Abstract
Key message
AGP-rich glycoproteins mediate pollen–ovule interactions and cell patterning in the embryo sac of apple before and after fertilization.
Abstract
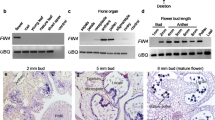
Glycoproteins are significant players in the dialog that takes place between growing pollen tubes and the stigma and style in the angiosperms. Yet, information is scarce on their possible involvement in the ovule, a sporophytic organ that hosts the female gametophyte. Apple flowers have a prolonged lapse of time between pollination and fertilization, offering a great system to study the developmental basis of glycoprotein secretion and their putative role during the last stages of the progamic phase and early seed initiation. For this purpose, the sequential pollen tube elongation within the ovary was examined in relation to changes in arabinogalactan proteins (AGPs) in the tissues of the ovule before and after fertilization. To evaluate what of these changes are developmentally regulated, unpollinated and pollinated flowers were compared. AGPs paved the pollen tube pathway in the ovules along the micropylar canal, and the nucellus entrance toward the synergids, which also developmentally accumulated AGPs at the filiform apparatus. Glycoproteins vanished from all these tissues following pollen tube passage, strongly suggesting a role in pollen–ovule interaction. In addition, AGPs marked the primary cell walls of the haploid cells of the female gametophyte, and they further built up in the cell walls of the embryo sac and developing embryo, layering the interactive walls of the three generations hosted in the ovule, the maternal sporophytic tissues, the female gametophyte, and the developing embryo.









Similar content being viewed by others
References
Arbeloa A, Herrero M (1987) The significance of the obturator in the control of pollen tube entry into the ovary in peach (Prunus persica). Ann Bot 60:681–685. https://doi.org/10.1093/oxfordjournals.aob.a087500
Bouman F (1984) The ovule. In: Johri BM (ed) Embryology of angiosperms. Springer, Berlin, pp 123–157
Castro AJ, Suárez C, Zienkiewicz K, de Dios AlchéJ, Zienkiewicz A, Rodríguez-García MI (2013) Electrophoretic profiling and immunocytochemical detection of pectins and arabinogalactan proteins in olive pollen during germination and pollen tube growth. Ann Bot 112:503–513. https://doi.org/10.1093/aob/mct118
Coimbra S, Almeida J, Junqueira V, Costa ML, Pereira LG (2007) Arabinogalactan proteins as molecular markers in Arabidopsis thaliana sexual reproduction. J Exp Bot 58:4027–4035. https://doi.org/10.1093/jxb/erm259
Coimbra S, Duarte C (2003) Arabinogalactan proteins may facilitate the movement of pollen tubes from the stigma to the ovules in Actinidia deliciosa and Amaranthus hypochondriacus. Euphytica 133:171–178. https://doi.org/10.1023/A:102556492
Coimbra S, Salema R (1997) Immunolocalization of arabinogalactan proteins in Amaranthus hypochondriacus L. ovules. Protoplasma 199:75–82. https://doi.org/10.1007/BF02539808
Costa Tura J, Mackenzie K (1990) Ovule and embryo sac development in Malus pumila L. cv. Cox’s Orange Pippin, from dormancy to blossom. Ann Bot 66:443–450. https://doi.org/10.1093/oxfordjournals.aob.a088046
Costa ML, Lopes AL, Amorim MI, Coimbra S (2017) Immunolocalization of AGPs and pectins in Quercus suber gametophytic structures. In: Schmidt A (ed) Plant Germline development. Methods in molecular biology, vol 1669. Humana Press, New York, pp, 117–137. https://doi.org/10.1007/978-1-4939-7286-9_11
Dardelle F, Lehner A, Ramdani Y, Bardor M, Lerouge P, Driouich A, Mollet J (2010) Biochemical and immunocytological characterizations of Arabidopsis pollen tube cell wall. Plant Physiol 153:1563–1576. https://doi.org/10.1104/pp.110.158881
Derksen J, Janssen GJ, Wolters-Arts M, Lichtscheidl I, Adlassnig W, Ovecka M, Doris F, Steer M (2011) Wall architecture with high porosity is established at the tip and maintained in growing pollen tubes of Nicotiana tabacum. Plant J 68:495–506. https://doi.org/10.1111/j.1365-313X.2011.04703.x
Dresselhaus T, Márton ML (2009) Micropylar pollen tube guidance and burst: adapted from defense mechanisms? Curr Opin Plant Biol 12:773–780. https://doi.org/10.1016/j.pbi.2009.09.015
Dresselhaus T, Sprunck S, Wessel GM (2016) Fertilization mechanisms in flowering plants. Curr Biol 26:R125–R139. https://doi.org/10.1016/j.cub.2015.12.032
Endress PK (2011) Angiosperm ovules: diversity, development, evolution. Ann Bot 107:1465–1489. https://doi.org/10.1093/aob/mcr120
Feder N, O’brien TP (1968) Plant microtechnique: some principles and new methods. Am J Bot 55:123–142. https://doi.org/10.1002/j.1537-2197.1968.tb06952.x
Gawecki R, Sala K, Kurczyńska EU, Świątek P, Płachno BJ (2017) Immunodetection of some pectic, arabinogalactan proteins and hemicellulose epitopes in the micropylar transmitting tissue of apomictic dandelions (Taraxacum, Asteraceae, Lactuceae). Protoplasma 254:657–668. https://doi.org/10.1007/s00709-016-0980-0
Gibbs PE (2014) Late-acting self-incompatibility—the pariah breeding system in flowering plants. New Phytol 203:717–734. https://doi.org/10.1111/nph.12874
Herrero M (2000) Changes in the ovary related to pollen tube guidance. Ann Bot 85:79–85. https://doi.org/10.1006/anbo.1999.1014
Herrero M (2001) Ovary signals for directional pollen tube growth. Sex Plant Reprod 14:3–7. https://doi.org/10.1007/s004970100082
Herrero M (2003) Male and female synchrony and the regulation of mating in flowering plants. Philos Trans R Soc B 358:1019–1024. https://doi.org/10.1098/rstb.2003.1285
Herrero M, Arbeloa A (1989) Influence of the pistil on pollen tube kinetics in peach (Prunus persica). Am J Bot 76:1441–1447. https://doi.org/10.2307/2444430
Herrero M, Dickinson HG (1979) Pollen–pistil incompatibility in Petunia hybrida: changes in the pistil following compatible and incompatible intraspecific crosses. J Cell Sci 36:1–18
Herrero M, Dickinson HG (1981) Pollen tube development in Petunia hybrida following compatible and incompatible intraspecific matings. J Cell Sci 47:365–383
Herrero M, Gascón M (1987) Prolongation of embryo sac viability in pear (Pyrus communis) following pollination or treatment with gibberellic acid. Ann Bot 60:287–293
Higashiyama T, Hamamura Y (2008) Gametophytic pollen tube guidance. Sex Plant Reprod 21:17–26. https://doi.org/10.1007/s00497-007-0064-6
Higashiyama T, Yabe S, Sasaki N, Nishimura Y, Miyagishima S, Kuroiwa H, Kuroiwa T (2001) Pollen tube attraction by the synergid cell. Science 293:1480–1483
Hofmeister W (1849) Die Entstehung des Embryos der Phanerogamen. Hofmeister, Leipzig
Hughes J, McCully ME (1975) The use of an optical brightener in the study of plant structure. Stain Technol 50:319. https://doi.org/10.3109/10520297509117082
Hülskamp M, Schneitz K, Pruitt RE (1995) Genetic evidence for a long-range activity that directs pollen tube guidance in Arabidopsis. Plant Cell 7:57–64. https://doi.org/10.1105/tpc.7.1.57
Jefferies CJ, Belcher AR (1974) A fluorescent brightener used for pollen tube identification in vivo. Stain Technol 49:199–202
Johansen DA (1940) Plant microtechnique. McGraw-Hill, New York
Johnson MA, Preuss D (2002) Plotting a course: multiple signals guide pollen tubes to their targets. Dev Cell 2:273–281. https://doi.org/10.1016/S1534-5807(02)00130-2
Jauh G, Lord E (1996) Localization of pectins and arabinogalactan-proteins in lily (Lilium longiflorum L.) pollen tube and style, and their possible roles in pollination. Planta 199:251–261. https://doi.org/10.1007/BF00196566
Kanaoka MM (2018) Cell–cell communications and molecular mechanisms in plant sexual reproduction. J Plant Res 131:37–47. https://doi.org/10.1007/s10265-017-0997-2
Kasahara RD, Portereiko MF, Sandaklie-Nikolova L, Rabiger DS, Drews GN (2005) MYB98 is required for pollen tube guidance and synergid cell differentiation in Arabidopsis. Plant Cell 17:2981–2992. https://doi.org/10.1105/tpc.105.034603
Kessler SA, Grossniklaus U (2011) She’s the boss: signalling in pollen tube reception. Curr Opin Plant Biol 14:622–627. https://doi.org/10.1016/j.pbi.2011.07.012
Knox JP, Linstead PJ, Cooper JP, Roberts K (1991) Developmentally regulated epitopes of cell surface arabinogalactan proteins and their relation to root tissue pattern formation. Plant J 1:317–326. https://doi.org/10.1046/j.1365-313X.1991.t01-9-00999.x
Leshem Y, Johnson C, Sundaresan V (2013) Pollen tube entry into the synergid cell of Arabidopsis is observed at a site distinct from the filiform apparatus. Plant Reprod 26:93–99. https://doi.org/10.1007/s00497-013-0211-1
Leszczuk A, Szczuka E (2018) Arabinogalactan proteins: immunolocalization in the developing ovary of a facultative apomict Fragaria × ananassa (Duch.). Plant Physiol Biochem 123:24–33. https://doi.org/10.1016/j.plaphy.2017.12.003
Leszczuk A, Kozioł A, Szczuka E, Zdunek A (2019a) Analysis of AGP contribution to the dynamic assembly and mechanical properties of cell wall during pollen tube growth. Plant Sci 281:9–18. https://doi.org/10.1016/j.plantsci.2019.01.005
Leszczuk A, Szczuka E, Zduneka A (2019b) Arabinogalactan proteins: distribution during the development of male and female gametophytes. Plant Physiol Biochem 135:9–18. https://doi.org/10.1016/j.plaphy.2018.11.023
Leszczuk A, Wydrych J, Szczuka E (2019c) The occurrence of calcium oxalate crystals and distribution of arabinogalactan proteins (AGPs) in ovary cells during Fragaria × ananassa (Duch.) development. J Plant Growth Regul. https://doi.org/10.1007/s00344-018-09912-7
Li HJ, Meng JG, Yang WC (2018) Multilayered signaling pathways for pollen tube growth and guidance. Plant Reprod 31:31–41. https://doi.org/10.1007/s00497-018-0324-7
Linskens HF (1975) Incompatibility in Petunia. Proc R Soc Lond B 188:299–311. https://doi.org/10.1098/rspb.1975.0021
Linskens HF, Esser KL (1957) Über eine spezifische anfärbung der pollenschläuche im griffel und die zahl der kallosepfropfen nach selbstung und fremdung. Naturwissenschaften 44:16. https://doi.org/10.1007/BF00629340
Liu J, Zhang H, Cheng Y, Kafkas S, Güney M (2014) Pistillate flower development and pollen tube growth mode during the delayed fertilization stage in Corylus heterophylla Fisch. Plant Reprod 27:145–152. https://doi.org/10.1007/s00497-014-0248-9
Lopes AL, Costa ML, Sobral R, Costa MM, Amorim MI, Coimbra S (2016) Arabinogalactan proteins and pectin distribution during female gametogenesis in Quercus suber L. Ann Bot 117:949–961. https://doi.org/10.1093/aob/mcw019
Lopez R, Renzaglia A (2016) Arabinogalactan proteins and arabinan pectins abound in the specialized matrices surrounding female gametes of the fern Ceratopteris richardii. Planta 243:947–957. https://doi.org/10.1007/s00425-015-2448-4
Lora J, Hormaza JI, Herrero M (2016) The diversity of the pollen tube pathway in plants: toward an increasing control by the sporophyte. Front Plant Sci 7:107. https://doi.org/10.3389/fpls.2016.00107
Lora J, Laux T, Hormaza JI (2018) The role of the integuments in pollen tube guidance in flowering plants. New Phytol. https://doi.org/10.1111/nph.15420
Losada JM, Herrero M (2012) Arabinogalactan-protein secretion is associated with the acquisition of stigmatic receptivity in the apple flower. Ann Bot 110:573–584. https://doi.org/10.1093/aob/mcs116
Losada JM, Herrero M (2014) Glycoprotein composition along the pistil of Malus × domestica and the modulation of pollen tube growth. BMC Plant Biol 14:1. https://doi.org/10.1186/1471-2229-14-1
Losada JM, Herrero M (2017) Pollen tube access to the ovule is mediated by glycoprotein secretion on the obturator of apple (Malus × domestica, Borkh). Ann Bot 119:989–1000. https://doi.org/10.1093/aob/mcw276
Losada JM, Hormaza JI, Lora J (2017) Pollen-pistil interaction in pawpaw (Asimina triloba L.), the northernmost species of the mainly tropical family Annonaceae. Am J Bot 104:1891–1903. https://doi.org/10.3732/ajb.1700319
Márton ML, Cordts S, Broadhvest J, Dresselhaus T (2005) Micropylar pollen tube guidance by egg apparatus 1 of maize. Science 307:573–576. https://doi.org/10.1126/science.1104954
Márton ML, Dresselhaus T (2010) Female gametophyte-controlled pollen tube guidance. Biochem Soc Trans 38:627–630. https://doi.org/10.1042/BST0380627
Márton ML, Fastner A, Uebler S, Dresselhaus T (2012) Overcoming hybridization barriers by the secretion of the maize pollen tube attractant ZmEA1 from Arabidopsis ovules. Curr Biol 22:1194–1198. https://doi.org/10.1016/j.cub.2012.04.061
Mendes SP, Mastroberti AA, Mariath JE, Vieira RC, De Toni KL (2014) Ovule and female gametophyte development in the Bromeliaceae: an embryological study of Pitcairnia encholirioides. Botany 92:883–894. https://doi.org/10.1139/cjb-2014-0114
Mizukami AG, Inatsugi R, Jiao J et al (2016) The AMOR arabinogalactan sugar chain induces pollen-tube competency to respond to ovular guidance. Curr Biol 26:1091–1097
Mizuta Y, Higashiyama T (2018) Chemical signalling for pollen tube guidance at a glance. J Cell Sci 131:208447. https://doi.org/10.1242/jcs.208447
Mollet JC, Kim S, Jauh GY, Lord EM (2002) Arabinogalactan proteins, pollen tube growth, and the reversible effects of Yariv phenylglycoside. Protoplasma 219:89–98. https://doi.org/10.1007/s007090200009
Moore JP, Nguema-Ona EE, Vicré-Gibouin M, Sørensen I, Willats WG, Driouich A, Farrant JM (2013) Arabinose-rich polymers as an evolutionary strategy to plasticize resurrection plant cell walls against desiccation. Planta 237:739–754. https://doi.org/10.1007/s00425-012-1785-9
Nguema-Ona E, Coimbra S, Vicré-Gibouin M, Mollet JC, Driouich A (2012) Arabinogalactan proteins in root and pollen-tube cells: distribution and functional aspects. Ann Bot 110:383–404. https://doi.org/10.1093/aob/mcs143
Okuda S, Tsutsui H, Shiina K, Sprunck S, Takeuchi H, Yui R et al (2009) Defensin-like polypeptide LUREs are pollen tube attractants secreted from synergid cells. Nature 458:357–361. https://doi.org/10.1038/nature07882
Pennell RI, Janniche L, Kjellbom P, Scofield GN, Peart JM, Roberts K (1991) Developmental regulation of a plasma membrane arabinogalactan protein epitope in oilseed rape flowers. Plant Cell 3:1317–1326. https://doi.org/10.1105/tpc.3.12.1317
Pereira AM, Lopes AL, Coimbra S (2016) Arabinogalactan proteins as interactors along the crosstalk between the pollen tube and the female tissues. Front Plant Sci 7:1895. https://doi.org/10.3389/fpls.2016.01895
Pereira AM, Masiero S, Nobre MS et al (2014) Differential expression patterns of arabinogalactan proteins in Arabidopsis thaliana reproductive tissues. J Exp Bot 65:5459–5471. https://doi.org/10.1093/jxb/eru300
Pereira LG, Coimbra S, Oliveira H, Monteiro L, Sottomayor M (2006) Expression of arabinogalactan protein genes in pollen tubes of Arabidopsis thaliana. Planta 223:374–380. https://doi.org/10.1007/s00425-005-0137-4
Pereira A, Pereira L, Coimbra S (2015) Arabinogalactan proteins: rising attention from plant biologists. Plant Reprod 28:1–15. https://doi.org/10.1007/s00497-015-0254-6
Pérez-Pérez Y, Carneros E, Berenguer E, Solís MT, Bárány I, Pintos B, Gómez-Garay A, Risueño MC, Testillano PS (2018) Pectin de-methylesterification and AGP increase promote cell wall remodeling and are required during somatic embryogenesis of Quercus suber. Front Plant Sci 9:1915. https://doi.org/10.3389/fpls.2018.01915
Punwani JA, Drews GN (2008) Development and function of the synergid cell. Sex Plant Reprod 21:7–15. https://doi.org/10.1007/s00497-007-0059-3
Punwani JA, Rabiger DS, Drews GN (2007) MYB98 positively regulates a battery of synergid-expressed genes encoding filiform apparatus-localized proteins. Plant Cell 19:2557–2568. https://doi.org/10.1105/tpc.107.052076
Sandaklie-Nikolova L, Palanivelu R, King EJ, Copenhaver GP, Drews GN (2007) Synergid cell death in Arabidopsis is triggered following direct interaction with the pollen tube. Plant Physiol 144:1753–1762. https://doi.org/10.1104/pp.107.098236
Sankaranarayanan S, Higashiyama T (2018) Capacitation in plant and animal fertilization. Trends Plant Sci 23:129–139. https://doi.org/10.1016/j.tplants.2017.10.006
Speranza A, Taddei AR, Gambellini G, Ovidi E, Scoccianti V (2009) The cell wall of kiwifruit pollen tubes is a target for chromium toxicity: alterations to morphology, callose pattern and arabinogalactan protein distribution. Plant Biol 11:179–193. https://doi.org/10.1111/j.1438-8677.2008.00129.x
Qin Y, Chen D, Zhao J (2007) Localization of arabinogalactan proteins in anther, pollen, and pollen tube of Nicotiana tabacum L. Protoplasma 231:43–53. https://doi.org/10.1007/s00709-007-0245-z
Sogo A, Tobe H (2006) Delayed fertilization and pollen-tube growth in pistils of Fagus japonica (Fagaceae). Am J Bot 93:1748–1756. https://doi.org/10.3732/ajb.93.12.1748
Sogo A, Tobe H (2008) Mode of pollen tube growth in pistils of Ticodendron incognitum (Ticodendraceae, Fagales) and the evolution of chalazogamy. Bot J Linn Soc 157:621–631. https://doi.org/10.1111/j.1095-8339.2008.00807.x
Solís MT, Pintos T, Prado MJ et al (2008) Early markers of in vitro microspore reprogramming to embryogenesis in olive (Olea europaea L.). Plant Sci 174:597–605. https://doi.org/10.1016/j.plantsci.2008.03.014
Su S, Higashiyama T (2018) Arabinogalactan proteins and their sugar chains: functions in plant reproduction, research methods, and biosynthesis. Plant Reprod 31:67–75. https://doi.org/10.1007/s00497-018-0329-2
Suárez C, Zienkiewicz A, Castro A, Zienkiewicz K, Majewska-Sawka A, Rodríguez-García M (2013) Cellular localization and levels of pectins and arabinogalactan proteins in olive (Olea europaea L.) pistil tissues during development: implications for pollen–pistil interaction. Planta 237:305–319. https://doi.org/10.1007/s00425-012-1774-z
Vaughn KC, Talbot MJ, Offler CE, McCurdy DW (2007) Wall ingrowths in epidermal transfer cells of Vicia faba cotyledons are modified primary walls marked by localized accumulations of arabinogalactan proteins. Plant Cell Physiol 48:159–168. https://doi.org/10.1093/pcp/pcl047
Yariv J, Lis H, Katchalski E (1967) Precipitation of arabic acid and some seed polysaccharides by glycosylphenylazo dyes. Biochem J 105:1C–2C
Zhong J, Ren Y, Yu M, Ma T, Zhang X, Zhao J (2011) Roles of arabinogalactan proteins in cotyledon formation and cell wall deposition during embryo development of Arabidopsis. Protoplasma 248:551–563. https://doi.org/10.1007/s00709-010-0204-y
Acknowledgements
Authors are grateful to Reyes Lopez for technical assistance, and Nuria Blanco-Moure for helping with graphs and data analysis. We thank William E. Friedman for his support and for providing laboratory equipment during part of this work. This work was supported by Ministerio de Ciencia e Innovación (MICINN)-FEDER [AGL2006-13529-C02-01, AGL 12621-C02-01, AGL 2012–40239] and Gobierno de Aragón [group A43]. JIM antibodies distribution was partly supported by NSF grants [DBI-0421683, RCN 009281]. JML was supported by a FPI fellowship [BES-2007-16059] from MICINN.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Tetsuya Higashiyama.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
AGPs labeled with JIM13 mAb in the nucellus of apple ovules before and after pollen tube penetration. A. Glycoprotein epitopes in the surface of a row of cells leading to the egg apparatus (white arrows), at anthesis (0 daa). B. At the time of pollen tube penetration and fertilization, these glycoproteins vanish from the areas of pollen tube growth. Two-micrometer sections of ovules were immunolocalized with JIM13 mAb and detected with an Alexa 488 anti-rat secondary antibody conjugated with FITC that shows a green color. Daa, days after anthesis; dap, days after pollination; nu, nucellus; pt, pollen tube. Scale bars: 20 µm. (TIFF 4493 kb)
Supplementary Fig. 2
Changes in the filiform apparatus of the synergids during the post-pollination stages in apple. A. While the embryo sac accumulates starch, the filiform apparatus of the synergids (black arrows) shows accumulation of polysaccharides three days after pollination (3 dap). B. Concomitant with pollen tube arrival, one synergid degenerates (black arrow). C. Following fertilization, upon zygote formation, the filiform apparatus (black arrows) degenerates, losing polysaccharide staining. Two-micrometer sections of ovules stained with periodic acid–Shiff’s reagent (PAS) for insoluble polysaccharides (pink to purple color). Dap, days after pollination; es, embryo sac; fa, filiform apparatus; pt, pollen tube; syn, synergid; zyg, zygote. Scale bars: 20 µm. (JPEG 2931 kb)
Rights and permissions
About this article
Cite this article
Losada, J.M., Herrero, M. Arabinogalactan proteins mediate intercellular crosstalk in the ovule of apple flowers. Plant Reprod 32, 291–305 (2019). https://doi.org/10.1007/s00497-019-00370-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-019-00370-z