Abstract
Objectives
Fibroblast growth factors consist of receptor tyrosine kinase binding proteins involved in growth, differentiation, and regeneration of a variety of tissues of the head and neck. Their role in the development of teeth has been documented, and their presence in human odontogenic cysts and tumors has previously been investigated. Odontoma–dysphagia syndrome (OMIM 164330) is a very rare disorder characterized by clustering of teeth as compound odontoma, dysplasia and aplasia of teeth, slight craniofacial abnormalities, and dysphagia. We have followed the clinical course of the disease in a family over more than 30 years and have identified a genetic abnormality segregating with the disorder.
Materials and methods
We evaluated clinical data from nine different family members and obtained venous blood probes for genetic studies from three family members (two affected and one unaffected).
Results
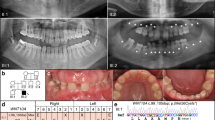
The present family with five patients in two generations has remained one out of only two known cases with this very rare syndrome. All those affected showed teeth dysplasia, oligodontia, and dysplasia and odontoma of the upper and lower jaw. Additional signs included dysphagia and strictures of the oesophagus. Comorbidity in one patient included aortic stenosis and coronary artery disease, requiring coronary bypasses and aortic valve replacement. Genome-wide SNP array analyses in three family members (two affected and one unaffected) revealed a microduplication of chromosome 11q13.3 spanning 355 kilobases (kb) and including two genes in full length, fibroblast growth factors 3 (FGF3) and 4 (FGF4).
Conclusion
The microduplication identified in this family represents the most likely cause of the odontoma–dysphagia syndrome and implies that the syndrome is caused by a gain of function of the FGF3 and FGF4 genes.
Clinical relevance
Mutations of FGF receptor genes can cause craniofacial syndromes such as odontoma–dysphagia syndrome. Following this train of thought, an evaluation of FGF gene family in sporadic odontoma could be worthwhile.








Similar content being viewed by others
References
Glickman I, Wuehrmann AH (1946) Compound composite odontoma; case report and histologic study. Am J Orthod Oral Surg 32:173–181
Nelson BL, Thompson LD (2010) Compound odontoma. Head Neck Pathol 4(4):290–291
Schmidseder R, Hausamen JE (1973) Familiäres Auftreten angeborener, multipler Odontome. Dtsch Zahnärztl Z 28:628–632
Bordini J Jr, Contar CM, Sarot JR, Fernandes A, Machado MA (2008) Multiple compound odontomas in the jaw: case report and analysis of the literature. J Oral Maxillofac Surg 66(12):2617–2620
Gardner EJ (1951) A genetic and clinical study of intestinal polyposis: a predisposing factor for carcinoma of the colon and rectum. Am J Hum Genet 3:167–176
Fotiadis C, Tsekouras DK, Antonakis P, Sfiniadakis J, Genetzakis M, Zografos GC (2005) Gardner’s syndrome: a case report and review of the literature. World J Gastroenterol 11(34):5408–5411
So F, Daley TD, Jackson L, Wysocki GP (2001) Immunohistochemical localization of fibroblast growth factors FGF-1 and FGF-2, and receptors FGFR2 and FGFR3 in the epithelium of human odontogenic cysts and tumors. J Oral Pathol Med 30(7):428–433
Ornitz DM (2005) FGF signaling in the developing endochondral skeleton. Cytokine Growth Factor Rev 16:205–213
Ornitz DM (2000) FGFs, heparan sulfate and FGFRs: complex interactions essential for development. Bioessays 22:108–112
Pispa J, Jung H-S, Jernvall J et al (1999) Cusp patterning defect in Tabby mouse teeth and its partial rescue by FGF. Dev Biol 216:521–534
Klein S, Roghani M, Rifkin DB (1997) Fibroblast growth factors as angiogenesis factors: new insights into their mechanism of action. In: Goldberg ID, Rosen EM (eds) Regulation of angiogenesis. Birkhauser Verlag, Basel, pp 159–183
Kettunen P, Thesleff I (1998) Expression and function of FGFs-4, -8, and -9 suggest functional redundancy and repetitive use as epithelial signals during tooth morphogenesis. Dev Dyn 211:256–268
Niswander L, Martin GR (1992) FGF-4 expression during gastrulation, myogenesis, limb and tooth development in the mouse. Development 114:755–768
Jehee FS, Bertola DR, Yelavarthi KK, Krepischi-Santos AC, Kim C, Vianna-Morgante AM, Vermeesch JR, Passos-Bueno MR (2007) An 11q11–q13.3 duplication, including FGF3 and FGF4 genes, in a patient with syndromic multiple craniosynostoses. Am J Med Genet A 143A(16):1912–1918
Herrmann M (1957) Über vom Zahnsystem ausgehende Tumore bei Kindern. Fortschr Kiefer Gesichtschir 3:257
Herrmann M (1958) Über vom Zahnsystem ausgehende Tumore bei Kindern. Fortschr Kiefer Gesichtschir 3:257
Yamaguchi TP, Rossant J (1995) Fibroblast growth factors in mammalian development. Curr Opin Genet Dev 5(4):485–491
Wilkinson DG, Bhatt S, McMahon AP (1989) Expression pattern of the FGF-related proto-oncogene int-2 suggests multiple roles in fetal development. Development 105(1):131–136
Jernvall J, Kettunen P, Karavanova I, Martin LB, Thesleff I (1994) Evidence for the role of the enamel knot as a control center in mammalian tooth cusp formation: non-dividing cells express growth stimulating Fgf-4 gene. Int J Dev Biol 38(3):463–469
Cohen MM Jr (1995) Craniosynostoses: phenotypic/molecular correlations. Am J Med Genet 56(3):334–339
Boder G (1967) Odontomatosis (multiple odontomas). Oral Surg 23:770–773
Schonberger W (1974) Angeborene multiple Odontome und Dysphagie bei Vater und Sohn-eine syndromhafte Verknuepfung? Z Kinderheilk 117:101–108
Gregory-Evans CY, Moosajee M, Hodges MD, Mackay DS, Game L, Vargesson N, Bloch-Zupan A, Ruschendorf F, Santos-Pinto L, Wackens G, Gregory-Evans K (2007) SNP genome scanning localizes oto-dental syndrome to chromosome 11q13 and microdeletions at this locus implicate FGF3 in dental and inner-ear disease and FADD in ocular coloboma. Hum Mol Genet 16:2482–2493
Hillbertz NHCS, Isaksson M, Karlsson EK, Hellmen E, Pielberg GR, Savolainen P, Wade CM, von Euler H, Gustafson U, Hedhammar A, Nilsson M, Lindblad-Toh K, Andersson L, Andersson G (2007) Duplication of FGF3, FGF4, FGF19 and ORAOV1 causes hair ridge and predisposition to dermoid sinus in Ridgeback dogs. Nat Genet 39:1318–1320
Acknowledgments
We would like to thank the family very much for their support of this study.
Conflicts of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ziebart, T., Draenert, F.G., Galetzka, D. et al. The original family revisited after 37 years: odontoma–dysphagia syndrome is most likely caused by a microduplication of chromosome 11q13.3, including the FGF3 and FGF4 genes. Clin Oral Invest 17, 123–130 (2013). https://doi.org/10.1007/s00784-012-0676-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-012-0676-6