Abstract
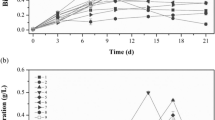
Glucose is the substrate most widely used as exogenous carbon source for heterotrophic cultivation of cyanobacteria. Due to limited information about the use of different carbohydrates as carbon sources to support cyanobacterial heterotrophic metabolism, the objective of this work was to evaluate different monosaccharides (arabinose, fructose, galactose, glucose, mannose and xylose), disaccharides (lactose, maltose, sucrose and trehalose) and polysaccharides (carboxymethylcellulose, cassava starch, Hi-maize®, maltodextrin Corn Globe 1805® and xylan) as exogenous carbon source for heterotrophic culture of cyanobacterium Phormidium sp. The batch cultivation using fructose as organic carbon source resulted in the highest (p < 0.05) cell biomass (5,540 mg/L) in parallel with the highest (p < 0.05) substrate yield coefficient (0.67 mgbiomass/mgfructose). Mannose was the carbon source with the highest (p < 0.05) substrate consumption rate (3,185.7 mg/L/day) and maltodextrin was the carbohydrate with major potential to produce biomass (1,072.8 mgbiomass/L/day) and lipids (160.8 mglipids/L/day). Qualitatively, the fatty acid profiles of the lipid extract from Phormidium sp. showed predominance of saturated chains for the cultures grown with most of the carbon sources, with the exception of the ones grown with xylose and maltodextrin.



Similar content being viewed by others
References
Queiroz MI, Hornes MO, Silva-Manetti AG, Jacob-Lopes E (2011) Single-cell oil production by cyanobacterium Aphanothece microscopica Nägeli cultivated heterotrophically in fish processing wastewater. Appl Energy 88:3438–3443
Perez-Garcia O, Escalante FME, de-Bashan LE, Bashan Y (2011) Heterotrophic cultures of microalgae: metabolism and potential products. Water Res 45:11–36
Smith AJ (1982) In: Carr NG, Whitton BA (eds) Modes of cyanobacterial carbon metabolism. The biology of cyanobacteria. University of California Press, Berkeley
Fay P (1983) The Blue–Greens (Cyanophyta–Cyanobacteria). In: Edward Arnold (ed) The Institute of Biology’s, Studies in Biology, no 160
Perez-Garcia O, Bashan Y, Puente ME (2011) Organic carbon supplementation of sterilized municipal wastewater is essential for heterotrophic growth and removing ammonium by the microalga Chlorella vulgaris. J Phycol 47:190–199
Lee YK (2004) Algal nutrition. Heterotrophic carbon nutrition. In: Richmond A (ed) Handbook of microalgal culture. Biotechnology and applied phycology. Blackwell Publishing, Oxford
Poole RJ (1978) Energy coupling for membrane transport. Annu Rev Plant Physiol 29:437–460
Al-Thukair AA, Abed RMM, Mohamed L (2007) Microbial community of cyanobacteria mats in the intertidal zone of oil-polluted coast of Saudi Arabia. Mar Pollut Bull 54:173–179
Cañizares-Villanueva RO, Ramos A, Lemus R, Gomez-Lojero C, Travieso L (1994) Growth of Phormidium sp. in aerobic secondary piggery wastewater. Appl Microbiol Biotechnol 42:487–491
Guiry MD, Guiry, GM (2013) AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. http://www.algaebase.org searched on October 2013
Rippka R, Derueles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
APHA, AWWA, WEF (2005) Standard methods for the examination of water and wastewater, 21st edn. Prot City Press, Baltimore
Bligh EG, Dyer JW (1959) A rapid method of total lipid extraction and purification. Can J Biochem Phisiol 37:911–917
Hartman L, Lago RCA (1976) A rapid determination of fatty acid methyl esters from lipids. Lab Prat 22:475–476
StatSoft, Inc. (2004) STATISTICA for Windows (Computer program manual). http://www.statsoftinc.com
Markou G, Georgakakis D (2011) Cultivation of filamentous cyanobacteria (blue–green algae) in agro-industrial wastes and wastewaters: a review. Appl Energy 88:3389–3401
Knowles VL, Plaxton WC (2003) From genome to enzyme: analysis of key glycolytic and oxidative pentose-phosphate pathway enzymes in the cyanobacterium Synechocystis sp. PCC 6803. Plant Cell Physiol 44:758–763
Wolk CP, Shaffer PW (1976) Heterotrophic micro- and macrocultures of a nitrogen-fixing cyanobacterium. Arch Microbiol 100:145–147
Raboy B, Padan E (1978) Active transport of glucose and α-methylglucoside in the cyanobacterium Plectonema boryanum. J Biol Chem 253:3287–3291
Hong SJ, Lee CG (2007) Evaluation of central metabolism based on a genomic database of Synechocystis PCC6803. Biotechnol Bioprocess Eng 12:165–173
Osanai T, Kanesaki Y, Nakano T, Takahashi H, Asayama M, Shirai M, Kanehisa M, Suzuki I, Murata N, Tanaka K (2005) Positive regulation of sugar catabolic pathways in the cyanobacterium Synechocystis sp. PCC 6803 by the Group 2 factor SigE. J Biol Chem 35:30653–30659
Fuchs B, Suttner P, Sterner S, Wastlhuber R, Loos E (1994) Disproportionating transglycosylase (d-enzyme) in green algae and cyanobacteria. Partial purification and characterization. Zeitschrift für Naturforschung 49:163–170
Reyes-Sosa FM, Molina-Heredia FP, De la Rosa MA (2010) A novel α-amylase from the cyanobacterium Nostoc sp. PCC 7119. Appl Microbiol Biotechnol 86:131–141
Gupta V, Natarajan C, Kumar K, Prasanna R (2011) Identification and characterization of endoglucanases for fungicidal activity in Anabaena laxa (Cyanobacteria). J Appl Phycol 23:73–81
Wood T (1885) The pentose phosphate pathway. Academic Press Inc, Orlando
Pearce J, Carr NG (1969) The incorporation and metabolism of glucose by Anabaena variabilis. J Gen Microbiol 54:451–462
Pelroy RA, Basshan JA (1972) Photosynthetic and dark carbon metabolism in unicellular blue–green algae. Archiv für Mikrobiologie 86:25–38
van den Hoek C, Mann DG, Jahns HM (1995) Algae—an introduction to phycology. Press Sindicate of the University of Cambridge, New York
Li XF, Xu QY (2007) Large-scale biodiesel production from microalga Chlorella protothecoides through heterotrophic cultivation in bioreactors. Biotechnol Bioeng 98:764–771
Lu Y, Zhai Y, Liu M, Wu Q (2010) Biodiesel production from algal oil using cassava (Manihot esculenta Crantz) as feedstock. J Appl Phycol 22:573–578
Acknowledgments
Funding for this research was provided by São Paulo Research Foundation (FAPESP, Brazil).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Francisco, É.C., Franco, T.T., Wagner, R. et al. Assessment of different carbohydrates as exogenous carbon source in cultivation of cyanobacteria. Bioprocess Biosyst Eng 37, 1497–1505 (2014). https://doi.org/10.1007/s00449-013-1121-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-013-1121-1