Abstract
Purpose
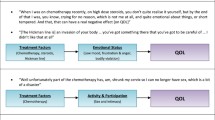
Little qualitative research exploring the impact of multiple myeloma (MM) and its treatment on the health-related quality of life (HRQL) of patients has been published. This study aimed to explore the burden of MM symptoms and treatment and the impact of these on HRQL. A model was developed to illustrate key concepts and their interrelationships.
Methods
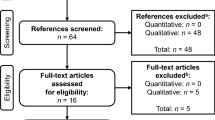
Patients with MM were recruited to this cross-sectional, qualitative study through a patient panel and at two clinical sites in the USA. An interview discussion guide was developed using a review of published literature and interviews with experienced MM clinicians. In-depth, semistructured telephone interviews with MM patients were conducted to explore their experiences of the disease and its treatment. Data were analyzed using a thematic analysis approach.
Results
Twenty MM patients at various stages of treatment participated in open-ended, semistructured interviews. Patients reported both current and previous MM symptoms; most had experienced fatigue and pain. Other commonly reported symptoms were fractures, anemia, neuropathy, aches, and infections. MM treatment was found to have a negative impact on patients’ HRQL; treatment-related adverse events included fatigue, neuropathy, insomnia, and gastrointestinal symptoms. MM treatment placed a substantial psychological and physical burden on patients, disrupting social activities, decreasing independence, and impacting on relationships. A model was developed to illustrate the relationship between these concepts.
Conclusion
The conceptual model developed in this study illustrates the many aspects of MM and its treatment and how they can have a negative impact on patients’ HRQL.

Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics, 2014. CA. Cancer J Clin 64:9–29
Richardson PG, Laubach J, Mitsiades CS, Schlossman R, Hideshima T, Redman K, Chauhan D, Ghobrial IM, Munshi N, Anderson KC (2011) Managing multiple myeloma: the emerging role of novel therapies and adapting combination treatment for higher risk settings. Br J Haematol 154:755–762
Cachia E, Eiser C, Ezaydi Y (2012) A study of pain, peripheral neuropathy and psychosocial late effects in multiple myeloma patients. BMJ Support Palliat Care 2:A3
Roodman GD (1997) Mechanisms of bone lesions in multiple myeloma and lymphoma. Cancer 80:1557–1563
Rosen LS, Gordon D, Kaminski M, Howell A, Belch A, Mackey J, Apffelstaedt J, Hussein MA, Coleman RE, Reitsma DJ, Chen BL, Seaman JJ (2013) Long-term efficacy and safety of zoledronic acid compared with pamidronate disodium in the treatment of skeletal complications in patients with advanced multiple myeloma or breast carcinoma: a randomized, double-blind, multicenter, comparative trial. Cancer 98:1735–1744
Mundy GR (1998) Myeloma bone disease. Eur J Cancer 34:246–251
Booker R, Olson K, Pilarski LM, Noon JP, Bahlis NJ (2009) The relationships among physiologic variables, quality of life, and fatigue in patients with multiple myeloma. Oncol Nurs Forum 36:209–216
Dodd MJ, Miaskowski C, Paul SM (2001) Symptom clusters and their effect on the functional status of patients with cancer. Oncol Nurs Forum 28:465–470
Bilezikian JP (1992) Management of acute hypercalcemia. N Engl J Med 326:1196–1203
Smith D, Yong K (2013) Multiple myeloma. BMJ 346:f3863
Laubach JP, Mitsiades CS, Mahindra A, Schlossman RL, Hideshima T, Chauhan D, Carreau NA, Ghobrial IM, Raje N, Munshi NC, Anderson KC, Richardson PG (2009) Novel therapies in the treatment of multiple myeloma. J Natl Compr Cancer Netw 7:947–960
Richardson PG, Schlossman RL, Weller E, Hideshima T, Mitsiades C, Davies F, LeBlanc R, Catley LP, Doss D, Kelly K, McKenney M, Mechlowicz J, Freeman A, Deocampo R, Rich R, Ryoo JJ, Chauhan D, Balinski K, Zeldis J, Anderson KC (2002) Immunomodulatory drug CC-5013 overcomes drug resistance and is well tolerated in patients with relapsed multiple myeloma. Blood 100:3063–3067
Osborne TR, Ramsenthaler C, Siegert RJ, Edmonds PM, Schey SA, Higginson IJ (2012) What issues matter most to people with multiple myeloma and how well are we measuring them? A systematic review of quality of life tools. Eur J Haematol 89:437–457
Mols F, Oerlemans S, Vos AH, Koster A, Verelst S, Sonneveld P, van de Poll-Franse LV (2012) Health-related quality of life and disease-specific complaints among multiple myeloma patients up to 10 yr after diagnosis: results from a population-based study using the PROFILES registry. Eur J Haematol 89:311–319
Sherman AC, Simonton S, Latif U, Plante TG, Anaissie EJ (2009) Changes in quality-of-life and psychosocial adjustment among multiple myeloma patients treated with high-dose melphalan and autologous stem cell transplantation. Biol Blood Marrow Transplant 15:12–20
Rothman ML, Beltran P, Cappelleri JC, Lipscomb J, Teschendorf B (2007) Patient-reported outcomes: conceptual issues. Value Health 10:S66–S75
Braun V, Clarke V (2006) Using thematic analysis in psychology. Qual Res Psychol 3:77–101
ATLAS.ti Qualitative Data Analysis Software (Version 5) (Software) (2014) Retrieved from http://atlasti.com/
Morse JM (1995) The significance of saturation. Qual Health Res 5:147–149
Maher K, deVries K (2011) An exploration of the lived experiences of individuals with relapsed multiple myeloma. Eur J Cancer Care (Engl) 20:267–275
Potrata B, Cavet J, Blair S, Howe T, Molassiotis A (2011) Understanding distress and distressing experiences in patients living with multiple myeloma: an exploratory study. Psychooncology 20:127–134
Molassiotis A, Wilson B, Blair S, Howe T, Cavet J (2011) Unmet supportive care needs, psychological well-being and quality of life in patients living with multiple myeloma and their partners. Psychooncology 20:88–97
Molassiotis A, Wilson B, Blair S, Howe T, Cavet J (2011) Living with multiple myeloma: experiences of patients and their informal caregivers. Support. Care Cancer 19:101–111
U.S.Cancer StatisticsWorking Group. United States Cancer Statistics: 1999–2010 Incidence and Mortality Web-based Report. Atlanta: U.S. (2013) Department of Health and Human Services, Centers for Disease Control and Prevention and National Cancer Institute. Available at http://www.cdc.gov/uscs
Sturges JE, Hanrahan KJ (2004) Comparing telephone and face-to-face qualitative interviewing: a research note. Qual Res 4(1):107–118
Irvine A (2011) Duration, dominance and depth in telephone and face-to-face interviews: a comparative exploration. Int J Qualitative Methods 10(3):202–220
Acknowledgments
The authors would like to acknowledge the editorial assistance of Jane Saunders and Helen Johns of FireKite, part of the KnowledgePoint360 Group, an Ashfield Company, during the development of this publication, which was funded by Millennium Pharmaceuticals, Inc. The authors would also like to thank Katie Breheny, Kendra Debusk, and Rene Dickerhoof from Icon Plc for their assistance with data collection and analysis.
Authorship contributions
KG, PS, HML, and DB designed and/or performed the research reported in this analysis; KG collected data; KG, PS, and DB analyzed the data; all authors interpreted the data; KG and PS wrote the manuscript. All authors reviewed and approved the manuscript.
Conflict of interest
RB: Research support from Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited, Celgene, Sanofi, and BMS
RB: Has full control of all primary data and agrees to allow the journal to review these data if requested
HML: Employment: Millennium Pharmaceuticals Inc.
A-MH: Employment: Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited
RDH: Research funding from Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited, Celgene, Novartis, and Onyx
KC: Has consulted for Millennium Pharmaceuticals Inc. and Celgene
KG: Employment: Icon plc, which received research funding from Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited
PS: Employment: MAPI consultancy, which received research funding from Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited
JL: Research funding from Novartis, Onyx, Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited, and Celgene
DB: Employment: Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited
PR: Advisory committee member for Celgene, Janssen Pharmaceuticals, and Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited, and has received research funding from Celgene and Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited
Author information
Authors and Affiliations
Corresponding author
Additional information
Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited, Cambridge, MA, USA.
Rights and permissions
About this article
Cite this article
Baz, R., Lin, H.M., Hui, AM. et al. Development of a conceptual model to illustrate the impact of multiple myeloma and its treatment on health-related quality of life. Support Care Cancer 23, 2789–2797 (2015). https://doi.org/10.1007/s00520-015-2644-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-015-2644-6