present the highest level of evidence by reporting the findings of existing Cochrane reviews in this field. Subsequently, we provide the first comprehensive overview of all reports on pharmacological treatment options for TS through a MEDLINE, PubMed, and EMBASE search for all studies that document the effect of pharmacological treatment of TS and other tic disorders between 1970 and November 2010. We present a summary of the current consensus on pharmacological treatment options for TS in Europe to guide the clinician in daily practice. This summary is, however, rather a status quo of a clinically helpful but merely low evidence guideline, mainly driven by expert experience and opinion, since rigorous experimental studies are scarce.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tic disorders including Tourette syndrome (TS) are neuropsychiatric disorders with higher prevalence rates than previously thought, of up to 3–4% for chronic motor or vocal tic disorders and 1% (range 0.05–3%) for TS [196], which is the combination of chronic motor and vocal tics persisting for at least one year. The typical age of onset of tics is between 4 and 8, and tics reach their peak severity early in the second decade of life often followed by a time of remission of tics [44, 228]. Overall, TS has often a favorable prognosis: follow-up studies of TS suggest that approximately one third of children with TS are essentially symptom-free as adults; another third will have mild tics that do not require clinical attention [22]. Adults who still have symptoms severe enough to come to clinical attention are therefore unusual representatives of all subjects who have received a diagnosis of TS.
Diagnosing a tic disorder including the differentiation of tics from other movement disorders is usually a simple task (see Cath et al. this issue). It is, however, essential to detect coexisting conditions and to assess the contribution of the tics and/or coexisting conditions to the patient’s psychosocial impairment in everyday life, because the coexisting conditions often are closely related to the latter, yet they do not explain fully the level of function [95].
Indications for treatment of TS
We use the term TS in these guidelines, although information also applies to other chronic tic disorders. Decisions about treatment of TS must be based on a thorough and broad diagnostic process. It is difficult to give guidelines with regard to indications for pharmacological treatment of TS, first, because persons with TS have a high interindividual variability of symptoms, secondly, due to the temporal fluctuations of tics and thirdly, because coexisting conditions may interfere with the treatment effects for the tics. Moreover, subjective impairment does not necessarily equate objective tic severity: some individuals with relatively severe tics experience only mild impairment, whereas in other cases mild tics may be associated with significant suffering [225].
Many children and adolescents with TS do not require treatment for their tics, since their tics do not interfere with daily life or recreational activities. Indeed, only a minority of individuals with tics seek medical advice [194]. Many patients do well with a watch and wait strategy after psychoeducation and reassurance. Psychoeducation in TS has the aim to improve the tolerance for symptoms and to support stress reduction. Psychoeducation includes information about the long- and short-term variability of tics, about the natural course and about possible coexisting problems. A watch and wait strategy is also justified by the fact that we still lack evidence of the effect that pharmacological treatment of TS has on the natural long-term course and hence on the prognosis of the disorder and how this kind of treatment may influence the natural course of brain development. All pharmacological treatment options are therefore mere symptomatic treatment that alleviate, but do not cure the tics [87].
Non-pharmacologic and/or pharmacologic interventions should be considered in addition to psychoeducation for persons with clear impairment associated with the tics, either at first referral or later, due to exacerbation of symptoms. A number of reviews (e.g. [87, 246]) have published lists of indications for pharmacological treatment of tics, but none of them reflects the consensus of experts. We recommend that treatment of tics should be considered in the following circumstances, especially when persisting for some days.
Tics cause subjective discomfort (e.g. pain or injury)
Pain in TS may arise from the actual performance of frequent or intense tics causing discomfort by sudden or repeated extreme exertion (e.g. with head or neck). This kind of pain is usually musculoskeletal, although rare examples of neuropathic pain may occur. Tics can, in rare cases, cause injuries [125], e.g., a fracture line of both peroneal bones in a 13-year-old boy with TS and obsessive–compulsive disorder (OCD) admitted to hospital because of pain in his legs [80]. Striking or being struck by a moving body part involved in large amplitude tics may also cause pain and is sometimes difficult to distinguish from deliberate self-injury. Additionally, some patients obtain relief from tics while experiencing pain, to such an extent that they will deliberately provoke pain to obtain benefit [193]. A smaller number of patients complain of pain associated with the irresistible urge to tic or with aggravating premonitory urges during voluntary efforts to suppress their tics. Some patients report that tics worsen their headaches or migraines. In those cases, tic-suppressive medication could be helpful in reducing the use of pain medication and should be considered.
Tics cause sustained social problems for the patient (e.g., social isolation or bullying)
Persistent complex motor tics and loud phonic tics can cause social problems. Tics may cause isolation, bullying, or social stigmatization; loud phonic tics may result in the child being put out of the classroom. In such cases, a tic reduction, in addition to psychoeducation for the teacher, can be socially very helpful.
However, tics do not lead to social impairments in all cases. Therefore, the issue of social problems needs to be assessed carefully. For example, parents of young children are often exceedingly worried about social problems, whereas adolescents sometimes overestimate the social consequences of their tics and children in the first elementary grades are often tolerant of tics. Coexisting conditions are more often the cause than tics, if a primary school child gets socially isolated by peers [54]. In higher school classes, bullying and social stigmatization due to tics becomes more common. After proper psychoeducation, many children and adolescents will accept their tic symptoms and await the natural remission; however, sometimes medication is indicated to avoid social stigmatization.
Tics cause social and emotional problems for the patient (e.g., reactive depressive symptoms)
In addition to the aforementioned, sustained social problems, consequent to negative reactions of the social environment, some patients develop depressive and anxious symptoms, low self-esteem, and/or social withdrawal. In those cases, it is not fully clear as to what extent coexisting (sub)clinical symptomatology and self-triggered reactions cause the patients social and emotional reactions to his/her tics.
Tics cause functional interference (e.g., impairment of academic achievements)
Functional interference due to tics is relatively rare [87]. However, especially homework and falling asleep can be prolonged by bouts of tics and sleep may be disturbed followed by hypoarousal during daytime. Frequent phonic tics can impair fluency of speech and thus conversations. Moreover, children can expend mental energy in the classroom to suppress their tics, thus reducing their attention to schoolwork and interfering with their academic performance [130].
Pharmacological treatment options for TS
Pharmacotherapy has probably the fastest onset when compared with behavioral treatment options but this clinical experience has never been tested in a clinical trial. The same holds true for the efficacy of tic reduction.
Genetic studies have so far not succeeded in pinpointing a clear deviation in the biochemical pathways in patients with TS. The existing models are mainly based on the efficacy of medication rather than on rigorous and replicable models. Findings from clinical medication studies, as well as from imaging studies and human material from blood, urine, cerebrospinal fluid, and postmortem brain tissue analyses in rather small samples led to the common hypotheses on neurochemical deviances in TS [97]. Although evidence is appealing for deviances in the dopaminergic system, other imbalances, such as in the serotoninergic, noradrenergic, glutamatergic, Gamma-aminobutyric acid (GABA)-ergic, cholinergic, and opioid metabolism in TS [97, 267] seem probable. Moreover, evidence grows that those systems play interactively together, especially the dopaminergic [263] and the serotonergic [160] system.
Studies supporting the strong hypothesis of an imbalance in the dopaminergic system have shown an increased number of striatal [285] and cortical [157, 290] dopamine receptors, as well as differences in binding to dopamine transporters in the basal ganglia [42, 233, 249, 286, 287] and release of dopamine following stimulant application [250]. Therefore, modulating the dopaminergic metabolism (particularly by blocking the post-synaptic D2-receptors) is the main action of drugs used in the pharmacologic treatment of tics.
Given that only a limited number of studies on pharmacological treatment options for TS met rigorous quality criteria, we have devised a two-stage approach. First, we present the highest level of evidence by reporting the findings of existing Cochrane reviews in this field. Subsequently, we provide the first comprehensive overview of all reports on pharmacological treatment options for TS through a MEDLINE, PubMed, and EMBASE search for all studies that document the effect of pharmacological treatment of TS and other tic disorders between 1970 and November 2010. We found additional studies by going through references of each article. Given the scarcity of well-designed and well-powered studies, we think it is timely to provide such a complete overview of all available studies in order to present all facets of pharmacologic treatment accumulated over the past decades. Finally, we present a summary of the current consensus on pharmacological treatment options for TS in Europe to guide the clinician in daily practice. This summary is, however, rather a status quo combined with a clinically helpful but merely low evidence guideline and is mainly driven by expert experience and opinion, since rigorous experimental studies which would allow to better guide through well based clinical evidence are scarce.
We do not grade the studies with respect to their quality and include all available studies in view of the small base of evidence of pharmacological treatment options for TS. We present all existing studies for the different pharmacological agents, with respect to their effects on tics and other accompanying symptoms and adverse reactions or interactions with other agents.
Cochrane reviews
Although broad clinical experience guides the pharmacologic treatment of tics, the actual evidence based on randomized controlled trials (RCT) is alarmingly limited. Therefore, it is not surprising that all three existing Cochrane reviews on the pharmacologic treatment of tics in TS [51, 186, 189] came to the same conclusion, i.e., that the evidence for efficacy and safety of the studied drugs does not allow firm recommendations.
Pringsheim et al. [189] included six randomized controlled trials on pimozide in TS (total 162 participants, age range 7–53 years). Pimozide was compared to placebo and haloperidol (two trials), placebo (one trial), haloperidol (one trial), and risperidone (two trials). In summary, the six studies showed that pimozide was more effective than placebo in reducing tics. It was slightly less effective than haloperidol but showed fewer adverse reactions. The two studies that compared pimozide and risperidone revealed no important differences between these medicines for either reduction of tics or adverse reactions.
A more recent Cochrane review searched for all randomized, controlled, double-blind studies comparing atypical antipsychotics with placebo for the treatment of tics in TS [186]. However, it did not include the two above-mentioned trials because both the studies compared the atypical agent, risperidone, with an active treatment modality, without a control group that received placebo medicine. Parallel-group and crossover studies of children or adults, at any dose and for any duration, were screened. Only three randomized placebo-controlled trials, two involving risperidone and one involving ziprasidone were thus identified. Risperidone was superior to placebo in one trial although the 95% confidence intervals were large. Two trials did not detect a statistically significant difference between treatment with risperidone and with ziprasidone against placebo. Risperidone caused several extrapyramidal adverse reactions and weight gain.
The third Cochrane review on the pharmacological treatment of TS [51] analyzed the effect of Delta 9-tetrahydrocannabinol (Delta 9-THC). A total of 28 different patients included in one double blind, crossover trial and in one double blind, parallel group trial were studied. Although both trials reported a positive effect of Delta 9-THC, the improvements in tic frequency and severity were small and only apparent on selected outcome measures.
In summary, all three available Cochrane reviews urgently advocate for future trials with longer durations and larger groups to investigate the safety and efficacy of pharmacological treatment in TS. Future trials should also use the Yale Global Tic Severity Scale (YGTSS) as primary outcome measure and standardized rating scales of adverse effects, e.g. the Extrapyramidal Symptom Rating Scale (ESRS).
Complete review
Antipsychotic agents
Positive effects for D2 dopamine receptor blockers have been reported in the treatment of tics since 40 years (in average a marked decrease of tics in about 70% of cases [237]). Particularly, the blockade of striatal D2 dopamine receptors is thought to lead to reduction of tics. However, a high blockade of the receptors correlates also with the rate of unfavorable adverse reactions, such as extrapyramidal symptoms (EPS) or tardive dyskinesia (TD) [27].
Typical antipsychotics For a long time, placebo-controlled treatment studies in TS have been conducted only to prove the efficacy of the typical antipsychotics, haloperidol and pimozide. In an early randomized, double-blind, placebo-controlled crossover study, both pimozide and haloperidol significantly decreased tic frequency in nine patients with TS [206]. The results of a subsequent randomized, double-blind, placebo-controlled study of the treatment of 57 patients with TS confirmed that both haloperidol and pimozide were more effective than placebo, but haloperidol was slightly more effective than pimozide. Adverse reactions occurred more frequently with haloperidol versus placebo, but the frequency was not significantly different for haloperidol as compared with pimozide [236]. The dosages used in this study ranged from 2 to 20 mg/day for haloperidol and from 2 to 48 mg/day for pimozide. The effect of the medicine with a strong blockade of D2 dopamine receptors reduced tics in up to 80% of the cases [236]. However, in daily clinical practice, lower doses such as 1–4 mg/day for haloperidol and 2–8 mg/day for pimozide are typically used nowadays to treat TS [128, 191, 224].
In a double-blind, 24-week, placebo-controlled, randomized, double-crossover study of more commonly used doses of haloperidol (mean of 3.5 mg/day) and pimozide (mean of 3.4 mg/day) conducted with 22 subjects, aged 7–16 years, pimozide was significantly more effective than placebo in reducing tics, whereas haloperidol failed to have a significant effect. Moreover, haloperidol exhibited a threefold higher frequency of serious adverse reactions and significantly greater extrapyramidal symptoms relative to pimozide [214]. In contrast to several other studies, haloperidol was not superior to placebo, possibly due to the limited study power.
Furthermore, a long-term naturalistic follow-up study (1–15 years) of 33 TS patients treated with pimozide (2–18 mg) or haloperidol (2–15 mg) suggested benefits of pimozide over haloperidol; both drugs produced comparable relief of symptoms at follow-up; significantly, more patients on haloperidol (8 of 17) as compared with those on pimozide (1 of 13) discontinued treatment [218]. In addition, haloperidol produced significantly more acute dyskinesia/dystonia than pimozide.
A third typical antipsychotic, fluphenazine, has been used particularly in the United States for many years to treat TS, though it has merely been studied systematically. In an open-label study that included both children and adults, fluphenazine was effective at doses ranging from 2 to 15 mg/day in 17 of 21 patients [91]. In a naturalistic follow-up of 41 patients, treatment with fluphenazine for at least 1 year was safe and effective [240]. A small controlled study of fluphenazine, trifluoperazine, and haloperidol found similar reduction of tics. However, fluphenazine was better tolerated [25]; haloperidol was associated with more sedation and extrapyramidal adverse reactions.
The high frequency of drowsiness and extrapyramidal-motoric adverse reactions (dystonia, akathisia, pseudo-Parkinsonism, probably due to the strong dopaminergic blockade in the nigrostriatal pathways) limits the use of the typical antipsychotics foremost in higher doses. It has also been reported that akathisia due to antipsychotic agents may worsen the tic symptoms [280]. Moreover, several case reports raised concerns about the risk of treatment with typical antipsychotics to induce tardive dyskinesia [93, 192, 241]. Although, it is difficult to confidently quantify the rates of tardive dyskinesia owing to the limited long-term data available, the risk of this potentially debilitating and treatment-persistent adverse reaction ought to be considered in the choice of treatment [284]. This is important with greater certainty as atypical antipsychotics have shown a significantly lower risk of tardive dyskinesia [155].
Other adverse reactions, e.g., the onset of anxiety [29, 138, 154] or hyperprolactinemia with its adverse reactions, such as gynecomastia, galactorrhea, irregular menses, and sexual dysfunction [205] are more common adverse reactions than tardive dyskinesia. Additionally, during long-term medication with haloperidol, the increased appetite may result in significant weight gain [114].
Benzamides The benzamides (tiapride, sulpiride, and amisulpride) are further selective D2 dopamine receptor antagonists but in contrast to the typical antipsychotics with low (sulpiride) or as good as no (tiapride) antipsychotic action.
In addition to tiapride binding to the supersensitive D2 dopamine receptors in the ventral striatum and parts of the limbic system (Locus coeruleus), a blockade of some serotonergic receptors (5HT3, 5HT4) is assumed. Since the 1970s, there have been reports about successful treatment of TS with tiapride [61, 124, 139, 145, 183]. Several placebo-controlled studies on small sample sizes followed [43, 74]. Only one randomized, double-blind, placebo-controlled crossover study has been published with tiapride (involving 17 children), indicating a significant reduction of tic symptomatology [68]. The main adverse reactions were drowsiness, moderate transient hyperprolactinemia, and weight gain (the maximum was 10 kg during 18 months in two children). Such massive weight gain is rather the exception than the rule, because the mean weight gain was 2–4 kg [151] with the dosage range of 100–900 mg/day. Tiapride had no adverse reactions on children’s cognitive performance. Neither neurophysiological parameters such as the EEG frequency analysis and sensory-evoked potentials were affected by tiapride nor were the neurosecretory, hypothalamic-hypophyseal regulation of the sex hormones, thyroid stimulating hormone, growth hormone, or thyroid hormone impaired. This rather advantageous profile of short- and long-term adverse reactions with doses effectively reducing tics has been proven in rats too [23, 227].
Since 1970 [291], the positive effects on tics have also been reported regularly for the benzamide sulpiride [199]. It is a highly selective D2-dopamine receptor antagonist associated with less extrapyramidal and vegetative adverse reactions than haloperidol [156]. An ongoing discussion focuses on whether that medication possibly has a specific binding in mesolimbic and mesocortical systems. In addition to its mild antipsychotic potency, it has some antidepressant effect in low doses (in particular 50–200 mg daily) as well as a stimulating and anxiolytic effect [176]. In an open-label retrospective review in which 63 out of 114 patients (55%) suffering from TS had been treated with sulpiride [197], worthwhile beneficial effects occurred in 37 patients (59%). In a 14-week, randomized, double-blind, placebo-controlled crossover study trial of fluvoxamine (a specific 5HT reuptake inhibitor) versus sulpiride followed by single-blind combined therapy (4 weeks) in 11 subjects with coexisting obsessive–compulsive disorder and TS [85], sulpiride monotherapy reduced tics and non-significantly improved obsessive–compulsive symptoms. Fluvoxamine, either alone or combined with sulpiride, non-significantly ameliorated tics and reduced obsessive–compulsive symptoms. Just recently in an open-label study with 189 children and adolescents with an average age of 8 years (range 3–15 years), 6 weeks’ treatment with sulpiride improved motor as well as vocal tics. The most commonly encountered adverse reaction was sedation (reported by 16.4%) [100]. Furthermore, in patients suffering from OCD without tics, sulpiride has proven its efficacy [14, 270]. In one case of treating TS with the combination of sulpiride and imipramine, the tics increased [69]. This might be attributed most likely to the reported effects of increase of serotonin associated with increase of tics.
The main adverse reactions of sulpiride treatment are sustained sedation or drowsiness (up to 25%) and, less frequently, depression, despite its antidepressant, drive-normalizing, and mood-brightening potential [197]. Patients have also complained about restlessness and sleep disturbances [209]. Another important problem with sulpiride is a strong stimulation of prolactin-secretion causing galactorrhea/amenorrhea and a commonly observed increased appetite leading to weight gain [12, 105, 281]. Other adverse reactions occur less frequently (hypotension, rarely long-QT syndrome, dry mouth, sweating, nausea, activation or sedation, insomnia, allergic rash, or pruritus). There has only been one case report about tardive dyskinesia in an adult treated with sulpiride for tics (Eapen, Katona et al. 1993).
Successful treatment of TS disorder with amisulpride has been published only in case reports [75, 272].
Atypical antipsychotics Atypical antipsychotics are effective in the treatment of TS too. The best evidence is available for risperidone. We will herein review all atypical antipsychotics in the order of their date of FDA approval for non-TS disorders.
Clozapine, a dibenzodiazepine with 5-HT2A, 5-HT2C, 5-HT3, and weaker D1 antagonist properties, and the first FDA-approved atypical antipsychotic agent (FDA approval: 1990), has not been found to be helpful in the treatment of TS in several case reports which also documented the serious adverse reactions associated with this agent [35]. On the contrary, it is reported that clozapine exacerbates tics [13] and induces stuttering, facial tics, and myoclonic seizures [15].
The atypical antipsychotic agent best studied for the treatment of TS is risperidone (FDA approval: 1993) with a high affinity for dopamine D2- and 5-HT2-receptors. However, in several case reports and open-label studies including small groups of patients, risperidone showed similar efficacy across different ages as haloperidol and pimozide did with less frequent and less severe adverse reactions [30, 58, 86, 122, 140, 198, 219, 238, 261, 275]. The efficacy of risperidone has been confirmed in two randomized, double-blind, placebo-controlled trials involving 26 children and 8 adults with an age range 6–62 years [226], and 48 adolescents and adults between 14 and 49 years, [60], respectively, with mean daily doses of about 2.5 mg (range 1–6 mg/day). Gaffney et al. subsequently [83] compared 8 weeks’ treatment effects of risperidone with clonidine in 21 subjects with TS aged 7 to 17 years in a randomized, double-blind study. Risperidone and clonidine appeared equally effective in the treatment of tics; however, in the cases with comorbid obsessive–compulsive symptoms, risperidone was superior. The most common adverse reaction seen with both treatments was mild-to-moderate sedation, which subsequently resolved with continued administration of the medication or with a dose reduction. No clinically significant extrapyramidal symptoms were observed.
Furthermore, in a 12-week, randomized, double blind, parallel group study, both risperidone (26 patients were treated with a mean daily dose of 3.8 mg) and pimozide (24 patients were treated with a mean daily dose of 2.9 mg) reduced tics, anxiety, and depressive mood [28], whereas obsessive–compulsive symptoms improved only in the risperidone group. The latter finding is in line with the superior efficacy of risperidone for coexisting obsessive–compulsive symptoms in TS in the study of Gaffney et al. [83] as well as in an earlier case report [86]. Although the severity of extrapyramidal adverse reactions was low in both the groups, fewer patients in the risperidone group reported extrapyramidal adverse reactions (n = 4) as compared with the pimozide group (n = 8). Depression, fatigue, and somnolence were reported as the most prominent adverse reactions in both treatment groups. This is in line with a retrospective study carried out on 58 adult and adolescent TS patients who were treated with risperidone; 17 patients (29.3%) developed a major depressive disorder, including 1 patient who later committed suicide, and 13 patients (22.4%) became dysphoric while taking risperidone [143]. In a randomized, double blind, crossover study of 19 TS children (ages, 7–17 years), who underwent a 4-week treatment with pimozide or risperidone, followed by the alternative treatment after a 2-week placebo washout, risperidone was more effective than pimozide in reducing tics, in contrast to Bruggeman et al.’s report [28], which suggested that risperidone and pimozide were equally efficacious in the treatment of TS.28 Risperidone, however, was associated with more weight gain during the 4-week treatment periods. No serious adverse reactions were reported [88].
Risperidone also appears to be effective in treating aggressive behavior in patients with TS. In a retrospective chart review of 28 children and adolescents (one female) aged 5–18 years with TS and aggression problems, 22 (78.5%) showed both decreased aggression scores and tic reduction when treated with a mean daily dose of 2 mg risperidone [219]. This is in accordance with the potential of risperidone to manage pediatric aggression in other disorders [177]. Moreover, positive effects of risperidone not only on tics but also on sleep disturbances have been reported in the case of a 12-year-old boy with no previous psychopharmacological treatment [7].
Finally, in line with the other agents, the problem of causality between treatment and the natural course of tic symptomatology has also been mentioned for risperidone leading to one report about induction of tics by risperidone [72].
Several case reports [17, 18, 117, 141] and open-label studies [33, 126, 148, 262] have suggested efficacy of olanzapine (FDA approval: 1996) in the treatment of TS in adolescents and adults during the last 10–15 years. In four patients with severe TS (aged 19–40 years), a 52-week, double blind, crossover study with olanzapine (5 and 10 mg daily) versus low-dose pimozide (2 and 4 mg daily) was performed [171]. The reduction in tic severity was highly significant with 10 mg olanzapine versus baseline and versus 2 mg pimozide, and was significant for 5 mg olanzapine versus 4 mg pimozide. Only moderate sedation was reported by one patient during olanzapine treatment, whereas three patients complained of minor motor adverse reactions and sedation during pimozide treatment. All patients opted for olanzapine treatment at the end of the study. Compared to other antipsychotics, olanzapine has a greater activity at serotonin 5-HT2 receptors than at D2 dopamine receptors. This may explain the lack of extrapyramidal effects. Additionally, olanzapine does not appear to block dopamine within the tubero-infundibular tract, explaining the lower incidence of hyperprolactinemia than with typical antipsychotic agents or risperidone. Nevertheless, the most widely reported adverse reactions were drowsiness/sedation and increased appetite frequently followed by weight gain [148]. In this context also metabolic adverse reactions (glucose and lipid metabolism) arise [184], although there seems to be no correlation between weight gain and metabolic disturbances [153].
Quetiapine (FDA approval: 1997) with its greater affinity for 5-HT2 receptors than for dopamine D2 receptors has shown its efficacy in reducing tics in two children with TS [179, 181, 182]. In an open-label trial with 12 subjects with a mean age of 11.4 ±2.4 years quetiapine reduced tics significantly [159]. Three subjects complained of sedation in the first week of treatment, but in the 8 weeks under investigation patients did not experience extrapyramidal adverse reactions and no statistically significant weight gain. Contrarily, in a retrospective study with longer observation period and higher dosage (175.0 SD 116.8 mg/day) of quetiapine the only noteworthy adverse reaction was weight increase. Quetiapine reduced tics also significantly in an open label study of 12 patients aged 8–18 years with TS [48]. Routine laboratory parameters and serum prolactin level were all normal and did not change throughout treatment.
Although there has been great hope for ziprasidone (FDA approval: 2001) as a potent treatment option in TS without the problem of weight gain [6], only one randomized, double blind, placebo-controlled study in 28 children and adolescents (7–17 years) [212] and one open open-label study in 24 children and adolescents (7–16 years) so far has proved this expectation [211, 213]. A mean daily dose of 28.2 mg ziprasidone reduced tics more effectively than placebo. Mild transient somnolence was the most common adverse reaction of low-dose exposure (5–20 mg/day), consistent with what is seen in clinical practice. This may be caused by enhanced 5-HT2C antagonistic activity of ziprasidone at low doses [260]. No patient experienced extrapyramidal symptoms, akathisia, or tardive dyskinesia, although administration of a single, low dose of ziprasidone may not be reflective of either higher doses or long-term risk in a naturalistic treatment setting [213]. In addition, there was no weight gain and changes of the analyzed laboratory parameters except of prolactin. Although QT prolongation has been discussed prominently in ziprasidone, a single dose of ziprasidone to treat TS was well tolerated without clinically significant effects on electrocardiograms collected around the time of maximum serum concentration [213] and even in higher doses no elevated risk of QT prolongation has been reported compared to other antipsychotics [266].
In addition to ziprasidone also aripiprazole (FDA approval: 2002) induced no weight gain during an 8-week, open-label trial with a flexible dosing strategy of aripiprazole in 72 children and adolescents with TS aged 6–18 years [49]. In a 10 week open-label, flexible-dose study with eleven subjects (10 males) with TS (age 9–19 years) who had not responded to or had not tolerated previous tic medication, effects of aripiprazole were promising [142], albeit with some weight gain in five patients. Finally, in an open-label, flexible-dose study including sixteen children (15 males) aged 8–17 years there was a mean increase of 2.3 kg after a 6-week trial with aripiprazole [167]. It provides a high affinity at dopamine D2 receptors but acts in contrast to other atypical antipsychotics as a partial agonist. Under treatment of clinical useful doses (10–30 mg/day) aripiprazole exhibits D2-receptor binding of 80–100% [96]. However, while binding at the active state of D2-receptors, aripiprazole shows 30% agonistic activity compared to dopamine [34]. Aripiprazole also acts as a partial agonist at 5-HT1A receptors and as a potent antagonist at 5-HT2A receptors [113]. This profile raised the hope that aripiprazole might be superior to previous pharmacological treatment options even in refractory cases. Excellent efficacy in the treatment of tics has been reported in a total of 201 cases, at least 31 of them adults [31, 47, 49, 52, 55, 63, 76, 103, 106, 118, 119, 142, 158, 166, 167, 173, 265, 288, 289]. A randomized, double blind, placebo-controlled study is, however, still lacking. Nevertheless, this drug should be considered because of its promising perspective based on actual clinical experiences. Even in “refractory” TS, aripiprazole has shown about 75% reduction of severe coprolalia in a 28-year-old man [16] as well as good efficacy in treating TS and coexisting OCD in an adult female [283]. Accordingly, Budman et al. [32] found in their retrospective, observational study of 37 children and adolescents with TS who were refractory to previous treatment that aripiprazole still reduced tics as well as explosive outbursts in these patients. Aripiprazole was tolerated reasonably well, although 8/37 (22%) children discontinued treatment; most common adverse reactions included weight gain, akathisia, and sedation at a mean daily dose of 12.3 (SD 7.50) mg in the 29 subjects who completed the study. In a 12-week, open-label trial with flexible dosing strategy aripiprazole revealed a good tic reduction in 15 participants, aged 7–19 years. Nausea and sedation were the most commonly reported adverse reactions that ameliorated in all participants within 2 weeks, with the exception of 1 participant who had continuously complained of sedation, but did not stop taking the drug [232]. The mean weight gain during this study was negligible.
For the newest atypical antipsychotic paliperidone (FDA approval: 2006) as well as for sertindole (not approved by the FDA for use in the USA) no data on the treatment of tics have been published.
Noradrenergic agents
In general, noradrenergic agents (clonidine, guanfacine, and atomoxetine) are mostly used in children and adolescents with a combination of attention-deficit/hyperactivity disorder (ADHD) and mild tics given their efficacy in treating ADHD symptoms in addition to tics [11]. Their tic-suppressing effects seem to be generally smaller, however, than those of antipsychotic agents.
Despite the frequent use of the α-2 adrenergic agonist clonidine for nearly three decades in the treatment of TS, controlled studies with clonidine are few in number. It is used more commonly in America than in Europe [195]. Case reports of clonidine’s efficacy in treating TS appeared in the early 1980s [150] and open-label trial evidence has been contradictory [45, 46, 234, 248]. A single-blind, placebo-controlled trial demonstrated a significant improvement in 6 out of 13 patients [133]. A randomized, placebo-controlled trial on 47 patients (7–48 years old) suffering from TS showed that treatment with clonidine reduced tic severity and frequency better than placebo [134], whereas another randomized, placebo-controlled study in 30 children and adults with TS found no difference [92]. A randomized, double blind, placebo-controlled study of desipramine and clonidine for the treatment of ADHD in TS revealed that clonidine did not alter tic severity in 34 children aged 7–13 years [247]. However, in the largest well-designed, randomized trial on orally administered clonidine, which included a placebo group, clonidine reduced tics significantly [271].
A transdermal clonidine preparation is also available and has been tested for the first time in nine patients in a placebo-controlled crossover trial. Although no objective improvement was recorded, most subjects felt they had improved [84]. A recent randomized, double blind, placebo-controlled multicentre trial using a clonidine adhesive patch revealed in the randomly assigned clonidine group (n = 326) a significant improvement of TS in 68.85% compared to 46.85% in the clinical control group (n = 111) [62]. Accordingly, clonidine transdermal patch treatment was effective in 53 out of 65 children with TS [116].
Adverse reactions of clonidine include sedation, dry mouth, headache, irritability, and midsleep awakening [62]. Blood pressure and pulse should be measured at baseline and monitored during dose adjustment. Specific guidelines for blood pressure monitoring during follow-up have not been established but regular monitoring of pulse and blood pressure changes, and symptoms suggestive of cardiovascular problems (e.g., exercise intolerance, dizziness, syncope) is recommended [53]. Baseline and follow-up electrocardiograms have been recommended in some practice guidelines [64], but not in others [53]. Although blood pressure is generally not a problem with clonidine, patients and families should be educated about the possibility of rebound hypertension, tics, and anxiety with abrupt discontinuation [19]. Although many authors report that the adverse reactions tend to be mild and transient, this view is not fully supported by others [89, 99, 137] especially when moderate to severe tics require higher dosage.
Guanfacine, another α-2 adrenergic agonist, has modest efficacy in reducing tics and in improving attention in children and adolescents. An open-label study of guanfacine in 10 children with TS [40] and in 25 medication-free children (23 males and 2 females) [24] with TS + ADHD aged 7–16 years revealed a significant decrease in tic severity and improvement in attention. In addition, a case report had described a 6-year-old boy with TS treated successfully with guanfacine [77]. These open label observations were confirmed by a randomized placebo-controlled double-blind trial in 34 children with TS + ADHD with a mean age of 10.4 years [223]. In contrast, in another double blind, placebo-controlled study on 24 children with TS aged 6–16 years guanfacine was not superior to placebo [50]. In summary, whether guanfacine would be effective for the treatment of moderate to severe tics remains unanswered [225]. In addition, the suggestion that guanfacine is a better tolerated alternative to clonidine remains unclear without a direct comparison study [217].
The most common adverse reactions of guanfacine are somnolence, headache, fatigue, sedation, dizziness, irritability, upper abdominal pain, and nausea. Somnolence, sedation, and fatigue adverse reactions emerge within the first 2 weeks of dosing and generally remit [210]. There is a concern that guanfacine has a propensity to induce mania in children with a personal or family history of bipolar disorder [102] as well as syncopal episodes probably due to drug-induced hypotension or bradycardia [123]. Guanfacine approved to treat hypertension in several European countries has been withdrawn from the market in several European countries probably due to lack of financial success.
The selective noradrenaline reuptake inhibitor atomoxetine had already been shown to be effective in randomized, placebo-controlled trials for treating ADHD in children [41]. Also in the treatment of ADHD with coexisting tics its efficacy was tested in a large, industry-sponsored multicenter study in 148 children [5]. Atomoxetine reduced both tics and ADHD symptoms in the study’s subgroup suffering from ADHD + TS [256]. Significant increases of mean pulse rate and rates of treatment-emergent nausea, decreased appetite, and decreased body weight were observed during medication with atomexetine. Concerns were raised, however, that children with severe ADHD or tics might have been unlikely to be enrolled in the study [87] which had a fairly high dropout rate in both treated (34%) and untreated (26%) groups during the double-blind portion of the trial. Moreover, case studies describe patients experiencing manifestation, recurrences, or exacerbation of tics following treatment with atomoxetine [136, 178, 180, 230].
Alternatives
Tetrabenazine, a vesicular monoamine transporter type 2 inhibitor, depletes presynaptic dopamine and serotonin stores and blocks postsynaptic dopamine receptors. In view of the hypothesized supersensitivity of dopaminergic receptors thought to be responsible for the tics in TS [231], tetrabenazine might be an alternative to antipsychotic treatment. Its divergent mechanism of action might result in different efficacy and adverse reactions profiles than the treatment with antipsychotics [109]. In some clinical studies on hyperkinetic movement disorders, including patients or samples with TS, tetrabenazine has shown its potential to ameliorate tics [108, 109, 111, 112, 174, 268, 278]. Results of two retrospective chart reviews enrolling only patients with TS (n = 77; mean age about 15 years; [120] and [188]) showed that 18–24 months’ treatment with tetrabenazine resulted in a moderate to marked improvement in functioning and TS-related symptoms in over 80% of patients. Adverse reactions included drowsiness/fatigue (36.4%), nausea (10.4%), depression (9.1%), insomnia (7.8%), and akathisia/parkinsonism (6.5%), but these symptoms improved with reduction in dosage [120]. Weight gain was less pronounced in doses of comparable efficacy than under treatment with antipsychotics and most patients who switched from an antipsychotic drug to tetrabenazine subsequently lost weight [170]. There were no reports of tardive dystonia or serious adverse reactions. In contrast there is a report about two patients with TS who developed tardive dystonia after treatment with antipsychotic agents. The dystonic movements persisted after the offending drugs were stopped and improved with tetrabenazine [252]. In summary, these findings encourage to conduct further studies.
Findings from preclinical studies in animals have suggested that nicotine might potentiate the effect of antipsychotic agents used to treat TS. Indeed, in 2 case reports negative effects of smoking cessation on TS have been reported [57, 59]. In initial open-label studies, chewing nicotine gum in addition to treatment with antipsychotics reduced tics in frequency and severity and improved concentration and attention [146, 216]. Similar effects were observed in a subsequent controlled trial involving nicotine gum plus haloperidol. Only in the group chewing the nicotine gum, tic frequency was reduced, while placebo gum alone had no effect on tic symptoms [147]. However, the short duration of effects as well as the bitter taste and gastrointestinal adverse reactions limit the compliance. Similar findings have been reported for application of transdermal nicotine patches to potentiate haloperidol in TS [242, 243]. In 11 poor-responders to antipsychotic treatment of TS, transdermal nicotine patches delivering 7 mg of nicotine in 24 h reduced tics 47% in frequency and 34% in severity [244]. In two of these patients tic reduction lasted even after removal of the transdermal nicotine patches. This result was in line with similar reports on tic reduction longer than 4 weeks after 48 h of nicotine administration by a transdermal patch [66, 67]. Correspondingly, retrospective case studies also found that application of a single transdermal nicotine patch delivering about 7 mg nicotine in 24 h resulted in a significant tic reduction for a mean of 10 days [239, 243]. The participants complained, however, about nausea and occasional headache and sedation. In the first randomized, double-blind study 70 patients with TS were treated with either transdermal nicotine (7 mg/24 h) or placebo patches in addition to their individual optimal dose of haloperidol [245]. In the patients who completed all 19 days of nicotine (n = 27) or placebo (n = 29), improvement of emotional and behavioral symptoms but also adverse reactions such as nausea and vomiting were more frequent under nicotine treatment. A subsequent randomized, double blind, placebo-controlled trial examined the acute (4 h) and sustained (2 weeks) effects of a single dose of transdermal nicotine on clinical (i.e., tics), attentional (continuous performance task, event-related potential, patient and parental reports), and behavioral symptoms in 23 children and adolescents with TS receiving treatment with antipsychotic agents. In the 14 evaluable patients with complete primary efficacy data, nicotine (compared to placebo) failed to alter symptoms at 4 h but counteracted ERP-P300 signs of diminished attention seen 2 weeks following placebo treatment. Secondary efficacy measures, including patient self-reports and parental ratings, found nicotine to reduce complex tics and improve behaviors related to inattention [104]. One study investigated neurophysiological mechanisms possibly underlying nicotine treatment of TS by using transcranial magnetic stimulation (TMS). A single dose of nicotine in 10 non-smoking and non-treated adults with TS reduced tic severity as assessed by blind video scoring in the majority of patients. In addition, nicotine abolished the reduced inhibition in patients compared to controls [172].
Tetrahydrocannabinol (THC) has been suggested to be effective and safe in the treatment of tics [162–164] without influence on neuropsychological performance [161]. This knowledge is based on a randomized, double blind, placebo-controlled study in which 24 adult patients with TS were treated over a 6-week period with up to 10 mg THC/day. No serious adverse reaction occurred and the reported mild adverse reactions were dizziness, tiredness, and dry mouth. Hasan et al. [98] reported about a 15-year-old boy with treatment refractory TS plus ADHD leading to severe physical and psychosocial impairment. For the first time after several years of unsuccessful medication even with a combination of different agents, the administration of THC improved tics considerably without adverse reactions, allowing parallel stimulant treatment of coexisting ADHD. Along with the THC treatment, TMS measured cortical inhibition was increased.
In addition to the use of pharmacological treatment options with systemic effects, there is increasing evidence for the efficacy of botulinum toxin injections to treat persistent well-localized (non-complex) motor and, sometimes, vocal tics by temporarily weakening the associated muscles. Initially, botulinum toxin injection was used for selected severe cases [3, 107, 125, 229]. Other case reports and case series followed also including children after the age of 8 years [4, 131, 215, 257, 273, 279]. In 35 of 186 patients, botulinum toxin injections were effectively controlling motor tics [8]. The effect on vocal tics was minimal. Adverse reactions included temporary soreness and mild muscle weakness. In 30 patients with vocal tics assessment after 15 days and then 4 times over a 12-month period botulinum toxin injection improved vocal tics in 93% of patients, with 50% being tic-free [187]. Mean response time was 5.8 days and mean duration of response was 102 days. Quality of life improved and premonitory experiences dropped from 53 to 20%. Hypophonia was the only adverse reaction of note (80% of patients). Just recently, the positive short-term and long-term (up to 10 years) treatment effects of botulinum toxin injections every 3 months on simple motor tics of 15 patients (mean age 43 years; range 18–84) could be shown [190]. Marras et al. [144] concluded from their randomized, double blind, controlled clinical trial that the treated tic frequency as well as the urge associated with the treated tic were reduced by botulinum toxin injection. Still, the patients’ subjective perception was that overall this treatment did not improve their condition. This is perhaps due to the fact that only selected subset of tics could be treated in each patient.
The dopamine autoagonist talipexole with putative preferential activity on presynaptic dopamine receptors was investigated one time in a randomized, double blind, placebo-controlled study [90]. In 13 adult men with TS, talipexole was poorly tolerated because of clinically significant sedation and dizziness. Tics did not improve at tolerable doses. These findings suggest that talipexole has no role in the regular management of tic disorders.
Clonazepam, a benzodiazepine which acts primarily on the GABAergic system, has a long history in the treatment of TS with dosages up to 6 mg/day [89]. Although there have been no placebo-controlled trials in TS, open-label studies have been carried out in adults [94, 274] and adolescents with TS [115, 264]. In a single-blind comparison with clonidine in 20 children, clonazepam was superior in suppressing tics [61]. In a single-blind clinical study of 20 patients with TS, those with high red blood cell-to-plasma choline ratios responded better to clonazepam than to haloperidol [152]. As with all benzodiazepines, tolerance and adverse reactions including sedation, short-term memory problems, ataxia, and paradoxic disinhibition often limit the use of clonazepam [89]. There are no data on other benzodiazepines except a case report about the therapeutic effect of low-dosage diazepam on facial tics in children [78].
The GABA B receptor agonist baclofen, which is used for the treatment of spasticity, has been examined in an open-label study in a large cohort of children with TS [8]. 250 of 264 patients on baclofen treatment experienced a significant decrease in the severity of tics. A small randomized, double blind, placebo-controlled study of baclofen in 10 children was inconclusive because there was a reduction in overall impairment but no changes in tic frequency or severity [251]. The results of these studies provide only modest support for the use of baclofen in TS. Common adverse reactions were sedation and drowsiness.
Other GABAergic drugs including the anticonvulsant levetiracetam have shown tic reduction in open studies on TS [9, 71]. Adverse reactions, however, as well as the finding that levetiracetam did not change the mean total YGTSS and Clinical Global Impression score in a small randomized, double blind, crossover study (n = 10) [99] as well as in a randomized, double blind, placebo-controlled, crossover trial in 22 children with TS (mean age 12.2 years) [253] question its usefulness in the treatment of TS.
Topiramate reduced tics in a small randomized, double-blind study on 20 patients of a broad age range (7–65 years) compared to placebo [110]. This is in line with a chart review on 41 patients with TS [127] as well as a previous report on two patients with TS who were successfully treated with topiramate while previous medications were tapered and discontinued during the first 2 weeks of treatment [1].
Lithium has been used successfully to reduce tics in five of ten children and adolescents [121], a 22-year-old male [277], and three adolescents suffering from TS who had been initially treated with haloperidol [70]. Failure has also been described, though [26], and firmer evidence is lacking.
Several case reports [81, 220–222] and a randomized, double-blind, placebo-controlled study involving 10 adults with TS suggest that tic reduction may be achieved with naloxone [129], an opioid receptor antagonist. Some studies indicated that difference in response to naloxone in TS subjects may be based on a dose–response effect [38, 276].
Some attention has also been given to the use of treatments that include a modulation of the body’s autoimmune-response. In children fulfilling criteria for pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections (PANDAS; a subgroup of children with OCD and/or tic disorder that experience symptom exacerbations following streptococcal infections), plasma exchange and intravenous immunoglobulin (IVIG) were both effective in lessening of symptoms [185, 293], although benefits through IVIG could not be confirmed in unselected patients with a tic disorder [101]. In a small prospective study, antibiotic prophylaxis with penicillin or azithromycin administered for 12 months in children fulfilling PANDAS criteria was associated with significant decreases in neuropsychiatric exacerbations [254]. A case study of a patient with TS reported benefits of treatment with celecoxib, a COX-2 inhibitor [165].
Finally, a wide range of further neuroactive agents have been examined non-systematically with divergent results concerning their efficacy in the treatment of TS. For example buspirone [65], carbamazepine [168, 292], metoclopramide [2, 169], physostigmine [258, 259], and spiradoline mesylate [39] have received some attention. A comprehensive overview of other case reports and non-blinded trials can be found elsewhere [195].
Treatment of tics in the context of comorbidities
Children and adolescents with TS are frequently affected by coexisting psychiatric conditions [79], which may be regarded the rule rather than the exception. In clinical samples of TS about half of the cases also meet criteria for ADHD and vice versa, TS is present in about 20% of children with ADHD [208, 228]. This co-occurrence of TS and ADHD is in most cases associated with a higher psychopathological, social, and academic impairment resulting from the negative impact of ADHD [10, 95, 200–202]. Besides, patients with TS also suffer more frequently from obsessive–compulsive symptoms or disorder (about 50%). Especially the need to achieve a ‘‘just right’’ feeling in TS has to be seen as an indicator for a continuum between TS and OCD [203].
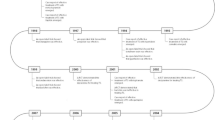
Coexisting disorders cause often more clinical impairment and may be more responsive to treatment than the tics themselves [19]. It is therefore crucial to select an appropriate treatment goal (tics or coexisting conditions), when deciding on treatment options. Treatment of tics and coexisting conditions should be prioritized according to the impairment caused by each problem (for a decision tree see Fig. 1). Thus, in many cases not the tics, but coexisting problems require treatment e.g. ADHD or OCD. Clinicians should thus avoid to start two medications simultaneously, for instance one for tics and one for ADHD symptoms. Primary treatment of a coexisting condition, such as ADHD may reduce stress and improve attentional resources, and sometimes reduce tics by enhancing the individual’s ability of tic suppression.
Decision tree for the treatment of tic disorders including Tourette syndrome. Indications for treatment are given in “Tics cause subjective discomfort (e.g. pain or injury)”, “Tics cause sustained social problems for the patient (e.g. social isolation or bullying)”, “Tics cause social and emotional problems for the patient (e.g. reactive depressive symptoms)” and “Tics cause functional interference (e.g. impairment of academic achievements)”. Solid arrow next level of evaluation/treatment, dashed-dotted arrow monitoring after successful treatment, dashed arrow alternating between two treatment options. Note: patient preference (after psychoeducation) and availability of therapists have to be considered in the choice of treatment. DBS deep brain stimulation, THC Tetrahydrocannabinol
Treatment algorithms of coexisting conditions in the context of TS are similar to treatment of these conditions without the presence of TS. Well-designed controlled clinical trials have not indicated a deterioration of tics in persons treated with stimulants [21] nor induction of first tics by stimulant treatment even in children at risk [175, 204].
Long-term treatment with methylphenidate (MPH) is not associated with increases in tic severity. In a two year prospective, open label study in which effects of MPH treatment were evaluated in 34 prepubertal children with ADHD and with chronic multiple tic disorder, the authors found no evidence that motor or vocal tics changed in frequency or severity during the MPH maintenance therapy, whereas initial behavioral improvements were maintained [82]. In a subsequent blinded placebo-controlled discontinuation trial in 19 children with ADHD and with chronic tic disorder who had received psychostimulants for a minimum of one year, tics did not change in their frequency or severity of motor or vocal tics during the maintenance dose of stimulant medication compared with the placebo condition. Treatment with the maintenance dose was, however, associated with behavioral improvement in ADHD symptoms, indicating continued efficacy. These studies prove that neither treatment nor discontinuation of treatment with MPH in patients with tics lead to an exacerbation of tics. Thus, abrupt withdrawal of stimulant medication in children receiving long-term maintenance therapy does not appear to result in worsening of tic frequency or severity.
Higher doses of stimulants, in the range of 45 mg b.i.d. of MPH and 22.5 mg b.i.d. of dexamphetamine, however, may still lead to (reversible) tic exacerbations [36]. Thus, in general, stimulants may be safely used in children with TS and ADHD, when using doses based on the typical clinical titration procedure [21]. Other treatment options for ADHD in the context of TS include clonidine [271], atomoxetine [5, 256], and desipramine [255].
Coexisting OCD in patients with TS may be less responsive to serotonin reuptake inhibitor monotherapy compared to OCD in patients without tics [149]. Co-administration of an antipsychotic agent may be helpful [20, 56].
Problems with clinical recommendations for the pharmacological treatment of TS
Unfortunately, there has not been great improvement in evidence concerning the pharmacological treatment of TS since the overview of Robertson and Stern [199] who stated that “the treatment of the Gilles de la Tourette syndrome has evolved from case reports, clinical experience and more recently blinded trials usually in small numbers of patients”. Ideally, according to the principles of evidence-based medicine to be recommended, an agent must have shown its efficacy in randomized, double-blind, placebo-controlled studies. However, even today, evidence for efficacy of many agents that might be considered in the pharmacological treatment of TS is often based on open studies or randomized, double-blind, placebo-controlled studies with quite small sample sizes [199]. Hence, there exists only one drug which has been approved for TS widely in Europe, which is haloperidol. However, because of its adverse reactions it is nowadays usually a drug of third line in clinical practice.
Particularly there is not a sufficient number of randomized, double-blind trials that have directly compared different pharmacological treatment options of TS including a placebo group [206, 214, 236]. Moreover, the heterogeneity of tic disorders with regard to the severity, frequency, localization, complexity of the tics as well as with regard to patterns of comorbidity demands further investigation in terms of the identification of factors that may predict or moderate response to different psychopharmacological agents [199]. Knowledge in this area could help clinicians to reach a more tailored choice of treatments. Currently, we have no data with regard to response to a second medication in patients who did not respond favorably to a first line agent. That is, for example, in patients who have not responded to risperidone, we do not have scientific data from trials whether response may be still expected from another antipsychotic, or rather from a different type of medication. Finally, durations of existing studies have not always taken into account the natural waxing and waning of tics (see Fig. 2). This calls for longer observation periods and better rating instruments than those of most existing studies. Investigations of long-term efficacy and adverse reactions are completely lacking. Nevertheless, the treating physician should be aware of the side effects profile of the drug in question and initiate adequate and suitable clinical and laboratory controls.
Evaluation of treatment efficacy in TS in light of natural waxing and waning. At date 1 a therapeutic intervention could be followed by tic reduction despite of its potential to increase tics or without an effect on tics. This has to be ascribed not to causal mechanisms of the intervention but to the natural waxing and waning of the tics. Correspondingly, a therapeutic intervention at date 2 could be followed by an increase of TS symptomatology despite its potential to reduce tics. The therapeutic intervention might attenuate the natural waxing of the tics. Conclusion: Meaningful appraisal of treatment efficacy in TS can only be given in most cases after longer time
Moreover, studies comparing the effectiveness of behavioral and pharmacological treatments in patients with TS are absent. Thus, currently no scientific data are available indicating whether behavioral treatment or medication should generally be tried first. An advantage of behavioral treatments may be its better long term effects, beyond the duration of the therapy, as well as their assumed less frequent and less severe adverse reactions. However, behavioral treatments require sufficient motivation and certain ability for introspection, which may limit its usefulness somewhat in younger patients (see also Verdellen et al., this issue). Patients’ treatment preference after thorough psychoeducation is an important aspect in deciding between medication and behavioral therapy. Definitely, pharmacologic treatment should be initiated if behavioral treatment reveals insufficient success. Conversely, drug-treated patients who do not experience sufficient tic reduction and/or suffer from non-tolerable adverse reactions may be stimulated to (re-)start behavioral interventions. In the rare cases of adults, who have extremely impairing tics that are not sufficiently alleviated through several pharmacological treatment options one should consider deep brain stimulation (see Mueller-Vahl et al., this issue).
Assessing response to treatment
The clinician should inform the patient and their parents that the goal of a pharmacological treatment of TS is not to completely eliminate the tics, but to achieve a reduction aimed at eliminating the psychosocial impairment caused by the tics. Unrealistic expectations on the efficacy of pharmacological treatment of TS will lead to frustration for the child, family, and physician. Also, the desire to completely suppress tics can lead to overmedication and adverse reactions that cause more problems than the tics themselves. A common example of this is the overtreating of children to the point of excessive daytime sedation or unhealthy weight gain. Families should be informed that medication typically only results in a 25 to 50% reduction in tic symptoms.
Also, clinicians should always be aware of the natural waxing and waning of tics in TS when evaluating effects of treatments (see Fig. 2). It is advisable to consistently use formal tic severity rating scales to more objectively assess responses to treatment over time. Perhaps the most suitable instrument is the YGTSS, a semi-structured interview which records the number, frequency, intensity, complexity, and interference of motor and vocal tics separately [135]. But also the Tourette Syndrome Severity Scale (TSSS) developed by Shapiro et al. [235], which is shorter and more easy to use can be recommended.
What specific agents can be recommended?
As previously stated, there is a great scarcity of studies directly comparing efficacy and safety of different psychopharmacological agents, foremost with regard to longer term effects. Therefore every general recommendation depends heavily on the experts’ own experiences and preferences.
After reviewing the existing literature, it appears that the best evidence arising from randomized, double-blind, placebo-controlled studies is still available for the typical antipsychotics haloperidol and pimozide, with some indications that pimozide may be more effective and may have a somewhat more favorable adverse reaction profile than haloperidol [189], with exception of its potential cardiac effect. In clinical practice in Europe, however, over the last years haloperidol and pimozide have been replaced stepwise by atypical antipsychotics. Here, the best evidence is undoubtedly available for risperidone [186, 189]. This is also the agent that has been studied best. A lower risk for adverse reactions compared to typical psychotics is assumed in clinical use. Still many adverse reactions, however, are similar to those associated with the use of typical antipsychotics, including sedation, akathisia, weight gain, extrapyramidal symptoms (EPS), neuromalignant syndrome, and tardive dyskinesia. Although atypical antipsychotics generally are associated with a lower incidence of EPS in youth [269], a rapid dose escalation is actually associated with higher risk of EPS [37]. In addition, longer experience with atypical antipsychotics reveals that new risks need to be considered, such as metabolic syndromes and QTc prolongation. The incidence of these risks in patients suffering from TS, especially in children and adolescents, cannot be easily predicted due to the paucity of long-term studies in this population.
The choice of pharmacological treatments is not only based on the efficacy and the rate of adverse reactions but also on the potential to show efficacy in refractory cases. In particular, aripiprazole is rather promising, given the lower probability of weight gain as adverse reaction and promising effects in patients who had not responded to previous treatments. Placebo-controlled studies with aripripazole are still missing, however.
Availability of clinical experience with agents also plays an important role in the choice of recommendable treatments. In the German-speaking world the benzamides, such as tiapride and sulpiride are commonly used as first line agents to treat TS particularly in children and adolescents. Indeed, tiapride is regarded as the medication of first choice in the German guidelines for the treatment of tic disorders without coexisting significant emotional/obsessive–compulsive symptoms [207], Tiapride and sulpiride are not available in the United States. This explains why these agents are not mentioned in reviews from US authors [87] and why their clinical efficacy in TS as well as their pharmacological properties have been underinvestigated in comparison to other antipsychotic compounds. This small base of evidence notwithstanding, Robertson and Stern [199] conclude in their review that tiapride and sulpiride are highly recommendable to treat TS in view of their excellent balance of efficacy and tolerability proven over decades in clinical practice.
Further, severity of tics and presence of comorbidity may affect choices of treatments. Although the evidence in favor of the tic-suppressing effects of clonidine may be less robust compared to the antipsychotics, clonidine may actually improve ADHD symptoms alongside with suppression of especially mild-to-moderate tics. In addition, clonidine tends to alleviate initial insomnia and reduce anxiety [217].
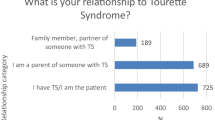
An important consideration, given the relative lack of controlled clinical studies, is the opinion of experts. Therefore, we sent by email a questionnaire to members of the European Society for the Study of TS (ESSTS). All clinicians with ample experience in the treatment of TS were asked what psychopharmacological agent they would consider first, second, third, and subsequent choices in the treatment of tics (provided there would be no contra-indication for any of the available agents, and there would be no comorbidity). We received 22 responses out of the 60 members. We rated each first choice agent with 4 points, a second choice agent with 3 points, a third-choice agent with 2 points, and additional agents with 1 point. As listed in Table 1, most support from the experts has been provided for risperidone, with considerable support for clonidine, aripiprazole, and pimozide as well (Table 2).
Based on the available evidence, experience with the drug, and experts’ preference, risperidone can be recommended as a first choice agent for the treatment of tics. Adverse reactions form the biggest limitation of risperidone, foremost so weight gain and sedation. Other drugs merit recommendation as well. Relatively good evidence with a better adverse reaction profile than haloperidol is available for pimozide. Tiapride and sulpiride can be recommended based on the broad clinical experience and favorable adverse reaction profile, although more controlled clinical studies are required. Aripipazole has great potential especially in treatment refractory cases and probably less pronounced risk of severe weight gain. Finally, clonidine can be given especially when coexisting ADHD is present. All other agents mentioned in Table 1 may be considered as alternatives, once response to one or more of the earlier mentioned medications has been unsatisfactory.
In case of coexisting OCD, risperidone forms a good first choice also, based on the results of clinical trials. This may be combined with a serotonin reuptake inhibitor. Given the continuum of tics and obsessive–compulsive symptoms, other agents recommended for the treatment of tics may be tried as well; when partial response occurs, addition of a serotonin reuptake inhibitor or of behavioral treatment may be considered. Coexisting ADHD may be treated with stimulants, atomoxetine, or clonidine. This may be combined with an (antipsychotic) agent for the tics.
The current guidelines do not contain dosage recommendations of each agent. In general, dosage should start low and gradually increase with close monitoring of response and adverse reactions. Most published studies have included both children and adults, up to date, no evidence suggests that the two age-groups should be treated in different ways apart from drug dosages [73, 199]. There are several hints that dosage of pharmacotherapy of TS is not different between children, adolescents, and adults once body weight has been taken into account [213, 282], but clear data are lacking. A commonly unrecognized problem is the miss of adapting the dosage to the increasing body weight during maturation.
To the best of our knowledge, only one drug is formally licensed for the indication tics or TS in most European countries: haloperidol. With all other medications (actual exceptions of a certain country cannot be excluded), prescription is on an off-label base, reflecting the paucity of efficacy and safety data, which would not be sufficient for approval by a registration authority for any of the mentioned agents. This should always be discussed with families prior to initiation of treatment.
The proposed principles of practice are considered as guidelines only. We hope that this guideline may contribute to an improvement in the pharmacological management of patients with tic disorders. The individual treatment of a patient should be planned by considering the available diagnostic information, the level of impairment associated with tics, the efficacy data and adverse reactions of treatment options as well as patient’s preference to gain the best result and adherence possible.
References
Abuzzahab FS, Brown VL (2001) Control of Tourette’s syndrome with topiramate. Am J Psychiatry 158:968
Acosta MT, Castellanos FX (2004) Use of the “inverse neuroleptic” metoclopramide in Tourette syndrome: an open case series. J Child Adolesc Psychopharmacol 14:123–128
Adler CH, Zimmerman RS, Lyons MK, Simeone F, Brin MF (1996) Perioperative use of botulinum toxin for movement disorder-induced cervical spine disease. Mov Disord 11:79–81
Aguirregomozcorta M, Pagonabarraga J, Diaz-Manera J, Pascual-Sedano B, Gironell A, Kulisevsky J (2008) Efficacy of botulinum toxin in severe Tourette syndrome with dystonic tics involving the neck. Parkinsonism Relat Disord 14:443–445
Allen AJ, Kurlan RM, Gilbert DL, Coffey BJ, Linder SL, Lewis DW, Winner PK, Dunn DW, Dure LS, Sallee FR, Milton DR, Mintz MI, Ricardi RK, Erenberg G, Layton LL, Feldman PD, Kelsey DK, Spencer TJ (2005) Atomoxetine treatment in children and adolescents with ADHD and comorbid tic disorders. Neurology 65:1941–1949
Allison DB, Casey DE (2001) Antipsychotic-induced weight gain: a review of the literature. J Clin Psychiatry 62 (Suppl 7):22–31
Arana-Lechuga Y, Sanchez-Escandon O, de Santiago-Trevino N, Castillo-Montoya C, Teran-Perez G, Velazquez-Moctezuma J (2008) Risperidone treatment of sleep disturbances in Tourette’s syndrome. J Neuropsychiatry Clin Neurosci 20:375–376
Awaad Y (1999) Tics in Tourette syndrome: new treatment options. J Child Neurol 14:316–319
Awaad Y, Michon AM, Minarik S (2005) Use of levetiracetam to treat tics in children and adolescents with Tourette syndrome. Mov Disord 20:714–718
Banaschewski T, Neale BM, Rothenberger A, Roessner V (2007) Comorbidity of tic disorders & ADHD: conceptual and methodological considerations. Eur Child Adolesc Psychiatry 16 (Suppl 1):5–14
Banaschewski T, Roessner V, Dittmann RW, Santosh PJ, Rothenberger A (2004) Non-stimulant medications in the treatment of ADHD. Eur Child Adolesc Psychiatry 13 (Suppl 1):I102–I116
Baptista T, Molina MG, Martinez JL, de Quijada M, Calanche de Cuesta I, Acosta A, Paez X, Martinez JM, Hernandez L (1997) Effects of the antipsychotic drug sulpiride on reproductive hormones in healthy premenopausal women: relationship with body weight regulation. Pharmacopsychiatry 30:256–262
Bastiampillai T, Dhillon R, Mohindra R (2008) Exacerbation of tics secondary to clozapine therapy. Aust N Z J Psychiatry 42:1068–1070
Baving L, Schmidt MH (2000) Obsessive-compulsive disorder, frontostriatal system and the effect of the serotonergic system. Z Kinder Jugendpsychiatr Psychother 28:35–44
Begum M (2005) Clozapine-induced stuttering, facial tics and myoclonic seizures: a case report. Aust N Z J Psychiatry 39:202
Ben Djebara M, Worbe Y, Schupbach M, Hartmann A (2008) Aripiprazole: a treatment for severe coprolalia in “refractory” Gilles de la Tourette syndrome. Mov Disord 23:438–440
Bengi Semerci Z (2000) Olanzapine in Tourette’s disorder. J Am Acad Child Adolesc Psychiatry 39:140
Bhadrinath BR (1998) Olanzapine in Tourette syndrome. Br J Psychiatry 172:366
Bloch MH (2008) Emerging treatments for Tourette’s disorder. Curr Psychiatry Rep 10:323–330
Bloch MH, Landeros-Weisenberger A, Kelmendi B, Coric V, Bracken MB, Leckman JF (2006) A systematic review: antipsychotic augmentation with treatment refractory obsessive-compulsive disorder. Mol Psychiatry 11:622–632
Bloch MH, Panza KE, Landeros-Weisenberger A, Leckman JF (2009) Meta-analysis: treatment of attention-deficit/hyperactivity disorder in children with comorbid tic disorders. J Am Acad Child Adolesc Psychiatry 48:884–893
Bloch MH, Peterson BS, Scahill L, Otka J, Katsovich L, Zhang H, Leckman JF (2006) Adulthood outcome of tic and obsessive-compulsive symptom severity in children with Tourette syndrome. Arch Pediatr Adolesc Med 160:65–69
Bock N, Moll GH, Wicker M, Pilz J, Ruther E, Banaschewski T, Huether G, Rothenberger A (2004) Early administration of tiapride to young rats without long-lasting changes in the development of the dopaminergic system. Pharmacopsychiatry 37:163–167
Boon-yasidhi V, Kim YS, Scahill L (2005) An open-label, prospective study of guanfacine in children with ADHD and tic disorders. J Med Assoc Thai 88 Suppl 8:S156–S162
Borison RL, Ang L, Chang S, Dysken M, Comaty JE, Davis JM (1982) New pharmacological approaches in the treatment of Tourette syndrome. Adv Neurol 35:377–382
Borison RL, Ang L, Hamilton WJ, Diamond BI, Davis JM (1983) Treatment approaches in Gilles de la Tourette syndrome. Brain Res Bull 11:205–208
Bressan RA, Jones HM, Pilowsky LS (2004) Atypical antipsychotic drugs and tardive dyskinesia: relevance of D2 receptor affinity. J Psychopharmacol 18:124–127
Bruggeman R, van der Linden C, Buitelaar JK, Gericke GS, Hawkridge SM, Temlett JA (2001) Risperidone versus pimozide in Tourette’s disorder: a comparative double-blind parallel-group study. J Clin Psychiatry 62:50–56
Bruun RD (1988) Subtle and underrecognized adverse reactions of neuroleptic treatment in children with Tourette’s disorder. Am J Psychiatry 145:621–624
Bruun RD, Budman CL (1996) Risperidone as a treatment for Tourette’s syndrome. J Clin Psychiatry 57:29–31
Bubl E, Perlov E, Tebartz Van Elst L (2006) Aripiprazole in patients with Tourette syndrome. World J Biol Psychiatry 7:123–125
Budman C, Coffey BJ, Shechter R, Schrock M, Wieland N, Spirgel A, Simon E (2008) Aripiprazole in children and adolescents with Tourette disorder with and without explosive outbursts. J Child Adolesc Psychopharmacol 18:509–515
Budman CL, Gayer A, Lesser M, Shi Q, Bruun RD (2001) An open-label study of the treatment efficacy of olanzapine for Tourette’s disorder. J Clin Psychiatry 62:290–294
Burris KD, Molski TF, Xu C, Ryan E, Tottori K, Kikuchi T, Yocca FD, Molinoff PB (2002) Aripiprazole, a novel antipsychotic, is a high-affinity partial agonist at human dopamine D2 receptors. J Pharmacol Exp Ther 302:381–389
Caine ED, Polinsky RJ, Kartzinel R, Ebert MH (1979) The trial use of clozapine for abnormal involuntary movement disorders. Am J Psychiatry 136:317–320
Castellanos FX, Giedd JN, Elia J, Marsh WL, Ritchie GF, Hamburger SD, Rapoport JL (1997) Controlled stimulant treatment of ADHD and comorbid Tourette’s syndrome: effects of stimulant and dose. J Am Acad Child Adolesc Psychiatry 36:589–596
Chang V, Friedman JH (2009) Motor adverse reactions of atypical antipsychotic drugs. Therapy 6:249–258
Chappell PB, Leckman JF, Riddle MA, Anderson GM, Listwack SJ, Ort SI, Hardin MT, Scahill LD, Cohen DJ (1992) Neuroendocrine and behavioral effects of naloxone in Tourette syndrome. Adv Neurol 58:253–262
Chappell PB, Leckman JF, Scahill LD, Hardin MT, Anderson G, Cohen DJ (1993) Neuroendocrine and behavioral effects of the selective kappa agonist spiradoline in Tourette’s syndrome: a pilot study. Psychiatry Res 47:267–280
Chappell PB, Riddle MA, Scahill L, Lynch KA, Schultz R, Arnsten A, Leckman JF, Cohen DJ (1995) Guanfacine treatment of comorbid attention-deficit hyperactivity disorder and Tourette’s syndrome: preliminary clinical experience. J Am Acad Child Adolesc Psychiatry 34:1140–1146
Cheng JY, Chen RY, Ko JS, Ng EM (2007) Efficacy and safety of atomoxetine for attention-deficit/hyperactivity disorder in children and adolescents-meta-analysis and meta-regression analysis. Psychopharmacology (Berl) 194:197–209
Cheon KA, Ryu YH, Namkoong K, Kim CH, Kim JJ, Lee JD (2004) Dopamine transporter density of the basal ganglia assessed with [123I]IPT SPECT in drug-naive children with Tourette’s disorder. Psychiatry Res 130:85–95
Chouza C, Romero S, Lorenzo J, Camano JL, Fontana AP, Alterwain P, Cibils D, Gaudiano J, Feres S, Solana J (1982) Clinical trial of tiapride in patients with dyskinesia (author’s transl). Sem Hop 58:725–733
Coffey BJ, Biederman J, Geller D, Frazier J, Spencer T, Doyle R, Gianini L, Small A, Frisone DF, Magovcevic M, Stein N, Faraone SV (2004) Reexamining Tic Persistence and Tic-Associated Impairment in Tourette’s Disorder: Findings From a Naturalistic Follow-Up Study. J Nerv Ment Dis 192:776–780
Cohen DJ, Detlor J, Young JG, Shaywitz BA (1980) Clonidine ameliorates Gilles de la Tourette syndrome. Arch Gen Psychiatry 37:1350–1357
Cohen DJ, Young JG, Nathanson JA, Shaywitz BA (1979) Clonidine in Tourette’s syndrome. Lancet 2:551–553
Constant EL, Borras L, Seghers A (2006) Aripiprazole is effective in the treatment of Tourette’s disorder. Int J Neuropsychopharmacol 9:773–774
Copur M, Arpaci B, Demir T, Narin H (2007) Clinical effectiveness of quetiapine in children and adolescents with Tourette’s syndrome: a retrospective case-note survey. Clin Drug Investig 27:123–130
Cui YH, Zheng Y, Yang YP, Liu J, Li J (2010) Effectiveness and tolerability of aripiprazole in children and adolescents with Tourette’s disorder: a pilot study in China. J Child Adolesc Psychopharmacol 20:291–298
Cummings DD, Singer HS, Krieger M, Miller TL, Mahone EM (2002) Neuropsychiatric effects of guanfacine in children with mild tourette syndrome: a pilot study. Clin Neuropharmacol 25:325–332
Curtis A, Clarke CE, Rickards HE (2009) Cannabinoids for Tourette’s Syndrome. Cochrane Database Syst Rev:CD006565
Davies L, Stern JS, Agrawal N, Robertson MM (2006) A case series of patients with Tourette’s syndrome in the United Kingdom treated with aripiprazole. Hum Psychopharmacol 21:447–453
Daviss WB, Patel NC, Robb AS, McDermott MP, Bukstein OG, Pelham WE Jr, Palumbo D, Harris P, Sallee FR (2008) Clonidine for attention-deficit/hyperactivity disorder: II. ECG changes and adverse events analysis. J Am Acad Child Adolesc Psychiatry 47:189–198
Debes N, Hjalgrim H, Skov L (2010) The presence of attention-deficit hyperactivity disorder (ADHD) and obsessive-compulsive disorder worsen psychosocial and educational problems in Tourette syndrome. J Child Neurol 25:171–181
Dehning S, Riedel M, Muller N (2005) Aripiprazole in a patient vulnerable to adverse reactions. Am J Psychiatry 162:625
Delgado PL, Goodman WK, Price LH, Heninger GR, Charney DS (1990) Fluvoxamine/pimozide treatment of concurrent Tourette’s and obsessive-compulsive disorder. Br J Psychiatry 157:762–765
Devor EJ, Isenberg KE (1989) Nicotine and Tourette’s syndrome. Lancet 2:1046
Diantoniis MR, Henry KM, Partridge PA, Soucar E (1996) Tics and risperidone. J Am Acad Child Adolesc Psychiatry 35:839–840
Dimitsopulos T, Kurlan R (1993) Tourette’s syndrome and nicotine withdrawal. J Neuropsychiatry Clin Neurosci 5:108–109
Dion Y, Annable L, Sandor P, Chouinard G (2002) Risperidone in the treatment of Tourette syndrome: a double-blind, placebo-controlled trial. J Clin Psychopharmacol 22:31–39
Drtílková I (1996) Clonazepam, clonidine and tiapride in children with tic disorder. Homeostasis 37:216
Du YS, Li HF, Vance A, Zhong YQ, Jiao FY, Wang HM, Wang MJ, Su LY, Yu DL, Ma SW, Wu JB (2008) Randomized double-blind multicentre placebo-controlled clinical trial of the clonidine adhesive patch for the treatment of tic disorders. Aust N Z J Psychiatry 42:807–813
Duane DD (2006) Aripiprazole in childhood and adolescence for Tourette syndrome. J Child Neurol 21:358
Dulcan M (1997) Practice parameters for the assessment and treatment of children, adolescents, and adults with attention-deficit/hyperactivity disorder. American Academy of Child and Adolescent Psychiatry. J Am Acad Child Adolesc Psychiatry 36:85S–121S
Dursun SM, Burke JG, Reveley MA (1995) Buspirone treatment of Tourette’s syndrome. Lancet 345:1366–1367
Dursun SM, Reveley MA (1997) Differential effects of transdermal nicotine on microstructured analyses of tics in Tourette’s syndrome: an open study. Psychol Med 27:483–487
Dursun SM, Reveley MA, Bird R, Stirton F (1994) Longlasting improvement of Tourette’s syndrome with transdermal nicotine. Lancet 344:1577
Eggers C, Rothenberger A, Berghaus U (1988) Clinical and neurobiological findings in children suffering from tic disease following treatment with tiapride. Eur Arch Psychiatry Neurol Sci 237:223–229
Eggers C, Schepker R, Oades RD (1993) Exacerbation and provocation of tics by imipramine and sulpiride. Eur Child Adolesc Psychiatry 2:169–176
Erickson HM Jr, Goggin JE, Messiha FS (1977) Comparison of lithium and haloperidol therapy in Gilles de la Tourette syndrome. Adv Exp Med Biol 90:197–205
Fernandez-Jaen A, Fernandez-Mayoralas DM, Munoz-Jareno N, Calleja-Perez B (2009) An open-label, prospective study of levetiracetam in children and adolescentes with Tourette syndrome. Eur J Paediatr Neurol
Feroz-Nainar C, Roy M (2006) Risperidone and late onset tics. Autism 10:302–307
Findling RL, Kauffman RE, Sallee FR, Carson WH, Nyilas M, Mallikaarjun S, Shoaf SE, Forbes RA, Boulton DW, Pikalov A (2008) Tolerability and pharmacokinetics of aripiprazole in children and adolescents with psychiatric disorders: an open-label, dose-escalation study. J Clin Psychopharmacol 28:441–446
Forner-Valero J, Aymerich VB, Oritz-Lopez A (1986) Comperative study of the effects of tiapride and a placebo on the involuntary movements and manual ability of children with cerebromotor disorders. Neuropsychiatrie de l’Enfance et de l’Adolescence 34:213–216
Fountoulakis KN, Iacovides A, St Kaprinis G (2004) Successful treatment of Tourette’s disorder with amisulpride. Ann Pharmacother 38:901
Fountoulakis KN, Siamouli M, Kantartzis S, Panagiotidis P, Iacovides A, Kaprinis GS (2006) Acute dystonia with low-dosage aripiprazole in Tourette’s disorder. Ann Pharmacother 40:775–777
Fras I (1996) Guanfacine for Tourette’s disorder. J Am Acad Child Adolesc Psychiatry 35:3–4
Frederiks JA (1970) Facial tics in children: the therapeutic effect of low-dosage diazepam. Br J Clin Pract 24:17–20
Freeman RD (2007) Tic disorders and ADHD: answers from a world-wide clinical dataset on Tourette syndrome. Eur Child Adolesc Psychiatry 16 (Suppl 1):15–23
Fusco C, Bertani G, Caricati G, Della Giustina E (2006) Stress fracture of the peroneal bone secondary to a complex tic. Brain Dev 28:52–54
Gadoth N, Gordon CR, Streifler J (1987) Naloxone in Gilles de la Tourette’s syndrome. Ann Neurol 21:415
Gadow KD, Sverd J, Sprafkin J, Nolan EE, Grossman S (1999) Long-term methylphenidate therapy in children with comorbid attention-deficit hyperactivity disorder and chronic multiple tic disorder. Arch Gen Psychiatry 56:330–336
Gaffney GR, Perry PJ, Lund BC, Bever-Stille KA, Arndt S, Kuperman S (2002) Risperidone versus clonidine in the treatment of children and adolescents with Tourette’s syndrome. J Am Acad Child Adolesc Psychiatry 41:330–336
Gancher S, Conant-Norville D, Angell R (1990) Treatment of Tourette’s syndrome with transdermal clonidine: a pilot study. J Neuropsychiatry Clin Neurosci 2:66–69
George MS, Trimble MR, Robertson MM (1993) Fluvoxamine And Sulpiride In Comorbid Obsessive-Compulsive Disorder And Gilles-De-La-Tourette Syndrome. Hum Psychopharmacol Clin Exp 8:327–334
Giakas WJ (1995) Risperidone treatment for a Tourette’s disorder patient with comorbid obsessive-compulsive disorder. Am J Psychiatry 152:1097–1098
Gilbert D (2006) Treatment of children and adolescents with tics and Tourette syndrome. J Child Neurol 21:690–700
Gilbert DL, Batterson JR, Sethuraman G, Sallee FR (2004) Tic reduction with risperidone versus pimozide in a randomized, double-blind, crossover trial. J Am Acad Child Adolesc Psychiatry 43:206–214
Goetz CG (1992) Clonidine and clonazepam in Tourette syndrome. Adv Neurol 58:245–251
Goetz CG, Stebbins GT, Thelen JA (1994) Talipexole and adult Gilles de la Tourette’s syndrome: double-blind, placebo-controlled clinical trial. Mov Disord 9:315–317
Goetz CG, Tanner CM, Klawans HL (1984) Fluphenazine and multifocal tic disorders. Arch Neurol 41:271–272
Goetz CG, Tanner CM, Wilson RS, Carroll VS, Como PG, Shannon KM (1987) Clonidine and Gilles de la Tourette’s syndrome: double-blind study using objective rating methods. Ann Neurol 21:307–310
Golden GS (1985) Tardive dyskinesia in Tourette syndrome. Pediatr Neurol 1:192–194
Gonce M, Barbeau A (1977) Seven cases of Gilles de la Tourette’s syndrome: partial relief with clonazepam: a pilot study. Can J Neurol Sci 4:279–283
Gorman DA, Thompson N, Plessen KJ, Robertson MM, Leckman JF, Peterson BS (2010) Psychosocial outcome and psychiatric comorbidity in older adolescents with Tourette syndrome: controlled study. Br J Psychiatry 197:36–44
Grunder G, Fellows C, Janouschek H, Veselinovic T, Boy C, Brocheler A, Kirschbaum KM, Hellmann S, Spreckelmeyer KM, Hiemke C, Rosch F, Schaefer WM, Vernaleken I (2008) Brain and plasma pharmacokinetics of aripiprazole in patients with schizophrenia: an [18F]fallypride PET study. Am J Psychiatry 165:988–995
Harris K, Singer HS (2006) Tic disorders: neural circuits, neurochemistry, and neuroimmunology. J Child Neurol 21:678–689
Hasan A, Rothenberger A, Munchau A, Wobrock T, Falkai P, Roessner V (2010) Oral Delta 9-tetrahydrocannabinol improved refractory Gilles de la Tourette syndrome in an adolescent by increasing intracortical inhibition: a case report. J Clin Psychopharmacol 30:190–192
Hedderick EF, Morris CM, Singer HS (2009) Double-blind, crossover study of clonidine and levetiracetam in Tourette syndrome. Pediatr Neurol 40:420–425
Ho CS, Chen HJ, Chiu NC, Shen EY, Lue HC (2009) Short-term sulpiride treatment of children and adolescents with Tourette syndrome or chronic tic disorder. J Formos Med Assoc 108:788–793
Hoekstra PJ, Minderaa RB, Kallenberg CG (2004) Lack of effect of intravenous immunoglobulins on tics: a double-blind placebo-controlled study. J Clin Psychiatry 65:537–542
Horrigan JP, Barnhill LJ (1999) Guanfacine and secondary mania in children. J Affect Disord 54:309–314
Hounie A, De Mathis A, Sampaio AS, Mercadante MT (2004) Aripiprazole and Tourette syndrome. Rev Bras Psiquiatr 26:213
Howson AL, Batth S, Ilivitsky V, Boisjoli A, Jaworski M, Mahoney C, Knott VJ (2004) Clinical and attentional effects of acute nicotine treatment in Tourette’s syndrome. Eur Psychiatry 19:102–112
Huang TL, Lu CY (2007) Correlations between weight changes and lipid profile changes in schizophrenic patients after antipsychotics therapy. Chang Gung Med J 30:26–32
Ikenouchi-Sugita A, Yoshimura R, Hayashi K, Ueda N, Umene-Nakano W, Hori H, Nakamura J (2009) A case of late-onset Tourette’s disorder successfully treated with aripiprazole: View from blood levels of catecholamine metabolites and brain-derived neurotrophic factor (BDNF). World J Biol Psychiatry 1–4
Jankovic J (1994) Botulinum toxin in the treatment of dystonic tics. Mov Disord 9:347–349
Jankovic J, Beach J (1997) Long-term effects of tetrabenazine in hyperkinetic movement disorders. Neurology 48:358–362
Jankovic J, Glaze DG, Frost JD Jr (1984) Effect of tetrabenazine on tics and sleep of Gilles de la Tourette’s syndrome. Neurology 34:688–692
Jankovic J, Jimenez-Shahed J, Brown L (2010) A randomized, double-blind, placebo-controlled study of topiramate in the treatment of Tourette syndrome. J Neurol Neurosurg Psychiatry 81:70–73
Jankovic J, Orman J (1988) Tetrabenazine therapy of dystonia, chorea, tics, and other dyskinesias. Neurology 38:391–394
Jankovic J, Rohaidy H (1987) Motor, behavioral and pharmacologic findings in Tourette’s syndrome. Can J Neurol Sci 14:541–546
Jordan S, Koprivica V, Chen R, Tottori K, Kikuchi T, Altar CA (2002) The antipsychotic aripiprazole is a potent, partial agonist at the human 5-HT1A receptor. Eur J Pharmacol 441:137–140
Joy CB, Adams CE, Lawrie SM (2006) Haloperidol versus placebo for schizophrenia. Cochrane Database Syst Rev CD003082
Kaim B (1983) A case of Gilles de la Tourette’s syndrome treated with clonazepam. Brain Res Bull 11:213–214
Kang H, Zhang YF, Jiao FY, Guo XY, Gao XM (2009) Efficacy of clonidine transdermal patch for treatment of Tourette’s syndrome in children. Zhongguo Dang Dai Er Ke Za Zhi 11:537–539
Karam-Hage M, Ghaziuddin N (2000) Olanzapine in Tourette’s disorder. J Am Acad Child Adolesc Psychiatry 39:139
Kastrup A, Schlotter W, Plewnia C, Bartels M (2005) Treatment of tics in tourette syndrome with aripiprazole. J Clin Psychopharmacol 25:94–96
Kawohl W, Schneider F, Vernaleken I, Neuner I (2008) Aripiprazole in the pharmacotherapy of Gilles de la Tourette syndrome in adult patients. World J Biol Psychiatry 1–5
Kenney CJ, Hunter CB, Mejia NI, Jankovic J (2007) Tetrabenazine in the treatment of Tourette syndrome J Pediatr Neurol 5 9–13
Kerbeshian J, Burd L (1988) Differential responsiveness to lithium in patients with Tourette disorder. Neurosci Biobehav Rev 12:247–250
Kim BN, Lee CB, Hwang JW, Shin MS, Cho SC (2005) Effectiveness and safety of risperidone for children and adolescents with chronic tic or tourette disorders in Korea. J Child Adolesc Psychopharmacol 15:318–324
King A, Harris P, Fritzell J, Kurlan R (2006) Syncope in children with Tourette’s syndrome treated with guanfacine. Mov Disord 21:419–420
Klepel H, Gebelt H, Koch RD, Tzenow H (1988) Treatment of extrapyramidal hyperkineses in childhood with tiapride. Psychiatr Neurol Med Psychol (Leipz) 40:516–522
Krauss JK, Jankovic J (1996) Severe motor tics causing cervical myelopathy in Tourette’s syndrome. Mov Disord 11:563–566
Krishnamoorthy J, King BH (1998) Open-label olanzapine treatment in five preadolescent children. J Child Adolesc Psychopharmacol 8:107–113
Kuo SH, Jimenez-Shahed J (2010) Topiramate in Treatment of Tourette Syndrome. Clin Neuropharmacol 33:32–34
Kurlan R (1997) Tourette syndrome. Treatment of tics. Neurol Clin 15:403–409
Kurlan R, Majumdar L, Deeley C, Mudholkar GS, Plumb S, Como PG (1991) A controlled trial of propoxyphene and naltrexone in patients with Tourette’s syndrome. Ann Neurol 30:19–23
Kurlan R, McDermott MP, Deeley C, Como PG, Brower C, Eapen S, Andresen EM, Miller B (2001) Prevalence of tics in schoolchildren and association with placement in special education. Neurology 57:1383–1388
Kwak CH, Hanna PA, Jankovic J (2000) Botulinum toxin in the treatment of tics. Arch Neurol 57:1190–1193
Leckman JF (2002) Tourette’s syndrome. Lancet 360:1577–1586
Leckman JF, Detlor J, Harcherik DF, Ort S, Shaywitz BA, Cohen DJ (1985) Short- and long-term treatment of Tourette’s syndrome with clonidine: a clinical perspective. Neurology 35:343–351
Leckman JF, Hardin MT, Riddle MA, Stevenson J, Ort SI, Cohen DJ (1991) Clonidine treatment of Gilles de la Tourette’s syndrome. Arch Gen Psychiatry 48:324–328
Leckman JF, Riddle MA, Hardin MT, Ort SI, Swartz KL, Stevenson J, Cohen DJ (1989) The yale global tic severity scale: initial testing of a clinician-rated scale of tic severity. J Am Acad Child Adolesc Psychiatry 28:566–573
Ledbetter M (2005) Atomoxetine use associated with onset of a motor tic. J Child Adolesc Psychopharmacol 15:331–333
Lichter DG, Jackson LA (1996) Predictors of clonidine response in Tourette syndrome: implications and inferences. J Child Neurol 11:93–97
Linet LS (1985) Tourette syndrome, pimozide, and school phobia: the neuroleptic separation anxiety syndrome. Am J Psychiatry 142:613–615
Lipcsey A (1983) Gilles de la Tourette’s disease. Sem Hop 59:695–696
Lombroso PJ, Scahill L, King RA, Lynch KA, Chappell PB, Peterson BS, McDougle CJ, Leckman JF (1995) Risperidone treatment of children and adolescents with chronic tic disorders: a preliminary report. J Am Acad Child Adolesc Psychiatry 34:1147–1152
Lucas Taracena MT, Montanes Rada F (2002) Olanzapine in Tourette’s syndrome: a report of three cases. Actas Esp Psiquiatr 30:129–132
Lyon GJ, Samar S, Jummani R, Hirsch S, Spirgel A, Goldman R, Coffey BJ (2009) Aripiprazole in children and adolescents with Tourette’s disorder: an open-label safety and tolerability study. J Child Adolesc Psychopharmacol 19:623–633
Margolese HC, Annable L, Dion Y (2002) Depression and dysphoria in adult and adolescent patients with Tourette’s disorder treated with risperidone. J Clin Psychiatry 63:1040–1044
Marras C, Andrews D, Sime E, Lang AE (2001) Botulinum toxin for simple motor tics: a randomized, double-blind, controlled clinical trial. Neurology 56:605–610
Mathe JF, Cler JM, Venisse JL (1978) Therapeutic use of tiapride in movement disorders. Sem Hop 54:517–520
McConville BJ, Fogelson MH, Norman AB, Klykylo WM, Manderscheid PZ, Parker KW, Sanberg PR (1991) Nicotine potentiation of haloperidol in reducing tic frequency in Tourette’s disorder. Am J Psychiatry 148:793–794
McConville BJ, Sanberg PR, Fogelson MH, King J, Cirino P, Parker KW, Norman AB (1992) The effects of nicotine plus haloperidol compared to nicotine only and placebo nicotine only in reducing tic severity and frequency in Tourette’s disorder. Biol Psychiatry 31:832–840
McCracken JT, Suddath R, Chang S, Thakur S, Piacentini J (2008) Effectiveness and tolerability of open label olanzapine in children and adolescents with Tourette syndrome. J Child Adolesc Psychopharmacol 18:501–508
McDougle CJ, Goodman WK, Leckman JF, Barr LC, Heninger GR, Price LH (1993) The efficacy of fluvoxamine in obsessive-compulsive disorder: effects of comorbid chronic tic disorder. J Clin Psychopharmacol 13:354–358
McKeith IG, Williams A, Nicol AR (1981) Clonidine in Tourette syndrome. Lancet 1:270–271
Meisel A, Winter C, Zschenderlein R, Arnold G (2004) Tourette syndrome: efficient treatment with ziprasidone and normalization of body weight in a patient with excessive weight gain under tiapride. Mov Disord 19:991–992
Merikangas JR, Merikangas KR, Kopp U, Hanin I (1985) Blood choline and response to clonazepam and haloperidol in Tourette’s syndrome. Acta Psychiatr Scand 72:395–399
Meyer JM (2002) A retrospective comparison of weight, lipid, and glucose changes between risperidone- and olanzapine-treated inpatients: metabolic outcomes after 1 year. J Clin Psychiatry 63:425–433
Mikkelsen EJ, Detlor J, Cohen DJ (1981) School avoidance and social phobia triggered by haloperidol in patients with Tourette’s disorder. Am J Psychiatry 138:1572–1576
Miller del D, Eudicone JM, Pikalov A, Kim E (2007) Comparative assessment of the incidence and severity of tardive dyskinesia in patients receiving aripiprazole or haloperidol for the treatment of schizophrenia: a post hoc analysis. J Clin Psychiatry 68:1901–1906
Miller LG, Jankovic J (1990) Sulpiride-induced tardive dystonia. Mov Disord 5:83–84
Minzer K, Lee O, Hong JJ, Singer HS (2004) Increased prefrontal D2 protein in Tourette syndrome: a postmortem analysis of frontal cortex and striatum. J Neurol Sci 219:55–61
Miranda CM, Castiglioni TC (2007) Aripiprazole for the treatment of Tourette syndrome. Experience in 10 patients. Rev Med Chil 135:773–776
Mukaddes NM, Abali O (2003) Quetiapine treatment of children and adolescents with Tourette’s disorder. J Child Adolesc Psychopharmacol 13:295–299
Muller-Vahl KR, Meyer GJ, Knapp WH, Emrich HM, Gielow P, Brucke T, Berding G (2005) Serotonin transporter binding in Tourette Syndrome. Neurosci Lett 385:120–125
Muller-Vahl KR, Prevedel H, Theloe K, Kolbe H, Emrich HM, Schneider U (2003) Treatment of Tourette syndrome with delta-9-tetrahydrocannabinol (delta 9-THC): no influence on neuropsychological performance. Neuropsychopharmacology 28:384–388
Muller-Vahl KR, Schneider U, Koblenz A, Jobges M, Kolbe H, Daldrup T, Emrich HM (2002) Treatment of Tourette’s syndrome with Delta 9-tetrahydrocannabinol (THC): a randomized crossover trial. Pharmacopsychiatry 35:57–61
Muller-Vahl KR, Schneider U, Kolbe H, Emrich HM (1999) Treatment of Tourette’s syndrome with delta-9-tetrahydrocannabinol. Am J Psychiatry 156:495
Muller-Vahl KR, Schneider U, Prevedel H, Theloe K, Kolbe H, Daldrup T, Emrich HM (2003) Delta 9-tetrahydrocannabinol (THC) is effective in the treatment of tics in Tourette syndrome: a 6-week randomized trial. J Clin Psychiatry 64:459–465
Muller N (2004) Anti-inflammatory therapy with a COX-2 inhibitor in Tourette’s syndrome. Inflammopharmacol 12:271–275
Murphy TK, Bengtson MA, Soto O, Edge PJ, Sajid MW, Shapira N, Yang M (2005) Case series on the use of aripiprazole for Tourette syndrome. Int J Neuropsychopharmacol 8:489–490
Murphy TK, Mutch PJ, Reid JM, Edge PJ, Storch EA, Bengtson M, Yang M (2009) Open label aripiprazole in the treatment of youth with tic disorders. J Child Adolesc Psychopharmacol 19:441–447
Neglia JP, Glaze DG, Zion TE (1984) Tics and vocalizations in children treated with carbamazepine. Pediatrics 73:841–844
Nicolson R, Craven-Thuss B, Smith J, McKinlay BD, Castellanos FX (2005) A randomized, double-blind, placebo-controlled trial of metoclopramide for the treatment of Tourette’s disorder. J Am Acad Child Adolesc Psychiatry 44:640–646
Ondo WG, Jong D, Davis A (2008) Comparison of weight gain in treatments for Tourette syndrome: tetrabenazine versus neuroleptic drugs. J Child Neurol 23:435–437
Onofrj M, Paci C, D’Andreamatteo G, Toma L (2000) Olanzapine in severe Gilles de la Tourette syndrome: a 52-week double-blind crossover study vs. low-dose pimozide. J Neurol 247:443–446
Orth M, Amann B, Robertson MM, Rothwell JC (2005) Excitability of motor cortex inhibitory circuits in Tourette syndrome before and after single dose nicotine. Brain 128:1292–1300
Padala PR, Qadri SF, Madaan V (2005) Aripiprazole for the treatment of Tourette’s disorder. Prim Care Companion J Clin Psychiatry 7:296–299
Paleacu D, Giladi N, Moore O, Stern A, Honigman S, Badarny S (2004) Tetrabenazine treatment in movement disorders. Clin Neuropharmacol 27:230–233
Palumbo D, Spencer T, Lynch J, Co-Chien H, Faraone SV (2004) Emergence of tics in children with ADHD: impact of once-daily OROS methylphenidate therapy. J Child Adolesc Psychopharmacol 14:185–194
Pani L, Gessa GL (2002) The substituted benzamides and their clinical potential on dysthymia and on the negative symptoms of schizophrenia. Mol Psychiatry 7:247–253
Pappadopulos E, Woolston S, Chait A, Perkins M, Connor DF, Jensen PS (2006) Pharmacotherapy of aggression in children and adolescents: efficacy and effect size. J Can Acad Child Adolesc Psychiatry 15:27–39
Parraga HC, Parraga KL, Harris DK, Campbell TS (2008) Abdominal tics during atomoxetine treatment in a child with ADHD: evaluation and differential diagnosis. CNS Spectr 13:E1
Parraga HC, Parraga MI (2001) Quetiapine treatment in patients with Tourette syndrome. Can J Psychiatry 46:184–185
Parraga HC, Parraga MI, Harris DK (2007) Tic exacerbation and precipitation during atomoxetine treatment in two children with attention-deficit hyperactivity disorder. Int J Psychiatry Med 37:415–424
Parraga HC, Parraga MI, Woodward RL, Fenning PA (2001) Quetiapine treatment of children with Tourette’s syndrome: report of two cases. J Child Adolesc Psychopharmacol 11:187–191
Parraga HC, Woodward RL (2001) Quetiapine for Tourette’s syndrome. J Am Acad Child Adolesc Psychiatry 40:389–391
Pasquier C, Pouplard F (1977) Reflections on child tics. Apropos of a therapeutic trial. Rev Neuropsychiatr Infant 25:645–651
Perez-Iglesias R, Mata I, Pelayo-Teran JM, Amado JA, Garcia-Unzueta MT, Berja A, Martinez-Garcia O, Vazquez-Barquero JL, Crespo-Facorro B (2009) Glucose and lipid disturbances after 1 year of antipsychotic treatment in a drug-naive population. Schizophr Res 107:115–121
Perlmutter SJ, Leitman SF, Garvey MA, Hamburger S, Feldman E, Leonard HL, Swedo SE (1999) Therapeutic plasma exchange and intravenous immunoglobulin for obsessive-compulsive disorder and tic disorders in childhood. Lancet 354:1153–1158
Pierce A, Rickards H (in press) Atypical Antipsychotics for Tourette’s Syndrome. Cochrane Database Syst Rev
Porta M, Maggioni G, Ottaviani F, Schindler A (2004) Treatment of phonic tics in patients with Tourette’s syndrome using botulinum toxin type A. Neurol Sci 24:420–423
Porta M, Sassi M, Cavallazzi M, Fornari M, Brambilla A, Servello D (2008) Tourette’s syndrome and role of tetrabenazine: review and personal experience. Clin Drug Investig 28:443–459
Pringsheim T, Marras C (2009) Pimozide for tics in Tourette’s syndrome. Cochrane Database Syst Rev CD006996
Rath JJ, Tavy DL, Wertenbroek AA, van Woerkom TC, de Bruijn SF (2010) Botulinum toxin type A in simple motor tics: short-term and long-term treatment-effects. Parkinsonism Relat Disord 16:478–481
Riddle MA, Carlson J (2001) Clinical psychopharmacology for Tourette syndrome and associated disorders. Adv Neurol 85:343–354
Riddle MA, Hardin MT, Towbin KE, Leckman JF, Cohen DJ (1987) Tardive dyskinesia following haloperidol treatment in Tourette’s syndrome. Arch Gen Psychiatry 44:98–99
Riley DE, Lang AE (1989) Pain in Gilles de la Tourette syndrome and related tic disorders. Can J Neurol Sci 16:439–441
Robertson MM (2008) The prevalence and epidemiology of Gilles de la Tourette syndrome. Part 2: tentative explanations for differing prevalence figures in GTS, including the possible effects of psychopathology, aetiology, cultural differences, and differing phenotypes. J Psychosom Res 65:473–486
Robertson MM (2000) Tourette syndrome, associated conditions and the complexities of treatment. Brain 123 Pt 3:425–462
Robertson MM, Eapen V, Cavanna AE (2009) The international prevalence, epidemiology, and clinical phenomenology of Tourette syndrome: a cross-cultural perspective. J Psychosom Res 67:475–483
Robertson MM, Schnieden V, Lees AJ (1990) Management of Gilles de la Tourette syndrome using sulpiride. Clin Neuropharmacol 13:229–235
Robertson MM, Scull DA, Eapen V, Trimble MR (1996) Risperidone in the treatment of Tourette syndrome: A retrospective case note study. Psychopharmacol 10:317–320
Robertson MM, Stern JS (2000) Gilles de la Tourette syndrome: symptomatic treatment based on evidence. Eur Child Adolesc Psychiatry 9 (Suppl 1):I60–I75
Roessner V, Banaschewski T, Fillmer-Otte A, Becker A, Albrecht B, Uebel H, Sergeant J, Tannock R, Rothenberger A (2008) Color perception deficits in co-existing attention-deficit/hyperactivity disorder and chronic tic disorders. J Neural Transm 115:235–239
Roessner V, Becker A, Banaschewski T, Rothenberger A (2007) Executive functions in children with chronic tic disorders with/without ADHD: new insights. Eur Child Adolesc Psychiatry 16 (Suppl 1):36–44
Roessner V, Becker A, Banaschewski T, Rothenberger A (2007) Psychopathological profile in children with chronic tic disorder and co-existing ADHD: additive effects. J Abnorm Child Psychol 35:79–85
Roessner V, Becker A, Banaschewski T, Rothenberger A (2005) Tic disorders and obsessive compulsive disorder: where is the link? J Neural Transm Suppl 69:69–99
Roessner V, Robatzek M, Knapp G, Banaschewski T, Rothenberger A (2006) First-onset tics in patients with attention-deficit-hyperactivity disorder: impact of stimulants. Dev Med Child Neurol 48:616–621
Roke Y, van Harten PN, Boot AM, Buitelaar JK (2009) Antipsychotic medication in children and adolescents: a descriptive review of the effects on prolactin level and associated adverse reactions. J Child Adolesc Psychopharmacol 19:403–414
Ross MS, Moldofsky H (1978) A comparison of pimozide and haloperidol in the treatment of Gilles de la Tourette’s syndrome. Am J Psychiatry 135:585–587
Rothenberger A, Banaschewski T, Roessner V (2007) Tic-Störungen. In: Deutsche Gesellschaft für Kinder- u. Jugendpsychiatrie PuP (ed) Leitlinien zur Diagnostik und Therapie von psychischen Störungen im Säuglings-, Kindes- und Jugendalter. Deutscher Ärzteverlag, Köln, pp 319–325
Rothenberger A, Roessner V, Banaschewski T, Leckman JF (2007) Co-existence of tic disorders and attention-deficit/hyperactivity disorder-recent advances in understanding and treatment. Eur Child Adolesc Psychiatry 16 (Suppl 1):1–4
Ruther E, Degner D, Munzel U, Brunner E, Lenhard G, Biehl J, Vogtle-Junkert U (1999) Antidepressant action of sulpiride. Results of a placebo-controlled double-blind trial. Pharmacopsychiatry 32:127–135
Sallee F, McGough J, Wigal T, Donahue J, Lyne A, Biederman J (2009) Guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder: a placebo-controlled trial. J Am Acad Child Adolesc Psychiatry 48:155–165
Sallee FR, Gilbert DL, Vinks AA, Miceli JJ, Robarge L, Wilner K (2003) Pharmacodynamics of ziprasidone in children and adolescents: impact on dopamine transmission. J Am Acad Child Adolesc Psychiatry 42:902–907
Sallee FR, Kurlan R, Goetz CG, Singer H, Scahill L, Law G, Dittman VM, Chappell PB (2000) Ziprasidone treatment of children and adolescents with Tourette’s syndrome: a pilot study. J Am Acad Child Adolesc Psychiatry 39:292–299
Sallee FR, Miceli JJ, Tensfeldt T, Robarge L, Wilner K, Patel NC (2006) Single-dose pharmacokinetics and safety of ziprasidone in children and adolescents. J Am Acad Child Adolesc Psychiatry 45:720–728
Sallee FR, Nesbitt L, Jackson C, Sine L, Sethuraman G (1997) Relative efficacy of haloperidol and pimozide in children and adolescents with Tourette’s disorder. Am J Psychiatry 154:1057–1062
Salloway S, Stewart CF, Israeli L, Morales X, Rasmussen S, Blitzer A, Brin MF (1996) Botulinum toxin for refractory vocal tics. Mov Disord 11:746–748
Sanberg PR, Fogelson HM, Manderscheid PZ, Parker KW, Norman AB, McConville BJ (1988) Nicotine gum and haloperidol in Tourette’s syndrome. Lancet 1:592
Sandor P (2003) Pharmacological management of tics in patients with TS. J Psychosom Res 55:41–48
Sandor P, Musisi S, Moldofsky H, Lang A (1990) Tourette syndrome: a follow-up study. J Clin Psychopharmacol 10:197–199
Sandor P, Stephens RJ (2000) Risperidone treatment of aggressive behavior in children with Tourette syndrome. J Clin Psychopharmacol 20:710–712
Sandyk R (1985) The effects of naloxone in Tourette’s syndrome. Ann Neurol 18:367–368
Sandyk R (1987) Naloxone abolishes obsessive-compulsive behavior in Tourette’s syndrome. Int J Neurosci 35:93–94
Sandyk R (1986) Naloxone withdrawal exacerbates Tourette syndrome. J Clin Psychopharmacol 6:58–59
Scahill L, Chappell PB, Kim YS, Schultz RT, Katsovich L, Shepherd E, Arnsten AF, Cohen DJ, Leckman JF (2001) A placebo-controlled study of guanfacine in the treatment of children with tic disorders and attention deficit hyperactivity disorder. Am J Psychiatry 158:1067–1074
Scahill L, Chappell PB, King RA, Leckman JF (2000) Pharmacologic treatment of tic disorders. Child Adolesc Psychiatr Clin N Am 9:99–117
Scahill L, Erenberg G, Berlin CM Jr, Budman C, Coffey BJ, Jankovic J, Kiessling L, King RA, Kurlan R, Lang A, Mink J, Murphy T, Zinner S, Walkup J (2006) Contemporary assessment and pharmacotherapy of Tourette syndrome. NeuroRx 3:192–206
Scahill L, Leckman JF, Schultz RT, Katsovich L, Peterson BS (2003) A placebo-controlled trial of risperidone in Tourette syndrome. Neurology 60:1130–1135
Scatton B, Cohen C, Perrault G, Oblin A, Claustre Y, Schoemaker H, Sanger DJ, Rouquier L, Porsolt R (2001) The preclinical pharmacologic profile of tiapride. Eur Psychiatry 16 Suppl 1:29s–34s
Schlander M, Schwarz O, Rothenberger A, Roessner V Tic disorders: prevalence and co-occurrence with attention-deficit/hyperactivity disorder in a German community sample. Eur Psychiatry. (in press)
Scott BL, Jankovic J, Donovan DT (1996) Botulinum toxin injection into vocal cord in the treatment of malignant coprolalia associated with Tourette’s syndrome. Mov Disord 11:431–433
Sears J, Patel NC (2008) Development of tics in a thirteen-year-old male following atomoxetine use. CNS Spectr 13:301–303
Segawa M (2003) Neurophysiology of Tourette’s syndrome: pathophysiological considerations. Brain Dev 25 (Suppl 1):S62–S69
Seo WS, Sung HM, Sea HS, Bai DS (2008) Aripiprazole treatment of children and adolescents with Tourette disorder or chronic tic disorder. J Child Adolesc Psychopharmacol 18:197–205
Serra-Mestres J, Ring H, Costa D (2004) Dopamine transporter binding in Gilles de la Tourette syndrome: A [123I]FP-CIT/SPECT study. Acta Psychiatr Scand 109:140–146
Shapiro AK, Shapiro E, Eisenkraft GJ (1983) Treatment of Gilles de la Tourette’s syndrome with clonidine and neuroleptics. Arch Gen Psychiatry 40:1235–1240
Shapiro AK, Shapiro ES, Young JG, Feinberg TE (1988) Measurement in tic disorders. In: Shapiro AK, Shapiro ES, Young JG, Feinberg TE (eds) Giles de la Tourette syndrome. Raven Press, New York, pp 127–193
Shapiro E, Shapiro AK, Fulop G, Hubbard M, Mandeli J, Nordlie J, Phillips RA (1989) Controlled study of haloperidol, pimozide and placebo for the treatment of Gilles de la Tourette’s syndrome. Arch Gen Psychiatry 46:722–730
Shapiro E, Shapiro E (1998) Treatment of tic disorders with haloperidol. In: Cohen DJ, Bruun RD, Leckman JF (eds) Tourette syndrome and tic disorders. Wiley, New York, pp 267–280
Shulman LM, Singer C, Weiner WJ (1995) Risperidone in Gilles de la Tourette syndrome. Neurology 45:1419
Shytle RD, Silver AA, Philipp MK, McConville BJ, Sanberg PR (1996) Transdermal nicotine for Tourette’s syndrome. Drug Dev Res 38:290–298
Silay YS, Vuong KD, Jankovic J (2004) The efficacy and safety of fluphenazine in patients with Tourette syndrome: P06.128. Neurology 62:A506
Silva RR, Magee HJ, Friedhoff AJ (1993) Persistent tardive dyskinesia and other neuroleptic-related dyskinesias in Tourette’s disorder. J Child Adoles Psychopharmacol 3:137–144
Silver AA, Sanberg PR (1993) Transdermal nicotine patch and potentiation of haloperidol in Tourette’s syndrome. Lancet 342:182
Silver AA, Shytle RD, Philipp MK, Sanberg PR (1996) Case study: long-term potentiation of neuroleptics with transdermal nicotine in Tourette’s syndrome. J Am Acad Child Adolesc Psychiatry 35:1631–1636
Silver AA, Shytle RD, Philipp MK, Sanberg PR (1995) Transdermal nicotine in Tourette’s Syndrome. In: Clarke PBS, Quik M, Thurau K (eds) The effects of nicotine on biological systems. Birkhauser Publishers, Boston, pp 293–299
Silver AA, Shytle RD, Philipp MK, Wilkinson BJ, McConville B, Sanberg PR (2001) Transdermal nicotine and haloperidol in Tourette’s disorder: a double-blind placebo-controlled study. J Clin Psychiatry 62:707–714
Singer HS (2010) Treatment of tics and Tourette syndrome. Curr Treat Options Neurol 12:539–561
Singer HS, Brown J, Quaskey S, Rosenberg LA, Mellits ED, Denckla MB (1995) The treatment of attention-deficit hyperactivity disorder in Tourette’s syndrome: a double-blind placebo-controlled study with clonidine and desipramine. Pediatrics 95:74–81
Singer HS, Gammon K, Quaskey S (1985) Haloperidol, fluphenazine and clonidine in Tourette syndrome: controversies in treatment. Pediatr Neurosci 12:71–74
Singer HS, Hahn IH, Moran TH (1991) Abnormal dopamine uptake sites in postmortem striatum from patients with Tourette’s syndrome. Ann Neurol 30:558–562
Singer HS, Szymanski S, Giuliano J, Yokoi F, Dogan AS, Brasic JR, Zhou Y, Grace AA, Wong DF (2002) Elevated intrasynaptic dopamine release in Tourette’s syndrome measured by PET. Am J Psychiatry 159:1329–1336
Singer HS, Wendlandt J, Krieger M, Giuliano J (2001) Baclofen treatment in Tourette syndrome: a double-blind, placebo-controlled, crossover trial. Neurology 56:599–604
Singh SK, Jankovic J (1988) Tardive dystonia in patients with Tourette’s syndrome. Mov Disord 3:274–280
Smith-Hicks CL, Bridges DD, Paynter NP, Singer HS (2007) A double-blind randomized placebo control trial of levetiracetam in Tourette syndrome. Mov Disord 22:1764–1770
Snider LA, Lougee L, Slattery M, Grant P, Swedo SE (2005) Antibiotic prophylaxis with azithromycin or penicillin for childhood-onset neuropsychiatric disorders. Biol Psychiatry 57:788–792
Spencer T, Biederman J, Coffey B, Geller D, Crawford M, Bearman SK, Tarazi R, Faraone SV (2002) A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry 59:649–656
Spencer TJ, Sallee FR, Gilbert DL, Dunn DW, McCracken JT, Coffey BJ, Budman CL, Ricardi RK, Leonard HL, Allen AJ, Milton DR, Feldman PD, Kelsey DK, Geller DA, Linder SL, Lewis DW, Winner PK, Kurlan RM, Mintz M (2008) Atomoxetine treatment of ADHD in children with comorbid Tourette syndrome. J Atten Disord 11:470–481
Srirompotong S, Saeseow P, Kharmwan S, Srirompotong S (2007) Ear wiggling tics: treatment with botulinum toxin injection. Eur Arch Otorhinolaryngol 264:385–387
Stahl SM, Berger PA (1980) Physostigmine in Gilles de la Tourette’s syndrome. N Engl J Med 302:298
Stahl SM, Berger PA (1981) Physostigmine in Tourette syndrome: evidence for cholinergic underactivity. Am J Psychiatry 138:240–242
Stahl SM, Shayegan DK (2003) The psychopharmacology of ziprasidone: receptor-binding properties and real-world psychiatric practice. J Clin Psychiatry 64 Suppl 19:6–12
Stamenkovic M, Aschauer H, Kasper S (1994) Risperidone for Tourette’s syndrome. Lancet 344:1577–1578
Stamenkovic M, Schindler SD, Aschauer HN, De Zwaan M, Willinger U, Resinger E, Kasper S (2000) Effective open-label treatment of tourette’s disorder with olanzapine. Int Clin Psychopharmacol 15:23–28
Steeves TD, Ko JH, Kideckel DM, Rusjan P, Houle S, Sandor P, Lang AE, Strafella AP (2010) Extrastriatal dopaminergic dysfunction in Tourette syndrome. Ann Neurol 67:170–181
Steingard RJ, Goldberg M, Lee D, DeMaso DR (1994) Adjunctive clonazepam treatment of tic symptoms in children with comorbid tic disorders and ADHD. J Am Acad Child Adolesc Psychiatry 33:394–399
Stenstrom AD, Sindo I (2008) Aripiprazole for the treatment of Tourette’s syndrome. Ugeskr Laeger 170:58
Stollberger C, Huber JO, Finsterer J (2005) Antipsychotic drugs and QT prolongation. Int Clin Psychopharmacol 20:243–251
Swain JE, Scahill L, Lombroso PJ, King RA, Leckman JF (2007) Tourette syndrome and tic disorders: a decade of progress. J Am Acad Child Adolesc Psychiatry 46:947–968
Sweet RD, Bruun R, Shapiro E, Shapiro AK (1974) Presynaptic catecholamine antagonists as treatment for Tourette syndrome. Effects of alpha methyl para tyrosine and tetrabenazine. Arch Gen Psychiatry 31:857–861
Thomas N, Swamidhas P, Russell S, Angothu H (2009) Tardive dyskinesia following risperidone treatment in Tourette’s syndrome. Neurol India 57:94–95
Toru M, Moriya H, Yamamoto K, Shimazono Y (1976) A double-blind comparison of sulpiride with chlordiazepoxide in neurosis. Folia Psychiatr Neurol Jpn 30:153–164
Tourette Syndrome Study Group (2002) Treatment of ADHD in children with tics: a randomized controlled trial. Neurology 58:527–536
Trillet M, Moreau T, Dalery J, de Villard R, Aimard G (1990) Treatment of Gilles de la Tourette’s disease with amisulpride. Presse Med 19:175
Trimble MR, Whurr R, Brookes G, Robertson MM (1998) Vocal tics in Gilles de la Tourette syndrome treated with botulinum toxin injections. Mov Disord 13:617–619
Troung DD, Bressman S, Shale H, Fahn S (1988) Clonazepam, haloperidol, and clonidine in tic disorders. South Med J 81:1103–1105
van der Linden C, Bruggeman R, van Woerkom TC (1994) Serotonin-dopamine antagonist and Gilles de la Tourette’s syndrome: an open pilot dose-titration study with risperidone. Mov Disord 9:687–688
van Wattum PJ, Chappell PB, Zelterman D, Scahill LD, Leckman JF (2000) Patterns of response to acute naloxone infusion in Tourette’s syndrome. Mov Disord 15:1252–1254
Varma SK, Messiha FS (1983) Endocrine aspects of lithium therapy in Tourette’s syndrome. Brain Res Bull 11:209–211
Vieregge P (1987) Tetrabenazine in the treatment of senile vocal tics. J Neurol 235:126–127
Vincent DA Jr (2008) Botulinum toxin in the management of laryngeal tics. J Voice 22:251–256
Weiden P, Bruun R (1987) Worsening of Tourette’s disorder due to neuroleptic-induced akathisia. Am J Psychiatry 144:504–505
Wetterling T, Mussigbrodt HE (1999) Weight gain: adverse reaction of atypical neuroleptics? J Clin Psychopharmacol 19:316–321
Wilner KD, Tensfeldt TG, Baris B, Smolarek TA, Turncliff RZ, Colburn WA, Hansen RA (2000) Single- and multiple-dose pharmacokinetics of ziprasidone in healthy young and elderly volunteers. Br J Clin Pharmacol 49 Suppl 1:15S–20S
Winter C, Heinz A, Kupsch A, Strohle A (2008) Aripiprazole in a case presenting with Tourette syndrome and obsessive-compulsive disorder. J Clin Psychopharmacol 28:452–454
Wolf DV, Wagner KD (1993) Tardive dyskinesia, tardive dystonia, and tardive Tourette’s syndrome in children and adolescents. J Child Adoles Psychopharmacol 3:175–198
Wong DF, Singer HS, Brandt J, Shaya E, Chen C, Brown J, Kimball AW, Gjedde A, Dannals RF, Ravert HT, Wilson PD, Wagner HN Jr (1997) D2-like dopamine receptor density in Tourette syndrome measured by PET. J Nucl Med 38:1243–1247
Yeh CB, Lee CH, Chou YH, Chang CJ, Ma KH, Huang WS (2006) Evaluating dopamine transporter activity with 99 mTc-TRODAT-1 SPECT in drug-naive Tourette’s adults. Nucl Med Commun 27:779–784
Yeh CB, Lee CS, Ma KH, Lee MS, Chang CJ, Huang WS (2007) Phasic dysfunction of dopamine transmission in Tourette’s syndrome evaluated with (99m)Tc TRODAT-1 imaging. Psychiatry Res 156:75–82
Yoo HK, Choi SH, Park S, Wang HR, Hong JP, Kim CY (2007) An open-label study of the efficacy and tolerability of aripiprazole for children and adolescents with tic disorders. J Clin Psychiatry 68:1088–1093
Yoo HK, Kim JY, Kim CY (2006) A pilot study of aripiprazole in children and adolescents with Tourette’s disorder. J Child Adolesc Psychopharmacol 16:505–506
Yoon DY, Gause CD, Leckman JF, Singer HS (2007) Frontal dopaminergic abnormality in Tourette syndrome: a postmortem analysis. J Neurol Sci 255:50–56
Yvonneau M, Bezard P (1970) Apropos of a case of Gilles de la Tourette’s disease blocked by sulpiride. Psycho-biological study. Encephale 59:439–459
Zawadzki Z (1972) Carbamazepine in the treatment of the maladie des tics. Pediatr Pol 47:1105–1110
Zykov VP, Shcherbina AY, Novikova EB, Shvabrina TV (2009) Neuroimmune aspects of the pathogenesis of Tourette’s syndrome and experience in the use of immunoglobulins in children. Neurosci Behav Physiol 39:635–638
Conflict of interest
Commercial firms and governmental organizations did not play a role in, or fund, the development of these guidelines. Kerstin J. Plessen, Renata Rizzo, Gerd Strand, Jeremy Stern, Cristiano Termine, Pieter J. Hoekstra declare that they have no conflict of interest. Veit Roessner: he has received lecture fees from Eli Lilly, Janssen-Cilag, Medice, Novartis, he was member of advisory boards of Eli Lilly, Novartis; Aribert Rothenberger (last three years): Advisory Board and Speakers Bureau of Lilly, Shire, Medice, Novartis, Research Support from Shire, German Research Society, Schwaabe, Travel Support from Shire, Educational Grant from Shire, Consultant of UCB/Shire, Lilly; Andrea G. Ludolph (last three years): she has received lecture fees from Janssen-Cilag, and research funding from Novartis, she was/is involved in clinical trials with Böhringer Ingelheim, Eli Lilly, Janssen-Cilag; Liselotte Skov received research support from the Lundbeck Foundation.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Members of the ESSTS Guidelines Group are listed in Appendix.
An erratum to this article can be found at http://dx.doi.org/10.1007/s00787-011-0195-z
Appendix: Members of the ESSTS Guidelines Group
Appendix: Members of the ESSTS Guidelines Group
Christos Androutsos, Harald Aschauer, Gillian Baird, Netty Bos-Veneman, Ariana Brambilla, Francesco Cardona, Danielle C. Cath, Andrea E. Cavanna, Virginie Czernecki, Sandra Dehning, Alan Eapter, Luca Farkas, Julia Gadaros, Andreas Hartmann, Elizabeth Hauser, Isabel Heyman, Tammy Hedderly, Pieter J. Hoekstra, Anne Korsgaard, Georgina M. Jackson, Linnea Larsson, Andrea G. Ludolph, Davide Martino, Claudia Menghetti, Nanette Mol Debes, Norbert Muller, Kirsten Muller-Vahl, Alexander Munchau, Tara Murphy, Richard Musil, Peter Nagy, Judith Nurnberger, Ben Oostra, Perry Paschou, Massimo Pasquini, Kerstin J. Plessen, Mauro Porta, Hugh Rickards, Renata Rizzo, Mary M. Robertson, Veit Roessner, Aribert Rothenberger, Domenico Servello, Liselotte Skov, Jeremy S. Stern, Gerd Strand, Zsannett Tarnok, Cristiano Termine, Jolande Van der Griendt, Cara Verdellen, Veerle Visser-Vandewalle, Ebba Wannag, Tomas Wolanczyck.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Roessner, V., Plessen, K.J., Rothenberger, A. et al. European clinical guidelines for Tourette syndrome and other tic disorders. Part II: pharmacological treatment. Eur Child Adolesc Psychiatry 20, 173–196 (2011). https://doi.org/10.1007/s00787-011-0163-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-011-0163-7