Abstract
Purpose
Clostridium difficile infection (CDI) is the leading cause of diarrhoea in hospitalised patients. Cancer populations are at high-risk for infection, but comprehensive evaluation in the current era of cancer care has not been performed. The objective of this study was to describe characteristics, risk factors, and outcomes of CDI in cancer patients.
Methods
Fifty consecutive patients with CDI at a large Australian cancer centre (2013–2015) were identified from the hospital pathology database. Each case was matched by ward and hospital admission date to three controls without toxigenic CDI. Treatment and outcomes of infection were evaluated and potential risk factors were analysed using conditional logistic regression.
Results
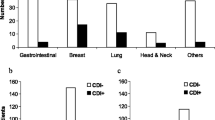
Patients with CDI had a mean age of 59.7 years and 74% had an underlying solid tumour. Healthcare-associated infection comprised 80% of cases. Recurrence occurred in 10, and 12% of cases were admitted to ICU within 30 days. Severe or severe-complicated infection was observed in 32%. Independent risk factors for infection included chemotherapy (odds ratio (OR) 3.82, 95% CI 1.67–8.75; p = 0.002), gastro-intestinal/abdominal surgery (OR 4.64, 95% CI 1.20–17.91; p = 0.03), proton pump inhibitor (PPI) therapy (OR 2.47, 95% CI 1.05–5.80; p = 0.04), and days of antibiotic therapy (OR 1.04, 95% CI 1.01–1.08; p = 0.02).
Conclusions
Severe or complicated infections are frequent in patients with cancer who develop CDI. Receipt of chemotherapy, gastro-intestinal/abdominal surgery, PPI therapy, and antibiotic exposure contribute to infection risk. More effective CDI therapy for cancer patients is required and dedicated antibiotic stewardship programs in high-risk cancer populations are needed to ameliorate infection risk.
Similar content being viewed by others
References
Kamboj M, Son C, Cantu S, Chemaly RF, Dickman J, Dubberke E, Engles L, Lafferty T, Liddell G, Lesperance ME, Mangino JE, Martin S, Mayfield J, Mehta SA, O’Rourke S, Perego CS, Taplitz R, Eagan J, Sepkowitz KA (2012) Hospital-onset Clostridium difficile infection rates in persons with cancer or hematopoietic stem cell transplant: a C3IC network report. Infect Control Hosp Epidemiol 33(11):1162–1165
Freeman J, Bauer MP, Baines SD, Corver J, Fawley WN, Goorhuis B, Kuijper EJ, Wilcox MH (2010) The changing epidemiology of Clostridium difficile infections. Clin Microbiol Rev 23(3):529–549
Dubberke ER, Reske KA, Srivastava A, Sadhu J, Gatti R, Young RM, Rakes LC, Dieckgraefe B, DiPersio J, Fraser VJ (2010) Clostridium difficile-associated disease in allogeneic hematopoietic stem-cell transplant recipients: risk associations, protective associations, and outcomes. Clin Transpl 24(2):192–198
Chopra T, Chandrasekar P, Salimnia H, Heilbrun LK, Smith D, Alangaden GJ (2011) Recent epidemiology of Clostridium difficile infection during hematopoietic stem cell transplantation. Clin Transpl 25(1):E82–E87
Alonso CD, Treadway SB, Hanna DB, Huff CA, Neofytos D, Carroll KC, Marr KA (2012) Epidemiology and outcomes of Clostridium difficile infections in hematopoietic stem cell transplant recipients. Clin Infect Dis 54(8):1053–1063
Willems L, Porcher R, Lafaurie M, Casin I, Robin M, Xhaard A, Andreoli AL, Rodriguez-Otero P, Dhedin N, Socié G, Ribaud P, Peffault de Latour R (2012) Clostridium difficile infection after allogeneic hematopoietic stem cell transplantation: incidence, risk factors, and outcome. Biol Blood Marrow Transplant 18(8):1295–1301
Krishna SG, Zhao W, Apewokin SK, Krishna K, Chepyala P, Anaissie EJ (2013) Risk factors, preemptive therapy, and antiperistaltic agents for Clostridium difficile infection in cancer patients. Transpl Infect Dis 15(5):493–501
Bruminhent J, Wang Z-X, Hu C, Wagner J, Sunday R, Bobik B, Hegarty S, Keith S, Alpdogan S, Carabasi M, Filicko-O’Hara J, Flomenberg N, Kasner M, Outschoorn UM, Weiss M, Flomenberg P (2014) Clostridium difficile colonization and disease in patients undergoing hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 20(9):1329–1334
Jain T, Croswell C, Urday-Cornejo V, Awali R, Cutright J, Salimnia H, Reddy Banavasi HV, Liubakka A, Lephart P, Chopra T, Revankar SG, Chandrasekar P, Alangaden G (2016) Clostridium difficile colonization in hematopoietic stem cell transplant recipients: a prospective study of the epidemiology and outcomes involving toxigenic and nontoxigenic strains. Biol Blood Marrow Transplant 22:157–163
Liu NW, Shatagopam K, Monn MF, Kaimakliotis HZ, Cary C, Boris RS, Mellon MJ, Masterson TA, Foster RS, Gardner TA, Bihrle R, House MG, Koch MO (2015) Risk for Clostridium difficile infection after radical cystectomy for bladder cancer: analysis of a contemporary series. Urol Oncol 33:503.e517–503.e522
Blot E, Escande MC, Besson D, Barbut F, Granpeix C, Asselain B, Falcou MC, Pouillart P (2003) Outbreak of Clostridium difficile-related diarrhoea in an adult oncology unit: risk factors and microbiological characteristics. J Hosp Infect 53(3):187–192
Han XH, Du CX, Zhang CL, Zheng CL, Wang L, Li D, Feng Y, DuPont HL, Jiang ZD, Shi YK (2013) Clostridium difficile infection in hospitalized cancer patients in Beijing, China is facilitated by receipt of cancer chemotherapy. Anaerobe 24:82–84
Kim J, Ward K, Shah N, Saenz C, McHale M, Plaxe S (2013) Excess risk of Clostridium difficile infection in ovarian cancer is related to exposure to broad-spectrum antibiotics. Support Care Cancer 21(11):3103–3107
Rodríguez Garzotto A, Mérida García A, Muñoz Unceta N, Galera Lopez MM, Orellana-Miguel MÁ, Díaz-García CV, Cortijo-Cascajares S, Cortes-Funes H, Agulló-Ortuño MT (2015) Risk factors associated with Clostridium difficile infection in adult oncology patients. Support Care Cancer 23(6):1569–1577
Slimings C, Armstrong P, Beckingham WD, Bull AL, Hall L, Kennedy KJ, Marquess J, McCann R, Menzies A, Mitchell BG, Richards MJ, Smollen PC, Tracey L, Wilkinson IJ, Wilson FL, Worth LJ, Riley TV (2014) Increasing incidence of Clostridium difficile infection, Australia, 2011–2012. Med J Aust 200(5):272–276
Cheng AC, Collins DA, Elliott B, Ferguson JK, Paterson DL, Thean S, Riley TV (2016) Laboratory-based surveillance of Clostridium difficile circulating in Australia, September - November 2010. Pathology 48(3):257–260
Worth LJ, Spelman T, Bull AL, Brett JA, Richards MJ (2016) Epidemiology of Clostridium difficile infections in Australia: enhanced surveillance to evaluate time trends and severity of illness in Victoria, 2010-2014. J Hosp Infect 93(3):280–285
Wacholder S, Silverman DT, McLaughlin JK, Mandel JS (1992) Selection of controls in case-control studies. III Design options Am J Epidemiol 135(9):1042–1050
Dubberke ER, Carling P, Carrico R, Donskey CJ, Loo VG, McDonald LC, Maragakis LL, Sandora TJ, Weber DJ, Yokoe DS, Gerding DN (2014) Strategies to prevent Clostridium difficile infections in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol 35:628–645
Bull AL, Worth LJ, Richards MJ (2012) Implementation of standardised surveillance for Clostridium difficile infections in Australia: initial report from the Victorian healthcare associated infection surveillance system. Intern Med J 42(6):715–718
McDonald LC, Coignard B, Dubberke E, Song X, Horan T, Kutty PK (2007) Recommendations for surveillance of Clostridium difficile–associated disease. Infect Control Hosp Epidemiol 28(02):140–145
Cohen S, Gerding D, Stuart J, Kelly C, Loo V, McDonald L, Pepin J, Wilcox M (2010) Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Infect Control Hosp Epidemiol 31(5):431–455
World Health Organization (1979) Handbook for reporting results of cancer treatment. In: Geneva. World Health Organization, Switzerland, pp 15–22
Hebbard AIT, Slavin MA, Reed C, Teh BW, Thursky KA, Trubiano JA, Worth LJ (2016) The epidemiology of Clostridium difficile infection in patients with cancer. Exp Rev Anti Infect Ther:DOI. doi:10.1080/14787210.14782016.11234376
Cornely OA, Miller MA, Fantin B, Mullane K, Kean Y, Gorbach S (2013) Resolution of Clostridium difficile-associated diarrhea in patients with cancer treated with fidaxomicin or vancomycin. J Clin Oncol 31(19):2493–2499
Trubiano JA, George A, Barnett J, Siwan M, Heriot A, Prince HM, Slavin MA, Teh BW (2015) A different kind of “allogeneic transplant”: successful fecal microbiota transplant for recurrent and refractory Clostridium difficile infection in a patient with relapsed aggressive B-cell lymphoma. Leuk Lymphoma 56(2):512–514
Yoon YK, Kim MJ, Sohn JW, Kim HS, Choi YJ, Kim JS, Kim ST, Park KH, Kim SJ, Kim BS, Shin SW, Kim YH, Park Y (2014) Predictors of mortality attributable to Clostridium difficile infection in patients with underlying malignancy. Support Care Cancer 22(8):2039–2048
Stevens V, Dumyati G, Fine LS, Fisher SG, van Wijngaarden E (2011) Cumulative antibiotic exposures over time and the risk of Clostridium difficile infection. Clin Infect Dis 53(1):42–48
Barlam TF, Cosgrove SE, Abbo LM, MacDougall C, Schuetz AN, Septimus EJ, Srinivasan A, Dellit TH, Falck-Ytter YT, Fishman NO, Hamilton CW, Jenkins TC, Lipsett PA, Malani PN, May LS, Moran GJ, Neuhauser MM, Newland JG, Ohl CA, Samore MH, Seo SK, Trivedi KK (2016) Implementing an antibiotic stewardship program: guidelines by the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Clin Infect Dis 62(10):e51–e77
Kamthan AG, Bruckner HW, Hirschman SZ, Agus SG (1992) Clostridium difficile diarrhea induced by cancer chemotherapy. Arch Intern Med 152(8):1715–1717
Nielsen H, Daugaard G, Tvede M, Bruun B (1992) High prevalence of Clostridium difficile diarrhoea during intensive chemotherapy for disseminated germ cell cancer. Brit J Cancer 66(4):666–667
Emoto M, Kawarabayashi T, Hachisuga T, Eguchi F, Shirakawa K (1996) Clostridium difficile colitis associated with cisplatin-based chemotherapy in ovarian cancer patients. Gynecol Oncol 61(3):369–372
Husain A, Aptaker L, Spriggs DR, Barakat RR (1998) Gastrointestinal toxicity and Clostridium difficile diarrhea in patients treated with paclitaxel-containing chemotherapy regimens. Gynecol Oncol 71(1):104–107
Kwok CS, Arthur AK, Anibueze CI, Singh S, Cavallazzi R, Loke YK (2012) Risk of Clostridium difficile infection with acid suppressing drugs and antibiotics: meta-analysis. Am J Gastroenterol 107(7):1011–1019
Janarthanan S, Ditah I, Adler DG, Ehrinpreis MN (2012) Clostridium difficile-associated diarrhea and proton pump inhibitor therapy: a meta-analysis. Am J Gastroenterol 107(7):1001–1010
McCaleb RV, Gandhi AS, Clark SM, Clemmons AB (2016) Clinical outcomes of acid suppressive therapy use in hematology/oncology patients at an academic medical center. Ann Pharmacother 50(7):541–547
Aquina CT, Probst CP, Becerra AZ, Hensley BJ, Iannuzzi JC, Noyes K, Monson JR, Fleming FJ (2016) High variability in nosocomial Clostridium difficile infection rates across hospitals after colorectal resection. Dis Colon rectum 59(4):323–331
Yasunaga H, Horiguchi H, Hashimoto H, Matsuda S, Fushimi K (2012) The burden of Clostridium difficile-associated disease following digestive tract surgery in Japan. J Hosp Infect 82(3):175–180
Abdelsattar ZM, Krapohl G, Alrahmani L, Banerjee M, Krell RW, Wong SL, Campbell DA, Aronoff DM, Hendren S (2015) Postoperative burden of hospital-acquired Clostridium difficile infection. Infect Control Hosp Epidemiol 36(1):40–46
Acknowledgements
Authors acknowledge the assistance of Jennifer Breen, Kee Ming, Badal Bhatt and Malcolm Eaton for extraction of surveillance and laboratory data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Funding
No external funding was used for this study.
Rights and permissions
About this article
Cite this article
Hebbard, A.I., Slavin, M.A., Reed, C. et al. Risks factors and outcomes of Clostridium difficile infection in patients with cancer: a matched case-control study. Support Care Cancer 25, 1923–1930 (2017). https://doi.org/10.1007/s00520-017-3606-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-017-3606-y