Abstract
We have developed a fast method for sensitive extraction and determination of the metal ions silver(I), gold(III), copper(II) and palladium(II). Fe3O4 magnetic nanoparticles were modified with polythiophene and used for extraction the metal ions without a chelating agent. Following extraction, the ions were determined by flow injection inductively coupled plasma optical emission spectrometry. The influence of sample pH, type and volume of eluent, amount of adsorbent, sample volume and time of adsorption and desorption were optimized. Under the optimum conditions, the calibration plots are linear in the 0.75 to 100 μg L−1 concentration range (R2 > 0.998), limits of detection in the range from 0.2 to 2.0 μg L−1, and enhancement factors in the range from 70 to 129. Precisions, expressed as relative standard deviations, are lower than 4.2 %. The applicability of the method was demonstrated by the successful analysis of tap water, mineral water, and river water.

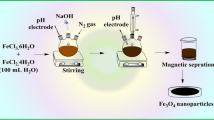
In the present work, polythiophene-coated Fe3O4 nanoparticles have been successfully synthesized and were applied as adsorbent for magnetic solid-phase extraction of some precious metal ions.



Similar content being viewed by others
References
Sheoran V, Sheoran AS (2009) Phytomining: a review. Miner Eng 22:1007–1019
Cui J, Zhang L (2008) Metallurgical recovery of metals from electronic waste: a review. J Hazard Mater 158:228–256
El-Menshawy AM, Kenawy IM, El-Asmy AA (2010) Modification of chloromethylated polystyrene with 2-mercabtobenzothiazole for application as a new sorbent for preconcentration and determination of Ag+ from different matrices. J Hazard Mater 173:523–527
Kolthoff IM, Elving PJ (1966) Treatise on Analytical Chemistry. Part II, Vol. 5. Interscience New York
Environmental Protection Agency (EPA) (1980) Ambient Water Quality Criteria for Silver. EPA 4405-80-071, Office of Water Regulations Washington DC
Meian E (Ed.) (1991) Metals and Their Compounds in the Environment, VCH, New York
Saavedra R, Soto C, Yañez J, Toral MI (2009) Determination of Pd(II) content in catalysts and tap water samples via photoacoustic spectroscopy analysis of Pd(II)-3-[2′-thiazolylazo]-2,6-diaminopyridine complex on solid phase. J Hazard Mater 167:970–975
Wiseman Clare L (2006) Palladium from catalytic converters: exposure levels and human risk. In: Zereini F, Alt F (eds) Palladium emissions in the environment. Springer, Berlin, pp 565–574
Jarvis K, Parry S, Piper J (2001) Temporal and spatial studies of autocatalyst derived platinum, rhodium, and palladium and selected vehicle-derived trace elements in the environment. Environ Sci Technol 35:1031–1036
World Health Organization (2002) Environmental Health Criteria 226-Palladium, World Health Organization, International Programme on Chemical Safety, Geneva
Kielhorn J, Melber C, Keller D, Mangelsdorf I (2002) Palladium-a review of exposure and effects to human health. Int J Hyg Environ Health 205:417–432
Huang X, Wang Y, Liao X, Shi B (2010) Adsorptive recovery of Au3+ from aqueous solutions using bayberry tannin-immobilized mesoporous silica. J Hazard Mater 183:793–798
Suwalsky M, González R, Villena F, Aguilar LF, Sotomayor CP, Bolognin S, Zatta P (2010) Human erythrocytes and neuroblastoma cells are affected in vitro by Au(III) ions. Biochem Biophys Res Commun 397:226–231
Sun X, Huang X, Xp L, Shi B (2011) Adsorptive removal of Cu(II) from aqueous solutions using collagen-tannin resin. J Hazard Mater 186:1058–1063
Rengaraj S, Kim Y, Joo CK, Yi J (2004) Removal of Cu(II) from aqueous solution by aminated and protonated mesoporous aluminas: kinetics and equilibrium. J Colloid Interface Sci 273:14–21
Lubick N (2007) Revisiting phosphorus in the Everglades. Environ Sci Technol 41:7954–7955
Senturk HB, Gundogdu A, Bulut VN, Duran C, Soylak M, Elci L, Tufekci M (2007) Separation and enrichment of gold(III) from environmental samples prior to its flame atomic absorption spectrometric determination. J Hazard Mater 149:317–323
Afzali D, Mostafavi A, Mirzaei M (2010) Preconcentration of gold ions from water samples by modified organo-nanoclay sorbent prior to flame atomic absorption spectrometry determination. J Hazard Mater 181:957–961
Yalcinkaya O, Kalfa OM, Turker AR (2011) Chelating agent free-solid phase extraction (CAF-SPE) of Co(II), Cu(II) and Cd(II) by new nano hybrid material (ZrO2/B2O3). J Hazard Mater 195:332–339
Mashhadizadeh MH, Karami Z (2011) Solid phase extraction of trace amounts of Ag, Cd, Cu, and Zn in environmental samples using magnetic nanoparticles coated by 3-(trimethoxysilyl)-1-propantiol and modified with 2-amino-5-mercapto-1,3,4-thiadiazole and their determination by ICP-OES. J Hazard Mater 190:1023–1029
Yamini Y, Moradi M, Tahmasebi E (2012) High-throughput quantification of palladium in water samples by ion pair based-surfactant assisted microextraction. Anal Chim Acta 728:26–30
Ranjbar L, Yamini Y, Saleh A, Seidi S, Faraji M (2012) Ionic liquid based dispersive liquid-liquid microextraction combined with ICP-OES for the determination of trace quantities of cobalt, copper, manganese, nickel and zinc in environmental water samples. Microchim Acta 177:119–127
Yamini Y, Rezaee M, Khanchi A, Faraji M, Saleh A (2010) Dispersive liquid–liquid microextraction based on the solidification of floating organic drop followed by inductively coupled plasma-optical emission spectrometry as a fast technique for the simultaneous determination of heavy metals. J Chromatogr A 1217:2358–2364
Jiang X, Huang K, Deng D, Xia H, Hou X, Zheng C (2012) Nanomaterials in analytical atomic spectrometry. Trends Anal Chem 39:38–59
Chen L, Wang T, Tong J (2011) Application of derivatized magnetic materials to the separation and the preconcentration of pollutants in water samples. Trends Anal Chem 30:1095–1108
Tahmasebi E, Yamini Y, Mehdinia A, Rouhi F (2012) Polyaniline-coated Fe3O4 nanoparticles: an anion exchange magnetic sorbent for solid-phase extraction. J Sep Sci 35:2256–2265
Jiang H, Yan Z, Zhao Y, Hu X, Lian H (2012) Zincon-immobilized silica-coated magnetic Fe3O4 nanoparticles for solid-phase extraction and determination of trace lead in natural and drinking waters by graphite furnace atomic absorption spectrometry. Talanta 94:251–256
Zhai Y, He Q, Han Q, Duan S (2012) Solid-phase extraction of trace metal ions with magnetic nanoparticles modified with 2,6-diaminopyridine. Microchim Acta 178:405–412
Cheng G, He M, Peng H, Hu B (2012) Dithizone modified magnetic nanoparticles for fast and selective solid phase extraction of trace elements in environmental and biological samples prior to their determination by ICP-OES. Talanta 88:507–515
Tahmasebi E, Yamini Y (2012) Facile synthesis of new nano sorbent for magnetic solid-phase extraction by self assembling of bis-(2,4,4-trimethylpentyl)-dithiophosphinic acid on Fe3O4@Ag core@shell nanoparticles: Characterization and application. Anal Chim Acta 756:13–22
Ngomsik AF, Bee A, Talbot D, Cote G (2012) Magnetic solid–liquid extraction of Eu(III), La(III), Ni(II) and Co(II) with maghemite nanoparticles. Sep Purif Technol 86:1–8
Sadeghi S, Azhdari H, Arabi H, Zeraatkar Moghaddam A (2012) Surface modified magnetic Fe3O4 nanoparticles as a selective sorbent for solid phase extraction of uranyl ions from water samples. J Hazard Mater 215–216:208–216
Jainae K, Sanuwong K, Nuangjamnong J, Sukpirom N, Unob F (2010) Extraction and recovery of precious metal ions in wastewater by polystyrene-coated magnetic particles functionalized with 2-(3-(2-aminoethylthio)propylthio)ethanamine. Chem Eng J 60:586–593
Tahmasebi E, Yamini Y, Moradi M, Esrafily A (2013) Polythiophene-coated Fe3O4 superparamagnetic nanocomposite: synthesis and application as a new sorbent for solid-phase extraction. Anal Chim Acta 770:68–74
Pearson GR (1963) Hard and soft acids and bases. J Am Chem Soc 85:3533–3539
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 226 kb)
Rights and permissions
About this article
Cite this article
Tahmasebi, E., Yamini, Y. Polythiophene-coated Fe3O4 nanoparticles as a selective adsorbent for magnetic solid-phase extraction of silver(I), gold(III), copper(II) and palladium(II). Microchim Acta 181, 543–551 (2014). https://doi.org/10.1007/s00604-013-1144-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-013-1144-y