Abstract
Background
The purpose of this study was to elucidate the effects of a dual-coated probiotic supplement (Duolac Care) on symptoms of diarrhea-predominant irritable bowel syndrome in a randomized double-blind clinical trial.
Methods
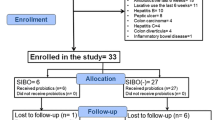
Fifty subjects with diarrhea-predominant irritable bowel syndrome were randomly assigned to either the non-coating group or the dual-coating group in order to receive two capsules per day of multi-species probiotics containing 5 billion bacteria per capsule for 4 weeks. Data from an adequate relief questionnaire were used in assessment of primary outcome. Daily records of stool frequencies and the Bristol stool scale, a weekly symptom diary using 100-mm visual analog scale, and Beck depression inventories were collected. Blood tests including blood cell counts, interleukin-10, tumor necrosis factor-alpha and inducible nitric oxide synthase, and regulatory T cells—CD4 + CD25high T cells, CD4 + LAP + T cells and CD25high + LAP + T cells—were analyzed before and after the study. The shift of gut microbiota was investigated using a quantitative real-time polymerase chain reaction assay.
Results
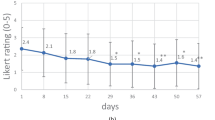
Responses to the adequate relief questionnaire indicated significant improvement in overall discomfort in the dual-coating group and the ratio of normal stools to hard or watery stools had a better effect from dual-coated probiotics compared to non-coated probiotics. This may be due to a shift of intestinal microbiota, as our correlation analysis showed significant negative correlation between Bifidobacterium and urgency of defecation.
Conclusions
Our result implies that dual-coating layers of probiotic supplement can be a candidate for treatment of diarrhea-predominant irritable bowel syndrome.





Similar content being viewed by others
References
Gralnek IM, Hays RD, Kilbourne A, et al. The impact of irritable bowel syndrome on health-related quality of life. Gastroenterology. 2000;119(3):654–60.
Fukudo S, Kaneko H, Akiho H, et al. Evidence-based clinical practice guidelines for irritable bowel syndrome. J Gastroenterol. 2015;50(1):11–30.
Öhman L, Simrén M. Pathogenesis of IBS: role of inflammation, immunity and neuroimmune interactions. Nat Rev Gastroenterol Hepatol. 2010;7(3):163–73.
Sayuk GS, Gyawali CP. Irritable bowel syndrome: modern concepts and management options. Am J Med. 2015;128(8):817–27.
King T, Elia M, Hunter J. Abnormal colonic fermentation in irritable bowel syndrome. The Lancet. 1998;352(9135):1187–9.
Schmulson M, Bielsa MV, Carmona-Sánchez R, et al. Microbiota, gastrointestinal infections, low-grade inflammation, and antibiotic therapy in irritable bowel syndrome (IBS): an evidence-based review. Rev de Gastroenterol de Méx. 2014;79(2):96–134 (English edition).
Lee KN, Lee OY. Intestinal microbiota in pathophysiology and management of irritable bowel syndrome. World J Gastroenterol: WJG. 2014;20(27):8886.
Kassinen A, Krogius-Kurikka L, Mäkivuokko H, et al. The fecal microbiota of irritable bowel syndrome patients differs significantly from that of healthy subjects. Gastroenterology. 2007;133(1):24–33.
Malinen E, Rinttilä T, Kajander K, et al. Analysis of the fecal microbiota of irritable bowel syndrome patients and healthy controls with real-time PCR. Am J Gastroenterol. 2005;100(2):373–82.
Moayyedi P, Ford AC, Talley NJ, et al. The efficacy of probiotics in the treatment of irritable bowel syndrome: a systematic review. Gut. 2010;59(3):325–32.
Minamida K, Nishimura M, Miwa K, et al. Effects of dietary fiber with Bacillus coagulans lilac-01 on bowel movement and fecal properties of healthy volunteers with a tendency for constipation. Biosci Biotechnol Biochem. 2015;79(2):300–6.
Kajander K, Myllyluoma E, Rajilić-Stojanović M, et al. Clinical trial: multispecies probiotic supplementation alleviates the symptoms of irritable bowel syndrome and stabilizes intestinal microbiota. Aliment Pharmacol Ther. 2008;27(1):48–57.
Sartor RB. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: antibiotics, probiotics, and prebiotics. Gastroenterology. 2004;126(6):1620–33.
Madsen K, Cornish A, Soper P, et al. Probiotic bacteria enhance murine and human intestinal epithelial barrier function. Gastroenterology. 2001;121(3):580–91.
Burgain J, Gaiani C, Linder M, et al. Encapsulation of probiotic living cells: from laboratory scale to industrial applications. J Food Eng. 2011;104(4):467–83.
Cha MK, Chung MJ, Kim JE, et al. Comparison of dual coated (Duolac™) and uncoated lactic acid bacteria from potential probiotics. Biotechnol Biotechnol Equip. 2011;25(3):2489–93.
Kang JY, Lee DK, Park JE, et al. Dual coating improves the survival of proviotic bifidobacterium strains during exposure to simulated gastro-intestinal conditions. Korean J Microbiol. 2013;49(3):275–81.
Yeun Y, Lee J. Effect of a double-coated probiotic formulation on functional constipation in the elderly: a randomized, double blind, controlled study. Arch Pharm Res. 2014;38(7):1–6.
Drossman DA, Li Z, Toner BB, et al. Functional bowel disorders. Dig Dis Sci. 1995;40(5):986–95.
Camilleri M, Northcutt AR, Kong S, et al. Efficacy and safety of alosetron in women with irritable bowel syndrome: a randomised, placebo-controlled trial. Lancet. 2000;355(9209):1035–40.
Heaton K, Radvan J, Cripps H, et al. Defecation frequency and timing, and stool form in the general population: a prospective study. Gut. 1992;33(6):818–24.
Kim HJ, Camilleri M, McKinzie S, et al. A randomized controlled trial of a probiotic, VSL# 3, on gut transit and symptoms in diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2003;17(7):895–904.
Beck AT, Steer RA, Carbin MG. Psychometric properties of the beck depression inventory: twenty-five years of evaluation. Clin Psychol Rev. 1988;8(1):77–100.
Guilarte M, Santos J, De Torres I, et al. Diarrhoea-predominant IBS patients show mast cell activation and hyperplasia in the jejunum. Gut. 2007;56(2):203–9.
Patacchioli F, Angelucci L, Dell’Erba G, et al. Actual stress, psychopathology and salivary cortisol levels in the irritable bowel syndrome (IBS). J Endocrinol Invest. 2001;24(3):173–7.
Cole JA, Rothman KJ, Cabral HJ, et al. Migraine, fibromyalgia, and depression among people with IBS: a prevalence study. BMC Gastroenterol. 2006;6(1):26.
Kendall PC, Hollon SD, Beck AT, et al. Issues and recommendations regarding use of the beck depression inventory. Cogn Ther Res. 1987;11(3):289–99.
Ringel Y, Ringel-Kulka T, Maier D, et al. Clinical trial: probiotic Bacteria Lactobacillus acidophilus NCFM and Bifidobacterium lactis Bi-07 Versus Placebo for the symptoms of bloating in patients with functional bowel disorders—a double-blind study. J Clin Gastroenterol. 2011;45(6):518.
Cappello C, Tremolaterra F, Pascariello A, et al. A randomised clinical trial (RCT) of a symbiotic mixture in patients with irritable bowel syndrome (IBS): effects on symptoms, colonic transit and quality of life. Int J Colorectal Dis. 2013;28(3):349–58.
Wong RK, Yang C, Song GH, et al. Melatonin regulation as a possible mechanism for probiotic (VSL# 3) in irritable bowel syndrome: a randomized double-blinded placebo study. Dig Dis Sci. 2015;60(1):186–94.
Kim S, Oh WY, Seung H, et al. Guideline for the clinical evaluation of drugs for the treatment of irritable bowel syndrome. Regul Res Food, Drug Cosmet. 2015;10(1):55–62.
FDA. Guidance for industry irritable bowel syndrome—clinical evaluation of drugs for treatment. US Department of Health and Human Sevices Food and Drug Administration Center for Drug Evaluation and Research. 2012.
Cryan JF, O’Mahony S. The microbiome-gut-brain axis: from bowel to behavior. Neurogastroenterol Motil. 2011;23(3):187–92.
Messaoudi M, Lalonde R, Violle N, et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr. 2011;105(05):755–64.
Desbonnet L, Garrett L, Clarke G, et al. The probiotic Bifidobacteria infantis: an assessment of potential antidepressant properties in the rat. J Psychiatr Res. 2008;43(2):164–74.
O’Mahony L, McCarthy J, Kelly P, et al. Lactobacillus and bifidobacterium in irritable bowel syndrome: symptom responses and relationship to cytokine profiles. Gastroenterology. 2005;128(3):541–51.
Rho Jh, Wright DP, Christie DL, et al. A novel mechanism for desulfation of mucin: identification and cloning of a mucin-desulfating glycosidase (sulfoglycosidase) from Prevotella strain RS2. J Bacteriol. 2005;187(5):1543–51.
Bergstrom KS, Xia L. Mucin-type O-glycans and their roles in intestinal homeostasis. Glycobiology. 2013:cwt045.
Ford A, Moayyedi P. Meta-analysis: factors affecting placebo response rate in the irritable bowel syndrome. Aliment Pharmacol Ther. 2010;32(2):144–58.
Triantafyllou K. Problems and challenges in the design of irritable bowel syndrome clinical trials. Focusing on the future with the experience from the past. Ann Gastroenterol. 2007;15(3):263–70.
Shah E, Pimentel M. Placebo effect in clinical trial design for irritable bowel syndrome. J neurogastroenterol Motil. 2014;20(2):163.
Spiller RC. Problems and challenges in the design of irritable bowel syndrome clinical trials: experience from published trials. Am J Med. 1999;107(5):91–7.
Whorwell PJ, Altringer L, Morel J, et al. Efficacy of an encapsulated probiotic Bifidobacterium infantis 35624 in women with irritable bowel syndrome. Am J Gastroenterol. 2006;101(7):1581–90.
Nikfar S, Rahimi R, Rahimi F, et al. Efficacy of probiotics in irritable bowel syndrome: a meta-analysis of randomized, controlled trials. Dis Colon Rectum. 2008;51(12):1775–80.
Acknowledgments
This study was supported financially by Cell Biotech.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H Kim received funds from CellBiotech. JG Seo is an employee of CellBiotech, which does not affect the result or interpretation of the study. Other authors do not have any conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Han, K., Wang, J., Seo, JG. et al. Efficacy of double-coated probiotics for irritable bowel syndrome: a randomized double-blind controlled trial. J Gastroenterol 52, 432–443 (2017). https://doi.org/10.1007/s00535-016-1224-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-016-1224-y