Abstract
The aim of this study was to assess the effect of (preventive) rehabilitation on swallowing and mouth opening after concomitant chemoradiotherapy (CCRT). Forty-nine patients with advanced oral cavity, oropharynx, hypopharynx and larynx, or nasopharynx cancer treated with CCRT were randomized into a standard (S) or an experimental (E) preventive rehabilitation arm. Structured multidimensional assessment (i.e., videofluoroscopy, mouth-opening measurement, structured questionnaires) was performed before and 10 weeks after CCRT. In both S and E arms, feasibility was good (all patients could execute the exercises within a week) and compliance was satisfactory (mean days practiced per week was 4). Nevertheless, mouth opening, oral intake, and weight decreased significantly. Compared to similar CCRT studies at our institute, however, fewer patients were still tube-dependent after CCRT. Furthermore, some functional outcomes seemed less affected than those of studies in the literature that did not incorporate rehabilitation exercises. Patients in the E arm practiced significantly fewer days in total and per week, but they obtained results comparable to the S arm patients. Preventive rehabilitation (regardless of the approach, i.e., experimental or standard) in head and neck cancer patients, despite advanced stage and burdensome treatment, is feasible, and compared with historical controls, it seems helpful in reducing the extent and/or severity of various functional short-term effects of CCRT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Curative treatment with concomitant chemoradiotherapy (CCRT) for patients with advanced cancer of the head and neck has adverse effects on many functions of the upper respiratory and digestive systems. Sequelae such as pain, edema, xerostomia, and fibrosis negatively affect mouth opening (trismus), chewing, swallowing, and speech [1]. Several studies that investigated the long-term effects of CCRT have concluded that dysfunction in swallowing and nutrition tends to be persistent and can be severe [2–4]. The anatomical structures involved in swallowing seem to be especially vulnerable during CCRT. Normally, retraction of the tongue base pushes the food bolus downward and prevents residue that may be aspirated, and the elevation and forward movement of the larynx contributes to closure of the airway and the opening of the esophagus, allowing the bolus to pass [5]. A number of studies have shown that CCRT has a negative effect on tongue base retraction and larynx elevation, resulting in delayed transport of the bolus, the occurrence of residue, and aspiration [2, 4, 6, 7]. Some patients are already confined to tube feeding before treatment [8] or will be during the course of the treatment [4, 7]. In these patients, muscle inactivity will cause atrophy of the swallowing muscles [2]. Trismus (limited mouth opening) is another negative effect of CCRT that has to be taken into consideration [9]. Trismus may cause poor oral hygiene, limit dental intervention and medical surveillance, and impair swallowing and speech even further [10].
Given these well-known functional sequelae of CCRT, one would have expected more studies on rehabilitation strategies, comparable to postlaryngectomy rehabilitation, for example [11]. Reliable information on the effect of rehabilitation is all the more urgent since, the use of CCRT is on the rise, especially in oral cavity, oropharynx, and nasopharynx cancers, as it has been shown to considerably improve life expectancy [12]. Therefore, more patients are subject to the aforementioned long-term functional deficits, which negatively influence their quality of life [13–15]. As yet, no prospective, randomized study has investigated the effects of (precautionary) rehabilitation exercises on (long-term) swallowing or mouth-opening problems caused by CCRT [15] in spite of many effective tongue and swallowing exercises described in literature [16–21] and the fact that delayed swallowing therapy is not effectual [22]. Some positive results that have been found are, for example, improvement of dysphagia-specific quality of life scores after implementation of pretreatment swallowing education and exercises [13]. Another (pilot) study provided evidence that pretreatment swallowing exercises do improve post-treatment swallowing function in head and neck cancer patients receiving CCRT [16]. However, both studies were nonrandomized and the sample sizes were small.
In the Dutch “Oral and Oropharyngeal Carcinoma Guideline,” it is stated that patients with head and neck cancer who are treated with curative (chemo)radiation should routinely receive appropriate logopedic rehabilitation, during and after treatment [23]. In the Netherlands, it is no longer considered ethical to withhold this form of rehabilitation from these patients, precluding the inclusion of a nonrehabilitation control group in any randomized controlled trial dealing with this topic. Although standard logopedic rehabilitation is taken to be routine, it is uncertain whether this still is the most optimal rehabilitation approach because alternative, innovative exercises have been developed recently and have proven to be effective [24]. Furthermore, it is unknown whether these relatively ill patients are able or willing to spend extra time and energy on such rehabilitation programs while undergoing burdensome CCRT.
Therefore, a prospective randomized clinical trial (RCT) was designed to assess the preventive effects of two rehabilitation programs for patients with advanced head and neck cancer treated with CCRT. In this article we focus on the feasibility of these programs, patient compliance, and the short-term functional effects (10 weeks post treatment).
Patients and Methods
Patients with advanced (stages III and IV) squamous cell carcinoma of the head and neck treated at The Netherlands Cancer Institute with concomitant chemoradiotherapy (CCRT) were invited to participate in this randomized controlled clinical trial. The study protocol was approved by the medical ethical review board of the institute, and written informed consent was obtained from all patients before entering the study. The patients were consecutively recruited over a period of 20 months (see Table 1 for the inclusion and exclusion criteria).
All patients received 100 mg/m2 cisplatin as a 40-min IV infusion on days 1, 22, and 43. Intensity-modulated radiotherapy (IMRT) of 70 Gy in 35 fractions was administered over 7 weeks starting concurrently with chemotherapy.
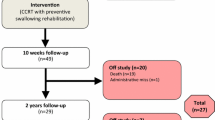
During the accrual period of the clinical trial, a total of 72 patients were treated with CCRT. Seventeen patients could not be included in this rehabilitation trial because of patient refusal (4), follow-up abroad (2), administrative miss (1), cognitive problems (6, mostly Korsakov’s disease), or physical problems (4, i.e., Bechterew’s disease, tetraplegia, 2× jaw abnormalities), leaving 55 patients (76%) for inclusion in the study. Patients were randomized into two rehabilitation groups (standard versus experimental, see below) and were categorized according to tumor site (oral cavity and oropharynx, hypopharynx and larynx, and nasopharynx). Complete post-treatment analysis was done for 49 patients; 6 patients had to be excluded because of change in treatment plan (1), progressive disease (2), death (2), and patient refusal (1). The Consort flow diagram of patient randomization and analysis is shown in Fig. 1 [25]. Patients were assessed according to a multidimensional protocol (see below) before treatment and approximately 10 weeks (mean 10 weeks; range = 9–12; one outliner had the measurements at 16 weeks post-treatment) after their last radiotherapy.
CONSORT flow diagram of patient randomization and analysis [25]
There were 39 males and 10 females, with a mean age of 57 years (range = 32–78 years). Patient characteristics, including sites and stages, are given in Table 2. Staging was accomplished according to the International Union against Cancer (UICC), 5th edition (2005).
Rationale for Exercises
All exercises chosen for this study were included to improve the most frequently occurring swallowing problems after CCRT. A systematic review of the literature showed that swallowing disorders of varying severity occurred after CCRT [15]. Most frequently, a reduction of the tongue base retraction was found. Other problems that often occurred were reduced tongue strength, slowed/delayed laryngeal vestibule closure, reduced laryngeal elevation, and problems with mouth opening [15, 26]. Only exercises and maneuvers relevant to the problems identified in the literature and with proven effectiveness in earlier studies were included in the rehabilitation programs.
With regard to standard logopedic rehabilitation, several studies have investigated the influence of stretch (or range of motion) and strength exercises [17–19, 21, 27–29]. Veis et al. [21] analyzed several stretch exercises and concluded that the gargle task elicited the greatest tongue base retraction in a group of subjects with suspected dysphagia (e.g., head/neck cancer, progressive and sudden onset neurologic damage, Parkinson’s disease, stroke, muscular dystrophy). Buchbinder et al. [27] compared three mobilization regimens for the prevention of jaw hypomobility and concluded that the initial maximum interincisor mouth opening (MIO) in all three groups increased, but increased significantly more in the group that used the TheraBite® Jaw Motion Rehabilitation System™ (Atos Medical AB, Hörby, Sweden). Dijkstra et al. [28] also found a significantly increased mouth opening after physical therapy (including the therapeutic tools of rubber plugs, tongue blades, dynamic bite opener, and the TheraBite). Incidentally, the increase in mouth opening was significantly greater for patients with trismus not related to cancer.
Lazarus et al. [18] evaluated the effects of several voluntary maneuvers in head and neck cancer patients and concluded that tongue base-pharyngeal wall pressure increased most with the use of the effortful swallow (greatest pressure) and the tongue-holding maneuvers (also known as the Masako). These two maneuvers have also been shown to be effective in healthy individuals. Hind et al. [17] found improved oral (tongue-palate) pressures in healthy middle-aged and older adults who used the effortful swallow maneuver during swallowing compared to those who did not. Fujiu et al. [29] found a significant increase in posterior pharyngeal wall bulging in young healthy adults who used the Masako maneuver. Another maneuver that has been proven effective is the super-supraglottic swallow. Logemann et al. [19] reported that this maneuver resulted in changes in airway entrance closure and hyolaryngeal movement. The authors observed fewer swallowing disorders in irradiated head and neck cancer patients who used the super-supraglottic swallow maneuver.
In this study, one group underwent a rehabilitation program based on the aforementioned most effective methods currently available in a logopedic setting [standard (S) rehabilitation], while the other group used a “new” exercise regime using a special device-based rehabilitation protocol [experimental (E) rehabilitation]. In the E rehabilitation group, the patients used the TheraBite Jaw Motion Rehabilitation System to stretch and strengthen the same swallowing and jaw muscles (and with the same frequency) as the S group. The TheraBite is a handheld portable medical device specifically designed to treat trismus and mandibular hypomobility [27, 30]. The rationale for the E rehabilitation was that tolerance of the TheraBite is good, it is easy to use, and compliance tends to be high [30]. Furthermore, Burkhead et al. [24] studied the effect of jaw and tongue positioning in suprahyoid muscle function during swallowing with the use of the TheraBite. These authors concluded that a relative change in muscle activity across conditions showed an incremental increase with jaw position. In addition, it was assumed that the swallowing exercises with the TheraBite would be less complex and easier to learn than standard logopedic exercises. Logopedic strengthening exercises are known to be quite complex and require the ability to follow instructions carefully and accurately [31, 32], which might influence compliance negatively.
Procedure
In a separate session approximately 2 weeks before the onset of CCRT, participants were provided with verbal and written instructions (by LvdM) so that they could perform the exercises independently and practice daily, in the hospital or at home.
Exercises
Specific instructions for both rehabilitation programs are given in the “Appendices” section. In summary, the S rehabilitation consisted of range-of-motion exercises and three strengthening exercises, i.e., the effortful swallow, the Masako maneuver, and the super-supraglottic swallow. The stretch exercise of the E rehabilitation consisted of a passive and slow opening of the mouth using the TheraBite device. The strengthening exercise consisted of swallowing with the tongue elevated to the palate while maintaining mouth opening at 50% of its maximum, thus training the suprahyoid muscles [24]. If needed (i.e., xerostomia), patients were allowed to use a sip of water either before or after initiating the swallow, but not during the exercise.
Schedule
Patients were encouraged to practice three times daily and to integrate the exercise into other daily activities such as brushing teeth. In this RCT we closely followed the recommendations of the American College of Sports Medicine Position Stand [33]. They recommend holding a stretch for 10–30 s at the point of mild discomfort since no greater benefit is derived from longer durations. They also recommend 8–12 repetitions. Even though these recommendations are related to healthy individuals and should be used in the context of “participant’s needs, goals, and initial abilities,” we expected the training schedule to be realistic and feasible in daily practice, even during CCRT. The specific schedule for each rehabilitation is given in the “Appendices” section.
Outcome Measures
Before onset and approximately 10 weeks after completing CCRT, a multidimensional assessment protocol was used to evaluate functional outcomes in all 49 patients. Details about this protocol have been reported previously [34]. In short, the protocol included standard videofluoroscopy to assess the laryngeal penetration and aspiration scale (validated PAS) [35] and the presence of contrast residue. Maximum interincisor mouth opening (MIO), weight changes, and body mass index (BMI) were measured. The functional oral intake scale (FOIS) was also included, as was a structured, study-specific questionnaire for quality-of-life evaluation, and a visual analog scale (VAS) for pain assessment [36].
To evaluate the feasibility of the two rehabilitation programs and compliance with the exercises, familiarity with the exercises was evaluated and scored (1 = very familiar with the exercises, 2 = fairly familiar with the exercises, and 3 = not at all familiar). Moreover, specific items were included in the questionnaire, such as the number of practice days per week (from 0 being less than 1 day a week to 7 being 7 days a week), the total number of practice days, and the reason for quitting.
Statistical Analysis
All statistical analyses were performed using SPSS v15.0.1 (SPSS, Inc., Chicago, IL). Randomization was performed centrally by computer at the Netherlands Cancer Institute using blocks of 6, stratified by tumor site (larynx and hypopharynx, nasopharynx, and oral cavity and oropharynx) (ALEA software).
An intention-to-treat analysis was carried out on all randomized patients. For associations of nominal variables with the rehabilitation, Fisher’s exact tests were applied, whereas for ordinal variables the Wilcoxon rank sum test or the Jonckheere Terpstra test was applied. Continuous variables were compared by means of the Wilcoxon rank sum test. For all analyses, p ≤ 0.05 was considered statistically significant. Inter- and intraobserver reliability for the aspiration and residue measurements (20% of the tapes rated by LvdM and AK) was determined using the kappa statistic.
Results
Feasibility and Compliance
To evaluate the feasibility of the program, the exercises themselves and also performing the rehabilitation concurrently with CCRT were analyzed. Thirty-four patients (69%) could perform the exercises immediately and the remaining 15 patients within a week. Twenty-six patients (53%) did not have any problems with any of the exercises. The remaining 23 patients (47%) had problems mainly with the swallowing (strength) maneuvers. With respect to the appreciation for the exercises, 10 patients (20%) liked the exercises, 25 patients (51%) were neutral about the training, 12 patients (25%) did not like them, and only 2 patients (4%) really disliked the exercises. There was no difference between the exercise groups in this respect. Reasons for disliking the exercises were “the exercises are strange” and “it is annoying,” and some disliked the daily practice requirement. The rehabilitation ran parallel to the burdensome CCRT and 28 patients (57%) stopped practicing after an average of 27 days (range = 2–98 days). Reasons such as “it went well” or “I forgot to practice” were mentioned by 10 of the patients (20%), and 18 patients (37%) stopped training because of pain in the mouth, nausea, and fatigue. The overall score for the expectation about the effectiveness of the exercises was good: 44 patients (90%) thought the exercises were helpful.
Compliance with the exercise program was evaluated by the “familiarity with the exercises” score and the trial-specific questionnaire. None of the patients had done logopedic therapy prior to their cancer treatment and therefore all patients initially scored “not at all familiar with the exercises.” After treatment, 28 patients (57%) scored “very familiar with the exercises,” 15 patients (31%) were “fairly familiar with the exercises,” and 6 (12%) were “not at all familiar with the exercises.” With respect to the intensity of the treatment, while patients were encouraged to train every day over the entire CCRT and post-treatment period, it appeared that only 7 patients (14%) did, while on average patients practiced 4 days per week (range = 0–7 days). The mean number of days practiced over the observation period of approximately 120 days was 50 days (range = 0–126 days). A further observation about compliance was that female patients practiced significantly more than males (p = 0.05; mean = 64 and 47 days, respectively), and that patients in the standard (S) group practiced significantly more than the patients in the experimental (E) group (p = 0.05; mean = 59 and 41 days, respectively). No other significant differences between the two rehabilitation arms with respect to feasibility and compliance were found.
Short-term Clinical Effects: Overall
Table 3 gives an overview of all pre- and post-treatment outcomes.
Swallowing function was assessed with videofluoroscopy, using the PAS, and determination of the presence of contrast residue. The interobserver and intraobserver reliabilities of the video assessments were good: for the PAS they were 0.98 and 0.88 and for the presence of residue they were 0.80 and 0.89, respectively. Combining laryngeal aspiration and penetration in the PAS, no significant difference was found between post- and pretreatment scores [12 patients (24%) vs. 9 patients (18%), respectively; p = 0.37), with (for comparison with the literature) a mean score of 0.95 vs. 1.15 for swallowing the different consistencies [16]. With respect to the presence of contrast residue, significantly less residue was found after treatment for three of the four different consistencies (5 cc liquid, p < 0.01; 3 cc paste, p = 0.02; cookie, p = 0.01).
Comparing the pre- and post-treatment maximum mouth opening (MIO), a significant decrease over time was found (from 50 to 47 mm, respectively; p < 0.01), but not in occurrence of trismus (MIO <35 mm; from 5 to 7 patients; p = 0.70).
With respect to weight, there was a significant mean weight loss of 6.1 kg (range = 78.1–72.0 kg; p < 0.01), but the BMI did not decrease significantly (p = 0.25) over the observation period.
Oral intake, as assessed with the overall functional oral intake scale (FOIS), significantly decreased during treatment from a mean score of 7 to a mean of 5 (a higher score indicates a more normal intake; p = 0.02). For comparison with the literature, the number of patients with a FOIS score of 1, 2, and 3, indicating an oral intake of less than 50%, and with the scores 4–7, indicating an oral intake of more than 50%, is also given [7, 32, 37, 38]. Two patients (4%) in the pretreatment and 17 patients (35%) in the post-treatment group had a FOIS score of 1–3. This is in concordance with the increase in tube dependency: before treatment 2 (4%) of the patients were tube dependent and at the 10-week post-treatment observation moment, 18 were tube dependent (37%; p < 0.01). However, this latter figure is a considerable improvement in comparison with the tube dependency at the last day of radiotherapy when 38 of the 49 patients (76%) still needed a (naso) gastric feeding tube. When compared with the 10-week post-treatment observation, this was a significant improvement, with 20 patients regaining their oral intake in this period (p < 0.01).
With respect to pain, no significant differences over time were noted: the pain scores (no, mild, moderate, and severe pain) did not vary much (p = 0.42). Patients’ perceived function and function-related quality of life pre- and post-treatment were assessed with a study-specific questionnaire. The reliability coefficients (Cronbach’s alpha) varied between 0.516 and 0.867, indicating that the internal consistency of the set of items in the different subscales was acceptable. Patients perceived a significantly smaller mouth opening after treatment than before the onset of treatment (p = 0.032). No differences between the pre- and post-treatment answers on the swallowing and information subscales were found.
Short-term Clinical Effects: Comparison of the Two Rehabilitation Groups
Randomization for the two rehabilitation arms did not show statistically significant differences with respect to site of the primary tumor and stage. As a consequence of the stratification with respect to tumor site only, there was a significant difference in the gender distribution, with 9 female patients of the 25 patients in the S arm and only 1 female of the 24 patients in the E arm (p = 0.01).
As already mentioned, female patients practiced significantly more days than males (mean = 64 and 47 days, respectively; p = 0.05), and patients in the S group practiced significantly more days than the patients in the E group (mean = 59 and 41 days, respectively; p = 0.05). Despite the fact that patients in the E arm practiced significantly fewer days in total and per week, they showed similar results compared to the patients in the S arm.
Another significant difference at 10-weeks post treatment was that the E group showed significantly less residue after swallowing Omnipaque-coated cake (p = 0.021) compared with the S group. For the other consistencies, no significant differences between the two rehabilitations were found. There were also no significant differences found for the PAS between groups. The only other statistically significant difference between the groups was that males in the E group reported significantly more pain than the males in the S group (p < 0.02).
Discussion
The high incidence of tumor-related functional sequelae in advanced head and neck cancer at initial diagnosis [34] and the burdensome treatment with concomitant chemoradiotherapy (CCRT) with its inherent local toxicity often result in complete cessation of oral intake and prolonged periods of tube feeding [15]. The consequent lack of masticatory and pharyngeal muscle activity is an additional reason for the frequent and significant delay in post-treatment swallowing recovery. Therefore, continued muscle activation throughout the treatment and post-treatment period to prevent or limit the necessity of tube feeding and maintain swallowing function seems self-evident. However, the feasibility of complying with a rehabilitation program while also undergoing oncologic treatment has not been systematically studied before. The present randomized clinical trial (RCT) shows that despite the burdensome CCRT, most patients (69%) were still able to perform the exercises immediately after instructions were given. With most of the patients training 4 days a week and 14% even training every day, compliance can also be considered acceptable.
The absence of a control group (without rehabilitation) may be considered a weakness of this study, but this could not be avoided since withholding rehabilitation is no longer considered ethical according to the guidelines of the Dutch Head and Neck Cooperative Group [23]. However, 76% tube feeding at the end of treatment and 37% at 10 weeks post treatment compare favorably with a comparable randomized CCRT study at our institute, in which these figures were 86% (157/183) at the end of treatment and 62% (112/180) at 12 weeks [39]. Including a (control) group that received the standard and the experimental exercises might have provided information about the added value of the experimental exercises, but a third therapy group would have required a much larger number of patients to power the study and, consequently, a logistically unfeasible increase in the accrual period. Furthermore, because chemoradiation treatment is very demanding, our aim was to attain the best result with the least possible burden to the patients. Since patient compliance with a standard exercise program was still unknown, exposing patients to a combined program seemed unreasonable to us.
Interestingly, patients who practiced daily did not differ significantly from those who did not in any of the outcome parameters. Another preventive study focusing on trismus did show a trend toward better results in patients who applied the exercises described by Santos and Buchbinder compared to those who did not exercise [40]. Carroll et al. [13, 16] also suggested that implementation of pretreatment exercises produces measurable improvements in post-treatment swallowing function or dysphagia-specific quality of life. Unfortunately, patients’ compliance was not addressed in these studies. To achieve good compliance and thus better results, training programs must be realistic. The American College of Sports Medicine Position Stand suggests that resistance training for the average healthy adult should be performed at least 2–3 days/week [33]. Keeping this recommendation in mind, the results of the present study, with most patients practicing 4 days a week, are quite good regarding exercise frequency, especially considering the burdensome cancer treatment this specific population had to undergo. Another important factor influencing compliance is the support and assistance given. Melchers et al. [41] explored factors that may influence adherence to performing the TheraBite exercise. They reported that internal motivation to exercise, the perceived effect, self-discipline, and having a clear exercise goal positively influenced adherence to the TheraBite exercise regimen. Perceiving no effect, limitation in TheraBite opening range, and reaching the exercise goal or a plateau in mouth opening were negative influences. Regular assistance/support is thus important to counteract these negative influences. If the assistance/support in this study had been more intensive (during and after treatment, when patients were at home), the clinical short-term effects might have been better.
In the present protocol, both rehabilitation protocols could not prevent a small (3 mm) but significant decrease in mouth opening, but the decrease might have been greater if no exercise was performed [22, 27, 28]. Since preventive exercises can address only muscle activity and not CCRT-related mucosal toxicity, the significant decrease in weight and oral intake might have been a reflection of pain and altered metabolism rather than the exercises. Interestingly, training seems to have a positive effect on the presence of post-swallow residue after CCRT: significantly less contrast residue was found for all consistencies except 1 cc liquid. However, the loss of tumor mass could have influenced these results. Comparing the two rehabilitation groups, it is interesting to note at this early stage of post-treatment evaluation that the E group showed significantly less contrast residue when swallowing Omnipaque-coated cake than the S group, which might suggest better mucosal clearance and improved (underlying) muscle activity, leading to greater pressure for clearance during pharyngeal constriction in the E group. We have no explanation for the perceived pain difference between the groups, but the fact that more males were included in the E group is noteworthy.
Randomization was stratified according to tumor site and not to sex, thus resulting in only one female included in the E group; therefore, no comparisons could be made between sex and the different exercise regimes. The sex differences between the groups could have influenced the results: patients in the E group practiced significantly fewer days in total and per week compared to the S group, and females practiced significantly more days/week than males. This under-representation of females in the E group could be an explanation for these differences. Furthermore, this might also explain why there was no evidence for our expectation that tolerance, compliance, and feasibility of the exercises would be better in the E group (using the TheraBite). Thus, in terms of the short-term functional outcomes, one has to conclude that both exercise regimes seem equally effective.
Comparison with the Literature
Aside from comparing the functional outcomes of this RCT with historical controls from our own institute [39], a comparison with the literature was made to assess whether patients in this trial did better than patients not exercising. Table 4 gives an overview of the relevant studies that reported on functional outcomes (aspiration, tube dependency, nutrition, weight, mouth opening), described in the systematic review by Van der Molen et al. [15] or Dijkstra et al. [26]. Studies published after the search period [37, 39, 40] and/or reporting on rehabilitation were also reviewed [13, 16, 40]. Only studies of patients undergoing CCRT (supplemented with two RT-only studies reporting on trismus) and which reported on early follow-up results (until 3 months post treatment) were analyzed.
The PAS in our study was a mean of 1.15, better than the 4.11 obtained in the study by Carroll et al. [16] in which patients also exercised during treatment. Also, mean weight loss was somewhat less in our study (6.1 kg, 7.8%), than in the studies of Newman et al. [42] (10%) and Oates et al. [43] (mean = 8.2 kg; 13%).
Other studies occasionally had better scores for the aspiration rate, mean reduction in mouth opening, oral intake, and tube dependency. For example, in the study by Newman et al. [43], less aspiration was found in the CCRT group (7%) than the present study (18%). In other studies, aspiration ranged from 23% to as high as 65% [2, 6, 16, 38, 44]. Whether the presence of residue in our study is better or comparable with that of studies that did not include groups receiving rehabilitation is unclear. Several studies used the oropharyngeal swallow efficiency (OPSE) to measure the percentage of oral and pharyngeal residue instead of the occurrence of residue above and/or below the valleculae [7, 38, 45]. The least reduction in mouth opening was found by Grandi et al. [40]. Their patients, who did the Santos exercises, showed a mean reduction in mouth opening of 1.38 mm. In the present study and in the other two groups of the study by Grandi et al. [40], a mean reduction of 3, 3.80, and 4.94 (no exercises) was found, respectively. The study by Buchbinder et al. [27] included only patients who already showed trismus, so it is not surprising that the mean mouth opening is much smaller. All patients in that study performed exercises using the TheraBite which showed a therapeutic effect that was indicated by an increase in mouth opening. However, this was a therapeutic and not a preventive trial and as such not comparable to our study. Analyzing the oral intake (less than 50% by mouth) and tube dependency, patients in the study by Lazarus et al. [37] showed the best scores (18 vs. 18%). In the present study, scores of 35 vs. 37% were found, with a wide range of 23–79% and 26–93% in other studies [7, 32, 38, 39, 42–44].
A disadvantage of this comparison obviously is the heterogeneity of the research coming from a range of institutes, the inclusion of different tumor sites, the different treatment protocols used, the mostly small sample sizes, and the different assessment tools used. This makes it difficult to come to any firm conclusions. Therefore, as already mentioned, a comparison with the results obtained earlier in our institute seems more relevant [39]. Thus, overall the results of this study are comparable or somewhat better than the results reported in the literature and much better when compared to earlier results from our institute, which is encouraging. Nevertheless, it is interesting to investigate why some functional deficits like trismus or aspiration still occur despite rehabilitation programs. An explanation might be that these patients received higher mean RT doses (Gy) on the muscles critical to swallowing function. An increasing number of studies focus on the relationship between the severity of swallowing problems or trismus and the mean radiation doses on the swallowing and jaw structures [2, 46, 47]. Teguh et al. [47] reported that a significant correlation was observed between dose in masseter and pterygoid muscles and trismus. They also concluded that with every additional 10 Gy to the pterygoid muscle, there is a 24% increase in the probability of trismus. It is therefore interesting to analyze whether patients in our study, who still showed severe functional problems after treatment, received a larger mean RT dose on the involved muscles. Such studies are presently under way and will be published separately.
Based on the results obtained in this study, it is not possible to conclude that pretreatment exercises are efficacious in preventing swallowing problems and trismus, but they seem to reduce the extent and severity of the functional problems that occur after CCRT. Further dose-effect and long-term clinical analyses are needed before any firm conclusions can be drawn.
Conclusion
This study shows that pretreatment rehabilitation is feasible and that acceptable compliance can be obtained despite the burdensome effects of CCRT. Early post-treatment functional outcomes for patients with advanced head and neck cancer seem to improve with either of the two exercise regimes used in this study. Long-term effects have to be awaited before definitive conclusions can be drawn about which of the two regimens, if any, is preferred.
References
Murry T, Madasu R, Martin A, Robbins KT. Acute and chronic changes in swallowing and quality of life following intraarterial chemoradiation for organ preservation in patients with advanced head and neck cancer. Head Neck. 1998;20:31–7.
Eisbruch A, Schwartz M, Rasch C, Vineberg K, Damen E, Van As CJ, Marsh R, Pameijer FA, Balm AJ. Dysphagia and aspiration after chemoradiotherapy for head-and-neck cancer: which anatomic structures are affected and can they be spared by IMRT? Int J Radiat Oncol Biol Phys. 2004;60:1425–39.
Smith RV, Kotz T, Beitler JJ, Wadler S. Long-term swallowing problems after organ preservation therapy with concomitant radiation therapy and intravenous hydroxyurea: initial results. Arch Otolaryngol Head Neck Surg. 2000;126:384–9.
Nguyen NP, Moltz CC, Frank C, Vos P, Smith HJ, Karlsson U, Dutta S, Midyett FA, Barloon J, Sallah S. Dysphagia following chemoradiation for locally advanced head and neck cancer. Ann Oncol. 2004;15:383–8.
Logemann JA. Evaluation and treatment of swallowing disorders. 2nd ed. Austin: TX: Pro-Ed; 1998.
Kotz T, Costello R, Li Y, Posner MR. Swallowing dysfunction after chemoradiation for advanced squamous cell carcinoma of the head and neck. Head Neck. 2004;26:365–72.
Newman LA, Robbins KT, Logemann JA, Rademaker AW, Lazarus CL, Hamner A, Tusant S, Huang CF. Swallowing and speech ability after treatment for head and neck cancer with targeted intraarterial versus intravenous chemoradiation. Head Neck. 2002;24:68–77.
Stenson KM, MacCracken E, List M, Haraf DJ, Brockstein B, Weichselbaum R, Vokes EE. Swallowing function in patients with head and neck cancer prior to treatment. Arch Otolaryngol Head Neck Surg. 2000;126:371–7.
Gaziano JE. Evaluation and management of oropharyngeal dysphagia in head and neck cancer. Cancer Control. 2002;9:400–9.
Goldstein M, Maxymiw WG, Cummings BJ, Wood RE. The effects of antitumor irradiation on mandibular opening and mobility: a prospective study of 58 patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88:365–73.
Hilgers FJ, Ackerstaff AH. Comprehensive rehabilitation after total laryngectomy is more than voice alone. Folia Phon Logop. 2000;52:65–73.
Carvalho AL, Nishimoto IN, Califano JA, Kowalski LP. Trends in incidence and prognosis for head and neck cancer in the United States: a site-specific analysis of the SEER database. Int J Cancer. 2005;114:806–16.
Kulbersh BD, Rosenthal EL, McGrew BM, Duncan RD, McColloch NL, Carroll WR, Magnuson JS. Pretreatment, preoperative swallowing exercises may improve dysphagia quality of life. Laryngoscope. 2006;116:883–6.
Louise KM, Brennan MT, Noll JL, Fox PC, Burri SH, Hunter JC, Lockhart PB. Radiation-induced trismus in head and neck cancer patients. Support Care Cancer. 2008;16:305–9.
van der Molen L, van Rossum MA, Burkhead LM, Smeele LE, Hilgers FJ. Functional outcomes and rehabilitation strategies in patients treated with chemoradiotherapy for advanced head and neck cancer: a systematic review. Eur Arch Otorhinolaryngol. 2009;266:889–900.
Carroll WR, Locher JL, Canon CL, Bohannon IA, McColloch NL, Magnuson JS. Pretreatment swallowing exercises improve swallow function after chemoradiation. Laryngoscope. 2008;118:39–43.
Hind JA, Nicosia MA, Roecker EB, Carnes ML, Robbins J. Comparison of effortful and noneffortful swallows in healthy middle-aged and older adults. Arch Phys Med Rehabil. 2001;82:1661–5.
Lazarus CL, Logemann JA, Rademaker A, Kahrilas PJ. Effects of voluntary maneuvres on tongue base function for swallowing. Folia Phoniatr Logop. 2002;54:171–6.
Logemann JA, Pauloski BR, Rademaker AW, Colangelo LA. Super-supraglottic swallow in irradiated head and neck cancer patients. Head Neck. 1997;19:535–40.
Ohmae Y, Logemann JA, Kaiser P, Hanson DG, Kahrilas PJ. Effects of two breath-holding maneuvers on oropharyngeal swallow. Ann Otol Rhinol Laryngol. 1996;105:123–31.
Veis S, Logemann JA, Colangelo L. Effects of three techniques on maximum posterior movement of the tongue base. Dysphagia. 2000;15:142–5.
Waters TM, Logemann JA, Pauloski BR, Rademaker AW, Lazarus CL, Newman LA, Hamner AK. Beyond efficacy and effectiveness: conducting economic analyses during clinical trials. Dysphagia. 2004;19:109–19.
van Zuiden M. Dutch guidelines “oral and oropharyngeal carcinoma” (NWHHT). Alphen aan den Rijn; 2004. p. 173–199.
Burkhead LM, Rosenbek JC, Sapienza CM, Musson N, Davenport P, Schechtman O. The effect of jaw and tongue position on suprayhoid muscle function during swallowing. Dysphagia. 2006;21:298.
Moher D, Schulz KF, Altman DG. The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomised trials. Lancet. 2001;357:1191–4.
Dijkstra PU, Kalk WW, Roodenburg JL. Trismus in head and neck oncology: a systematic review. Oral Oncol. 2004;40:879–89.
Buchbinder D, Currivan RB, Kaplan AJ, Urken ML. Mobilization regimens for the prevention of jaw hypomobility in the radiated patient: a comparison of three techniques. J Oral Maxillofac Surg. 1993;51:863–7.
Dijkstra PU, Sterken MW, Pater R, Spijkervet FK, Roodenburg JL. Exercise therapy for trismus in head and neck cancer. Oral Oncol. 2007;43:389–94.
Fujiu M, Logemann JA. Effect of a tongue-holding maneuver on posterior pharyngeal wall movement during deglutition. Am J Speech Lang Pathol. 1995;5:23–30.
Maloney GE, Mehta N, Forgione AG, Zawawi KH, Al-Badawi EA, Driscoll SE. Effect of a passive jaw motion device on pain and range of motion in TMD patients not responding to flat plane intraoral appliances. Cranio. 2002;20:55–66.
Lazarus CL. Effects of radiation therapy and voluntary maneuvers on swallow functioning in head and neck cancer patients. Clin Commun Disord. 1993;3:11–20.
Logemann JA, Pauloski BR, Rademaker AW, Lazarus CL, Gaziano J, Stachowiak L, Newman L, MacCracken E, Santa D, Mittal B. Swallowing disorders in the first year after radiation and chemoradiation. Head Neck. 2008;30(2):148–58.
American College of Sports Medicine Position Stand. The recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Med Sci Sports Exerc. 1998;30:975–91.
van der Molen L, van Rossum MA, Ackerstaff AH, Smeele LE, Rasch CR, Hilgers FJ. Pretreatment organ function in patients with advanced head and neck cancer: clinical outcome measures and patients’ views. BMC Ear Nose Throat Disord. 2009;9:10.
Rosenbek JC, Robbins JA, Roecker EB, Coyle JL, Wood JL. A penetration-aspiration scale. Dysphagia. 1996;11:93–8.
Jensen MP, Chen C, Brugger AM. Interpretation of visual analog scale ratings and change scores: a reanalysis of two clinical trials of postoperative pain. J Pain. 2003;4:407–14.
Lazarus C, Logemann JA, Pauloski BR, Rademaker AW, Helenowski IB, Vonesh EF, MacCracken E, Mittal BB, Vokes EE, Haraf DJ. Effects of radiotherapy with or without chemotherapy on tongue strength and swallowing in patients with oral cancer. Head Neck. 2007;29:632–7.
Logemann JA, Rademaker AW, Pauloski BR, Lazarus CL, Mittal BB, Brockstein B, MacCracken E, Haraf DJ, Vokes EE, Newman LA, Liu D. Site of disease and treatment protocol as correlates of swallowing function in patients with head and neck cancer treated with chemoradiation. Head Neck. 2006;28:64–73.
Ackerstaff AH, Balm AJ, Rasch CR, De Boer JP, Wiggenraad R, Rietveld DH, Gregor RT, Kroger R, Hilgers FJ. First-year quality of life assessment of an intra-arterial (RADPLAT) versus intravenous chemoradiation phase III trial. Head Neck. 2009;31:77–84.
Grandi G, Silva ML, Streit C, Wagner JC. A mobilization regimen to prevent mandibular hypomobility in irradiated patients: an analysis and comparison of two techniques. Med Oral Patol Oral Cir Bucal. 2007;12:E105–9.
Melchers LJ, van Weert E, Beurskens CH, Reintsema H, Slagter AP, Roodenburg JL, Dijkstra PU. Exercise adherence in patients with trismus due to head and neck oncology: a qualitative study into the use of the Therabite. Int J Oral Maxillofac Surg. 2009;38:947–54.
Oates JE, Clark JR, Read J, Reeves N, Gao K, Jackson M, Boyer M, O’Brien CJ. Prospective evaluation of quality of life and nutrition before and after treatment for nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg. 2007;133:533–40.
Newman LA, Vieira F, Schwiezer V, Samant S, Murry T, Woodson G, Kumar P, Robbins KT. Eating and weight changes following chemoradiation therapy for advanced head and neck cancer. Arch Otolaryngol Head Neck Surg. 1998;124:589–92.
Nguyen NP, Frank C, Moltz CC, Vos P, Smith HJ, Bhamidipati PV, Karlsson U, Nguyen PD, Alfieri A, Nguyen LM, Lemanski C, Chan W, Rose S, Sallah S. Aspiration rate following chemoradiation for head and neck cancer: an underreported occurrence. Radiother Oncol. 2006;80:302–6.
Graner DE, Foote RL, Kasperbauer JL, Stoeckel RE, Okuno SH, Olsen KD, Sabri AN, Maragos NE, Cha SS, Sargent DJ, Strome SE. Swallow function in patients before and after intra-arterial chemoradiation. Laryngoscope. 2003;113:573–9.
Levendag PC, Teguh DN, Voet P, van der Est H, Noever I, de Kruijf WJ, Kolkman-Deurloo IK, Prevost JB, Poll J, Schmitz PI, Heijmen BJ. Dysphagia disorders in patients with cancer of the oropharynx are significantly affected by the radiation therapy dose to the superior and middle constrictor muscle: a dose-effect relationship. Radiother Oncol. 2007;85:64–73.
Teguh DN, Levendag PC, Voet P, van der Est H, Noever I, de Kruijf W, van Rooij P, Schmitz PI, Heijmen BJ. Trismus in patients with oropharyngeal cancer: relationship with dose in structures of mastication apparatus. Head Neck. 2008;30:622–30.
Acknowledgments
The authors express gratitude to Anne Kornman, SLP, for her assistance in videofluoroscopy analyses. Harm van Tinteren, PhD, certified statistician, is acknowledged for his statistical support. Part of the study was supported by an unrestricted research grant from Atos Medical, Hörby, Sweden.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendices
Appendix A: Standard Rehabilitation


Appendix B: TheraBite Rehabilitation


Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
van der Molen, L., van Rossum, M.A., Burkhead, L.M. et al. A Randomized Preventive Rehabilitation Trial in Advanced Head and Neck Cancer Patients Treated with Chemoradiotherapy: Feasibility, Compliance, and Short-term Effects. Dysphagia 26, 155–170 (2011). https://doi.org/10.1007/s00455-010-9288-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00455-010-9288-y