Abstract
Aim
It has been suggested that glucose levels ≥155 mg/dl at 1-h during an oral glucose tolerance test (OGTT) may predict development of type 2 diabetes and cardiovascular events among adults with normal glucose tolerance (NGT 1 h-high). Studies showed a link between increased blood viscosity and type 2 diabetes. However, whether blood viscosity is associated with dysglycemic conditions such as NGT 1 h-high, impaired glucose tolerance (IGT) or impaired fasting glucose (IFG) is unsettled.
Methods
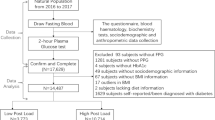
1723 non-diabetic adults underwent biochemical evaluation and OGTT. A validated formula based on hematocrit and total plasma proteins was employed to estimate whole blood viscosity. Subjects were categorized into NGT with 1 h glucose <155 mg/dL (NGT-1 h-low), NGT-1 h-high, IFG and/or IGT.
Results
Hematocrit and blood viscosity values appeared significantly higher in individuals with NGT 1 h-high, IFG and/or IGT as compared to NGT 1 h-low subjects. Blood viscosity was significantly correlated with age, waist circumference, blood pressure, HbA1c, fasting, 1- and 2-h post-challenge insulin levels, total cholesterol and low-density lipoprotein, triglycerides, fibrinogen, white blood cell, and inversely correlated with high-density lipoprotein and insulin sensitivity. Of the four glycemic parameters, 1-h post-challenge glucose showed the strongest correlation with blood viscosity (β = 0.158, P < 0.0001) in a multivariate regression analysis model including several atherosclerosis risk factors.
Conclusions
Our results demonstrate a positive relationship between blood viscosity and 1-h post-challenge plasma glucose. They also suggest that a subgroup of NGT individuals with 1-h post-challenge plasma >155 mg/dl have increased blood viscosity comparable to that observed in subjects with IFG and/or IGT.

Similar content being viewed by others
References
Ciuffetti G, Schillaci G, Lombardini R, Pirro M, Vaudo G, Mannarino E (2005) Prognostic impact of low-shear whole blood viscosity in hypertensive men. Eur J Clin Invest 35:93–98
Rillaerts E, van Gaal L, Xiang DZ, Vansant G, De Leeuw I (1989) Blood viscosity in human obesity: relation to glucose tolerance and insulin status. Int J Obes 13:739–745
Sloop GD, Garber DW (1997) The effects of low-density lipoprotein and high-density lipoprotein on blood viscosity correlate with their association with risk of atherosclerosis in humans. Clin Sci 92:473–479
Ernst E (1995) Haemorheological consequences of chronic cigarette smoking. J Cardiovasc Risk 2:435–439
Letcher RL, Chien S, Pickering TG, Laragh JH (1983) Elevated blood viscosity in patients with borderline essential hypertension. Hypertension 5:757–762
Irace C, Carallo C, Scavelli F, De Franceschi MS, Esposito T, Gnasso A (2014) Blood viscosity in subjects with normoglycemia and prediabetes. Diabetes Care 37:488–492
Devereux RB, Drayer JIM, Chien S et al (1984) Whole blood viscosity as a determinant of cardiac hypertrophy in systemic hypertension. Am J Cardiol 54:592–595
Zannad F, Voisin P, Brunotte F, Bruntz JF, Stoltz JF, Gilgenkrantz JM (1988) Haemorheological abnormalities in arterial hypertension and their relation to cardiac hypertrophy. J Hypertens 6:293–297
Lee AJ, Mowbray PI, Lowe GD, Rumley A, Fowkes FG, Allan PL (1998) Blood viscosity and elevated carotid intima-media thickness in men and women: the Edinburgh Artery study. Circulation 97:1467–1473
Zhao HY, Li J, Xu M et al (2015) Elevated whole blood viscosity is associated with insulin resistance and non-alcoholic fatty liver. Clin Endocrinol 83:806–811
Tripolino C, Irace C, Carallo C et al (2016) Association between blood viscosity and common carotid artery elasticity. Clin Hemorheol Microcirc 62:55–62
Tohgi H, Yamanouchi H, Murakami M, Kameyama M (1978) Importance of the hematocrit as a risk factor in cerebral infarction. Stroke 9:369–374
Neumann FJ, Katus HA, Hoberg E (1991) Increased plasma viscosity and erythrocyte aggregation: indicators of an unfavourable clinical outcome in patients with unstable angina pectoris. Br Heart J 66:425–430
Yarnell JWG, Baker IA, Sweetnam PM et al (1991) Fibrinogen, viscosity, and white blood cell count are major risk factors for ischemic heart disease. The Caerphilly and Speedwell collaborative heart disease studies. Circulation 83:836–844
Lowe G, Lee A, Rumley A, Price J, Fowkes F (1997) Blood viscosity and risk of cardiovascular events: the Edinburgh Artery study. Br J Haematol 96:168–173
Høieggen A, Fossum E, Moan A, Enger E, Kjeldsen SE (1998) Whole-blood viscosity and the insulin-resistance syndrome. J Hypertens 16:203–210
Nordby G, Moan A, Kjeldsen SE, Os I (1995) Relationship between hemorheological factors and insulin sensitivity in normotensive and hypertensive premenopausal women. Am J Hypertens 8:439–444
Catalano C, Muscelli E, Natali A et al (1997) Reciprocal association between insulin sensitivity and the haematocrit in man. Eur J Clin Invest 27:634–637
Facchini FS, Carantoni M, Jeppesen J, Reaven GM (1998) Hematocrit and hemoglobin are independently related to insulin resistance and compensatory hyperinsulinemia in healthy, non-obese men and women. Metabolism 47:831–835
Hanley AJ, Retnakaran R, Qi Y et al (2009) Association of hematological parameters with insulin resistance and beta-cell dysfunction in nondiabetic subjects. J Clin Endocrinol Metab 94:3824–3832
Medalie JH, Papier CM, Goldbourt U, Herman JB (1975) Major factors in the development of diabetes mellitus in 10,000 men. Arch Intern Med 135:811–817
Wannamethee SG, Perry IJ, Shaper AG (1996) Hematocrit and risk of NIDDM. Diabetes 45:576–579
Nakanishi N, Suzuki K, Tatara K (2004) Haematocrit and risk of development of type 2 diabetes mellitus in middle-aged Japanese men. Diabet Med 21:476–482
Tulloch-ReidMK Hanson RL, Saremi A, Looker HC, Williams DE, Krakoff J, Knowler WC (2004) Hematocrit and the incidence of type 2 diabetes in the Pima Indians. Diabetes Care 27:2245–2246
Tamariz LJ, Young JH, Pankow JS et al (2008) Blood viscosity and hematocrit as risk factors for type 2 diabetes mellitus: the atherosclerosis risk in communities (ARIC) study. Am J Epidemiol 168:1153–1160
Capoğlu I, Unüvar N, Bektaş Y, Yilmaz O, Kaya MD (2002) The effects of high haematocrit levels on glucose metabolism disorders. J Int Med Res 30:433–437
Richards RS, Nwose EU (2010) Blood viscosity at different stages of diabetes pathogenesis. Br J Biomed Sci 67(2):67–70
Abdul-Ghani MA, Abdul-Ghani T, Ali N, Defronzo RA (2008) One-hour plasma glucose concentration and the metabolic syndrome identify subjects at high risk for future type 2 diabetes. Diabetes Care 31:1650–1655
Abdul-Ghani MA, Lyssenko V, Tuomi T, DeFronzo RA, Groop L (2009) Fasting versus postload plasma glucose concentration and the risk for future type 2 diabetes: results from the Botnia study. Diabetes Care 32:281–286
Fiorentino TV, Marini MA, Andreozzi F et al (2015) One-hour postload hyperglycemia is a stronger predictor of type 2 diabetes than impaired fasting glucose. J Clin Endocrinol Metab 100:3744–3751
Jagannathan R, Sevick MA, Fink D et al (2016) The 1-h post-load glucose level is more effective than HbA1c for screening dysglycemia. Acta Diabetol 53:543–550
Marini MA, Succurro E, Frontoni S et al (2012) Insulin sensitivity, β-cell function, and incretin effect in individuals with elevated 1-h postload plasma glucose levels. Diabetes Care 35:868–872
Andreozzi F, Mannino GC, Perticone M, Perticone F, Sesti G (2017) Elevated 1-h post-load plasma glucose levels in subjects with normal glucose tolerance are associated with a pro-atherogenic lipid profile. Atherosclerosis 256:15–20
Fiorentino TV, Andreozzi F, Mannino GC et al (2016) One-hour postload hyperglycemia confers higher risk of hepatic steatosis to HbA1c-defined prediabetic subjects. J Clin Endocrinol Metab 101:4030–4038
Succurro E, Marini MA, Arturi F et al (2009) Elevated one-hour post-load plasma glucose levels identifies subjects with normal glucose tolerance but early carotid atherosclerosis. Atherosclerosis 207:245–249
Sciacqua A, Maio R, Miceli S et al (2012) Association between one-hour post-load plasma glucose levels and vascular stiffness in essential hypertension. PLoS ONE 7:e44470
Sciacqua A, Miceli S, Carullo G et al (2011) One-hour postload plasma glucose levels and left ventricular mass in hypertensive patients. Diabetes Care 34:1406–1411
Perticone M, Tassone EJ, Scarpino PE et al (2016) Sympathovagal balance and 1-h postload plasma glucose in normoglucose tolerant hypertensive patients. Acta Diabetol 53:41–47
de Simone G, Devereux RB, Chien S, Alderman MH, Atlas SA, Laragh JH (1990) Relation of blood viscosity to demographic and physiologic variables and to cardiovascular risk factors in apparently normal adults. Circulation 81:107–117
Matsuda M, De Fronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22:1462–1470
Manco M, Panunzi S, Macfarlane DP et al (2010) One-hour plasma glucose identifies insulin resistance and beta-cell dysfunction in individuals with normal glucose tolerance: cross-sectional data from the relationship between insulin sensitivity and cardiovascular risk (RISC) study. Diabetes Care 33:2090–2097
Sesti G, Fiorentino TV, Succurro E et al (2014) Elevated 1-hour post-load plasma glucose levels in subjects with normal glucose tolerance are associated with unfavorable inflammatory profile. Acta Diabetol 51:927–932
Duncan BB, Schmidt MI, Pankow JS et al (2003) Low-grade systemic inflammation and the development of type 2 diabetes. The arteriosclerosis risk in communities study. Diabetes 52:1799–1805
Cavalot F, Petrelli A, Traversa M et al (2006) Postprandial blood glucose is a stronger predictor of cardiovascular events than fasting blood glucose in type 2 diabetes mellitus, particularly in women: lessons from the San Luigi Gonzaga diabetes study. J Clin Endocrinol Metab 91:813–819
Bergman M, Chetrit A, Roth J, Dankner R (2016) One-hour post-load plasma glucose level during the OGTT predicts mortality: observations from the Israel study of glucose intolerance, obesity and hypertension. Diabet Med 33:1060–1066
Acknowledgments
This work was supported, in part, by the Foundation for Diabetes Research of the Italian Diabetes Society, through the award of “Fondazione Diabete Ricerca FO.DI.RI.—MSD” scholarships in 2014 and 2015 to Gaia Chiara Mannino.
Author information
Authors and Affiliations
Contributions
M.A.M, T.V.F: researched data, reviewed and edited the manuscript; F.A, A.S: contributed to research data; G.C.M, M.P: reviewed the manuscript; F.P: contributed to the discussion and reviewed the manuscript; G.S. designed the study, analysed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All procedures performed in this study were in accordance with the ethical standards and all participants gave their informed consent in writing prior to inclusion in the study.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (Comitato Etico Azienda Ospedaliera “Mater Domini”, Italy) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Managed by Antonio Secchi.
Rights and permissions
About this article
Cite this article
Marini, M.A., Fiorentino, T.V., Andreozzi, F. et al. Elevated 1-h post-challenge plasma glucose levels in subjects with normal glucose tolerance or impaired glucose tolerance are associated with whole blood viscosity. Acta Diabetol 54, 775–784 (2017). https://doi.org/10.1007/s00592-017-1004-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-017-1004-z