Abstract
Introduction
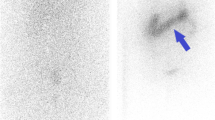
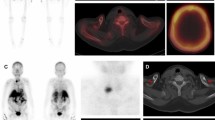
Brown tumors (BT) are non-neoplastic bone lesions infrequently occurring in patients with long-standing severe hyperparathyroidism (HPT). BT may be identified and characterized using 18-F-sodium fluoride-positron-emission-tomography/computed tomography (18F-NaF-PET/CT). We present a retrospective series of eight primary hyperparathyroidism (pHPT) patients with BT imaged with 18F-NaF-PET/CT.
Materials and methods
Imaging assessment included location, diameter, maximum standardized uptake value (SUVmax), metabolically active lesion volume (PETvol) of BT, total metabolically active bone volume (TMBvol) per patient and several computed tomography (CT) features of BT. Where appropriate, we analyzed the association between characteristic features of BT in 18F-NaF-PET/CT, histopathology, clinical symptomatology and laboratory parameters.
Results
In our cohort of 8 patients (median age, 49 years, range, 26–73), 72 BT were found. The mean PETvol of BT was 89.48 cm3 ± 122.81 cm3 and the mean SUVmax was 17.5 ± 7.8. The total PETvol of BT per patient correlated positively with serum calcium (r = 0.810, p = 0.015), and negatively with glomerular filtration rate (GFR) (r = − 0.762, p = 0.028). TMBvol correlated significantly with serum PTH (r = 0.810, p = 0.015), alkaline phosphatase (r = 0.762, p = 0.028), and duration of postoperative hospitalization (r = 0.826, p = 0.011, 24.3 days ± 19.8 days).
Conclusion
18F-NaF-PET/CT is a valuable non-invasive whole-body imaging technique for the assessment of patients with pHPT and BT. TMBvol is associated with PTH and alkaline phosphatase, and the requirement for intense postoperative calcium substitution, which determines the duration of hospitalization.



Similar content being viewed by others
Abbreviations
- 18F-NaF:
-
Sodium fluoride labeled with fluorine-18
- 25-OH-ViD:
-
25-Hydroxy-vitamin-D3
- 18F-FDG:
-
Fluorodeoxyglucose labeled with fluorine-18
- BMD:
-
Bone mineral density
- BT:
-
Brown tumors
- CT:
-
Computed tomography
- DXA:
-
Dual energy X-ray absorptiometry
- FOV:
-
Field of view
- GFR:
-
Glomerular filtration rate
- HBS:
-
Hungry bone syndrome
- MBq:
-
Megabecquerel
- OFC:
-
Osteitis fibrosa cystica
- PET/CT:
-
Positron-emission-tomography/computed tomography
- PETvol :
-
Metabolic volume
- pHPT:
-
Primary hyperparathyroidism
- PTH:
-
Parathyroid hormone
- SD:
-
Standard deviation
- sHPT:
-
Secondary hyperparathyroidism
- SUVmax :
-
Maximum standardized uptake value
- TMBvol :
-
Total metabolically active bone volume per patient
- VOI:
-
Volume of interest
References
Collip JP (1925) The extraction of a parathyroid hormone which will prevent or control parathyroid tetany and which regulates the level of blood calcium. J Biol Chem 63:395
Albright F, Aub JC, Bauer W (1934) Hyperparathyroidism: a common and polymorphic condition as illustrated by seventeen proved cases from one clinic. JAMA 104:9–35
Albright F, Reifenstein EC (1948) The parathyroid glands and metabolic bone disease, selected studies. The Williams and Wilkins Company, Baltimore
Varma R, Kim YJ, Garjian K, Barank D (2014) Hyperparathyroidism and hungry bone syndrome revisited. Clin Nucl Med 39:704–706. https://doi.org/10.1097/RLU.0000000000000500
Irie T, Mawatari T, Ikemura S, Matsui G, Iguchi T, Mitsuyasu H (2015) Brown tumor of the patella caused by primary hyperparathyroidism: a case report. Korean J Radiol 16:613–616. https://doi.org/10.3348/kjr.2015.16.3.613
Schnyder MA, Stolzmann P, Huber GF, Schmid C (2017) A patient with a history of breast cancer and multiple bone lesions: a case report. J Med Case Rep 11:127. https://doi.org/10.1186/s13256-017-1296-1
Brasier AR, Nussbaum SR (1988) Hungry bone syndrome: clinical and biochemical predictors of its occurrence after parathyroid surgery. Am J Med 84:654–660
Guney E, Yigitbasi OG, Bayram F, Ozer V, Canoz O (2001) Brown tumor of the maxilla associated with primary hyperparathyroidism. Auris Nasus Larynx 28:369–372
Scott SN, Graham SM, Sato Y, Robinson RA (1999) Brown tumor of the palate in a patient with primary hyperparathyroidism. Ann Otol Rhinol Laryngol 108:91–94. https://doi.org/10.1177/000348949910800114
Hermoye A, Malghem J, Lecouvet F, Goffin E, Lonneux M, Lhommel R (2009) F-18 FDG PET/CT as a noninvasive diagnostic and follow-up tool in brown tumors due to secondary hyperparathyroidism. Clin Nucl Med 34:330–332. https://doi.org/10.1097/RLU.0b013e31819e52a9
Kuwahara K, Izawa S, Murabe H, Murakami N, Yokota T, Wani Y, Notohara K, Tsukayama C, Ishimori T, Watanabe Y (2007) Increased 18F-fluorodeoxyglucose uptake in a brown tumor in a patient with primary hyperparathyroidism. J Clin Endocrinol Metab 92:2408–2409. https://doi.org/10.1210/jc.2007-0591
Gahier Penhoat M, Drui D, Ansquer C, Mirallie E, Maugars Y, Guillot P (2017) Contribution of 18-FDG PET/CT to brown tumor detection in a patient with primary hyperparathyroidism. Jt Bone Spine 84:209–212. https://doi.org/10.1016/j.jbspin.2016.06.007
Sager S, Aliyev A, Halac M, Ozturk T (2012) Positron emission tomography/computed tomography imaging of brown tumors mimicking multiple skeletal metastases in patient with primary hyperparathyroidism. Indian J Endocrinol Metab 16:850–852. https://doi.org/10.4103/2230-8210.100682
Huellner MW, Appenzeller P, Kuhn FP, Husmann L, Pietsch CM, Burger IA, Porto M, Delso G, von Schulthess GK, Veit-Haibach P (2014) Whole-body nonenhanced PET/MR versus PET/CT in the staging and restaging of cancers: preliminary observations. Radiology 273:859–869. https://doi.org/10.1148/radiol.14140090
Bastawrous S, Bhargava P, Behnia F, Djang DS, Haseley DR (2014) Newer PET application with an old tracer: role of 18F-NaF skeletal PET/CT in oncologic practice. Radiographics 34:1295–1316. https://doi.org/10.1148/rg.345130061
Czernin J, Satyamurthy N, Schiepers C (2010) Molecular mechanisms of bone 18F-NaF deposition. J Nucl Med 51:1826–1829. https://doi.org/10.2967/jnumed.110.077933
Even-Sapir E, Metser U, Flusser G, Zuriel L, Kollender Y, Lerman H, Lievshitz G, Ron I, Mishani E (2004) Assessment of malignant skeletal disease: initial experience with 18F-fluoride PET/CT and comparison between 18F-fluoride PET and 18F-fluoride PET/CT. J Nucl Med 45:272–278
Fischer DR, Zweifel K, Treyer V, Hesselmann R, Johayem A, Stumpe KD, von Schulthess GK, Hany TF, Strobel K (2011) Assessment of successful incorporation of cages after cervical or lumbar intercorporal fusion with [(18)F]fluoride positron-emission tomography/computed tomography. Eur Spine J 20:640–648. https://doi.org/10.1007/s00586-010-1643-z
Fischer DR, Pfirrmann CW, Zubler V, Stumpe KD, Seifert B, Strobel K, Tamborrini G, von Schulthess GK, Michel BA, Ciurea A (2012) High bone turnover assessed by 18F-fluoride PET/CT in the spine and sacroiliac joints of patients with ankylosing spondylitis: comparison with inflammatory lesions detected by whole body MRI. EJNMMI Res 2:38. https://doi.org/10.1186/2191-219X-2-38
Jadvar H, Desai B, Conti PS (2015) Sodium 18F-fluoride PET/CT of bone, joint, and other disorders. Semin Nucl Med 45:58–65. https://doi.org/10.1053/j.semnuclmed.2014.07.008
Hardoff R, Frajewicki V (1996) Bone scintigraphy in hungry bone syndrome following parathyroidectomy. J Nucl Med 37:1371–1373
Meng Z, Zhu M, He Q, Tian W, Zhang Y, Jia Q, Tan J (2011) Clinical implications of brown tumor uptake in whole-body 99 mTc-sestamibi scans for primary hyperparathyroidism. Nucl Med Commun 32:708–715. https://doi.org/10.1097/MNM.0b013e328347b582
Fonager RF, Zacho HD, Langkilde NC, Fledelius J, Ejlersen JA, Haarmark C, Hendel HW, Lange MB, Jochumsen MR, Mortensen JC, Petersen LJ (2017) Diagnostic test accuracy study of (18)F-sodium fluoride PET/CT, (99m)Tc-labelled diphosphonate SPECT/CT, and planar bone scintigraphy for diagnosis of bone metastases in newly diagnosed, high-risk prostate cancer. Am J Nucl Med Mol Imaging 7:218–227
Strobel K, Vali R (2012) (18)F NaF PET/CT versus conventional bone scanning in the assessment of benign bone disease. PET Clin 7:249–261. https://doi.org/10.1016/j.cpet.2012.04.007
Schirrmeister H, Guhlmann A, Elsner K, Kotzerke J, Glatting G, Rentschler M, Neumaier B, Trager H, Nussle K, Reske SN (1999) Sensitivity in detecting osseous lesions depends on anatomic localization: planar bone scintigraphy versus 18F PET. J Nucl Med 40:1623–1629
Beheshti M, Mottaghy FM, Paycha F, Behrendt FFF, Van den Wyngaert T, Fogelman I, Strobel K, Celli M, Fanti S, Giammarile F, Krause B, Langsteger W (2015) (18)F-NaF PET/CT: EANM procedure guidelines for bone imaging. Eur J Nucl Med Mol Imaging 42:1767–1777. https://doi.org/10.1007/s00259-015-3138-y
Hetzel M, Arslandemir C, Konig HH, Buck AK, Nussle K, Glatting G, Gabelmann A, Hetzel J, Hombach V, Schirrmeister H (2003) F-18 NaF PET for detection of bone metastases in lung cancer: accuracy, cost-effectiveness, and impact on patient management. J Bone Miner Res 18:2206–2214. https://doi.org/10.1359/jbmr.2003.18.12.2206
Mick CG, James T, Hill JD, Williams P, Perry M (2014) Molecular imaging in oncology: (18)F-sodium fluoride PET imaging of osseous metastatic disease. AJR Am J Roentgenol 203:263–271. https://doi.org/10.2214/AJR.13.12158
Pontes FSC, Lopes MA, de Souza LL, Santos Dos, da Mata Rezende D, Santos-Silva AR, Jorge J Jr, da Silva WG, Pires FR, Rocha AC, de Campos WG, Caldato MCF, Martin RM, Fonseca FP, Pontes HAR (2018) Oral and maxillofacial manifestations of chronic kidney disease-mineral and bone disorder: a multicenter retrospective study. Oral Surg Oral Med Oral Pathol Oral Radiol 125:31–43. https://doi.org/10.1016/j.oooo.2017.09.011
Knowles NG, Smith DL, Outwater EK (2008) MRI diagnosis of brown tumor based on magnetic susceptibility. J Magn Reson Imaging 28:759–761. https://doi.org/10.1002/jmri.21441
Rejnmark L, Vestergaard P, Mosekilde L (2011) Nephrolithiasis and Renal Calcifications in Primary Hyperparathyroidism. J Clin Endocrinol Metabol 96:2377–2385
Sommerauer M, Graf C, Schafer N, Huber G, Schneider P, Wuthrich R, Schmid C, Steinert H (2015) Sensitivity and specificity of dual-isotope 99mTc-tetrofosmin and 123I sodium iodide single photon emission computed tomography (SPECT) in hyperparathyroidism. PLoS One 10:e0129194. https://doi.org/10.1371/journal.pone.0129194
Jorda M, Rey L, Hanly A, Ganjei-Azar P (2000) Fine-needle aspiration cytology of bone: accuracy and pitfalls of cytodiagnosis. Cancer 90:47–54
Khalbuss WE, Teot LA, Monaco SE (2010) Diagnostic accuracy and limitations of fine-needle aspiration cytology of bone and soft tissue lesions: a review of 1114 cases with cytological-histological correlation. Cancer Cytopathol 118:24–32. https://doi.org/10.1002/cncy.20058
Vigorita VJ, Anand VS, Einhorn TA (1986) Sampling error in diagnosing hyperparathyroid changes in bone in small needle biopsies. Am J Surg Pathol 10:140–142. https://doi.org/10.1097/00000478-198602000-00007
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Study design: C.G., M.H., O.T., C.S. Study conduct: C.S. Data collection: C.G., M.H., B.B., C.S. Data analysis: C.G., M.H., C.S. Data interpretation: C.G., M.H., O.T., B.B., C.S. Drafting manuscript: C. G., M.H. Revising manuscript content: C.G., M.H., O.T., B.B., C.S. Approving final version of manuscript: C.G., M.H., O.T., B.B., C.S. C.G., M.H. and C.S. take responsibility for the integrity of the data analysis.
Corresponding author
Ethics declarations
Conflict of interest
MH is a recipient of IIS grants, institutional grants, and speaker’s fees from GE Healthcare, and has received funding from the Alfred and Annemarie von Sick grant for translational and clinical cardiac and oncological research. All other authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Graf, C., Huellner, M., Tschopp, O. et al. 18F-NaF-PET/CT in patients with primary hyperparathyroidism and brown tumors. J Bone Miner Metab 38, 299–309 (2020). https://doi.org/10.1007/s00774-019-01059-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-019-01059-z