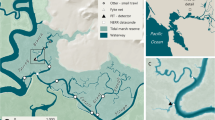
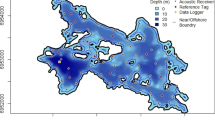
Abstract
Measuring an organism’s movement and habitat use is highly dependent on the spatial and temporal scale of the study, with most studies measuring distributions once a day or at less frequent intervals. Yet, to fully understand the rates of intra- and interspecific encounters among individuals, observations at finer spatial and temporal scales might be necessary. We used passive integrated transponder tags and antenna arrays to continuously monitor habitat use and vagility of three stream minnows; southern redbelly dace Chrosomus erythrogaster, central stoneroller Campostoma anomalum, and creek chub Semotilus atromaculatus, among and within pools of an intermittent stream. Most fish remained in the pool where they were caught and released, or returned after emigrating from the pool. Despite largely remaining within the release pool, distribution among four microhabitats differed significantly over six, 4-h time periods for all three species. Vagility, the summed distance moved among antennas, differed significantly among species. Individual vagility (m day−1) increased significantly with body length for stoneroller and chub, but not dace. Some individuals moved as much as 110 m day−1 within the pool, showcasing extensive movement at fine scales. Finally, we found no evidence that feeding activity changed as a result of differential habitat use over a 24-h period. Our findings indicate considerable variation in habitat use and movement occurs among species over a 24-h period. This suggests ecologists can broaden the interpretation of processes influencing community structure (e.g., resource partitioning, avoidance of predators) by quantifying species distributions across a range of spatial and temporal scales.






Similar content being viewed by others
References
Ahlgren MO (1990) Nutritional significance of facultative detritivory to the juvenile white sucker (Catostomus commersonii). Can J Fish Aquat Sci 47:49–54. https://doi.org/10.1139/f90-005
Albanese B, Angermeier PL, Dorai-Raj S (2004) Ecological correlates of fish movement in a network of Virginia streams. Can J Fish Aquat Sci 61:857–869. https://doi.org/10.1139/f04-096
Apps CD, McLellan BN, Kinley TA, Flaa JP (2001) Scale-dependent habitat selection by Mountain Caribou, Columbia Mountains, British Columbia. J Wildl Manag 65:65–77. https://doi.org/10.2307/3803278
Baker JA, Ross ST (1981) Spatial and temporal resource utilization by southeastern cyprinids. Copeia 1981:178–189. https://doi.org/10.2307/1444052
Bertrand KN, Gido KB (2007) Effects of the herbivorous minnow, southern redbelly dace (Phoxinus erythrogaster), on stream productivity and ecosystem structure. Oecologia 151:69–81. https://doi.org/10.1007/s00442-006-0569-y
Brown JS (1988) Patch use as an indicator of habitat preference, predation risk, and competition. Behav Ecol Sociobiol 22:37–47. https://doi.org/10.1007/BF00395696
Brown JS, Kotler BP (2004) Hazardous duty pay and the foraging cost of predation. Ecol Lett 7:999–1014. https://doi.org/10.1111/j.1461-0248.2004.00661.x
Brown JS, Kotler BP, Smith RJ, Wirtz WO II (1988) The effects of owl predation on the foraging of heteromyid rodents. Oecologia 76:408–415. https://doi.org/10.1007/BF00377036
Clough S, Ladle M (1997) Diel migration and site fidelity in a stream-dwelling cyprinid, Leuciscus leuciscus. J Fish Biol 50:1117–1119. https://doi.org/10.1111/j.1095-8649.1997.tb01635.x
Eberle ME (2014) Creek Chub, Semotilus atromaculatus Mitchill 1818. In: Kansas Fishes Committee (ed) Kansas fishes committee. University Press of Kansas, Lawrence, pp 246–248
Evans-White MA, Dodds WK, Whiles MR (2003) Ecosystem significance of crayfishes and stonerollers in a prairie stream: functional differences between co-occurring omnivores. J N Am Benthol Soc 22:423–441. https://doi.org/10.2307/1468272
Fausch KD, Nakano S, Ishigaki K (1994) Distribution of two congeneric charrs in streams of Hokkaido Island, Japan: considering multiple factors across scales. Oecologia 100:1–12. https://doi.org/10.1007/BF00317124
Fausch KD, Toorgersen CE, Baxter CV, Li HW (2002) Landscapes to riverscapes: bridging the gap between research and conservation of stream fishes. Bioscience 52:483–498. https://doi.org/10.1641/0006-3568(2002)052[0483:LTRBTG]2.0.CO;2
Felley JD, Hill LG (1983) Multivariate assessment of environmental preferences of cyprinid fishes of the Illinois River, Oklahoma. Am Midl Nat 109:209–221. https://doi.org/10.2307/2425400
Fowler JF, Taber CA (1985) Food habits and feeding periodicity in two sympatric stonerollers (Cyprinidae). Am Midl Nat 113:217–224. https://doi.org/10.2307/2425567
Fox J, Weisberg S (2011) An {R} companion to applied regression, 2nd edn. SAGE Publications Inc., Thousand Oaks, CA
Franssen NR, Gido KB, Guy CS, Tripe JA, Shrank SJ, Strakosh TR, Bertrand KN, Franssen CM, Pitts KL, Paukert CP (2006) Effects of floods on fish assemblages in an intermittent prairie stream. Freshw Biol 51:2072–2086. https://doi.org/10.1111/j.1365-2427.2006.01640.x
Gerking SD (1959) The restricted movement of fish populations. Biol Rev 34:221–242. https://doi.org/10.1111/j.1469-185X.1959.tb01289.x
Gorman OT (1988) The dynamics of habitat use in a guild of Ozark minnows. Ecol Monogr 58:1–18. https://doi.org/10.2307/1942631
Gowan C, Young MK, Fausch KD, Riley SC (1994) Restricted movement in resident stream salmonids: a paradigm lost? Can J Fish Aquat Sci 51:2626–2637. https://doi.org/10.1139/f94-262
Greenberg LA, Giller PS (2000) The potential of flat-bed passive integrated transponder antennae for studying habitat use by stream fishes. Ecol Freshw Fish 9:74–80. https://doi.org/10.1034/j.1600-0633.2000.90108.x
Hayes JW, Hughes NF, Kelly LH (2007) Process-based modeling of invertebrate drift transport, net energy intake and reach carrying capacity for drift-feeding salmonids. Ecol Modell 207:171–188. https://doi.org/10.11016/j.ecolmodel.2007.04.032
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Stat 6:65–70
Horton TB, Guy CS, Pontius JS (2004) Influence of time interval on estimations of movement and habitat use. N Am J Fish Manag 24:690–696. https://doi.org/10.1577/M02-069.1
Hughes JJ, Ward D, Perrin MR (1994) Predation risk and competition affect habitat selection and activity of Namib Desert Gerbils. Ecology 75:1397–1405. https://doi.org/10.2307/1937463
Kotler BP (1997) Patch use by gerbils in a risky environment: manipulating food and safety to test four models. Oikos 78:274–282. https://doi.org/10.2307/3546294
Logue JB, Mouquet N, Peter H, Hillebrand H, The Metacommunity Working Group (2011) Empirical approaches to metacommunities: a review and comparison with theory. TREE 26:482–491. https://doi.org/10.1016/j.tree.2011.04.009
MacArthur RH (1958) Population ecology of some warblers of northeastern coniferous forests. Ecology 39:599–619. https://doi.org/10.2307/1931600
Martin EC, Whitney JE, Gido KB, Hase KJ (2013) Habitat associations of stream fishes in protected tallgrass prairie streams. Am Mid Nat 170:39–51. https://doi.org/10.1674/0003-0031-170.1.39
Matthews WJ (1998) Patterns in freshwater fish ecology. Chapman and Hall, New York
Matthews WJ, Hill LG (1980) Habitat partitioning in the fish community of a southwestern river. Southwest Nat 25:51–66. https://doi.org/10.2307/3671211
Nakano S (1995) Competitive interactions for foraging microhabitats in a size-structured interspecific dominance hierarchy of two sympatric stream salmonids in a natural habitat. Can J Zool 73:1845–1854. https://doi.org/10.1139/z95-217
Nakano S, Furukawa-Tanaka T (1994) Intra- and interspecific dominance hierarchies and variation in foraging tactics of two species of stream dwelling chars. Ecol Res 9:9–20. https://doi.org/10.1007/BF02347237
Pennock CA, Frenette BD, Waters MJ, Gido KB (2016) Survival of and tag retention in Southern Redbelly Dace injected with two sizes of PIT tags. N Am J Fish Manag 36:1386–1394. https://doi.org/10.1080/02755947.2016.1227403
Petty JT, Grossman GD (2010) Giving-up densities and ideal pre-emptive patch use in a predatory benthic stream fish. Freshw Biol 55:780–793. https://doi.org/10.1111/j.1365-2427.2009.02321.x
Phillips GL (1969) Diet of minnow Chrosomus erythrogaster (Cyprinidae) in a Minnesota stream. Am Midl Nat 82:99–109. https://doi.org/10.2307/2423820
Pinaud D, Weimerskirch H (2005) Scale-dependent habitat use in a long-ranging central place predator. J Anim Ecol 74:852–863. https://doi.org/10.1111/j.1365-2656.2005.00984.x
Power ME (1987) Predator avoidance by grazing fishes in temperate and tropical streams: importance of stream depth and prey size. In: Kerfoot WC, Sih A (eds) Predation: direct and indirect impacts in aquatic communities. Univ. Press of New England, Dartmouth, pp 333–351
Power ME, Stewart AJ, Matthews WJ (1988) Grazer control of algae in an Ozark mountain stream: effects of short-term exclusion. Ecology 69:1894–1898. https://doi.org/10.2307/194116
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed Nov 2017
Radinger J, Wolter C (2014) Patterns and predictors of fish dispersal in rivers. Fish Fish 15:456–473. https://doi.org/10.1111/faf.12028
Rodriguez MA (2002) Restricted movement in stream fish: the paradigm is incomplete, not lost. Ecology 83:1–13. https://doi.org/10.1890/0012-9658(2002)083[0001:RMISFT]2.0.CO;2
Ross ST (1986) Resource partitioning in fish assemblages: a review of field studies. Copeia 1986:352–388. https://doi.org/10.2307/1444996
Samu F, Szirányi A, Kiss B (2003) Foraging in agricultural fields: local ‘sit-and-move’ strategy scales up to risk-averse habitat use in a wolf spider. Anim Behav 66:939–947. https://doi.org/10.1006/anbe.2003.2265
Schlosser IJ (1987) The role of predation in age- and size-related habitat use by stream fishes. Ecology 68:651–659. https://doi.org/10.2307/1938470
Schlosser IJ (1991) Stream fish ecology: a landscape perspective. Bioscience 41:704–712. https://doi.org/10.2307/1311765
Schlosser IJ, Angermeier PL (1995) Spatial variation in demographic processes of lotic fishes: conceptual models, empirical evidence, and implications for conservation. Am Fish Symp No 17:392–401
Schmider E, Ziegler M, Danay E, Beyer L, Markus B (2010) Is it really robust? Reinvestigating the robustness of ANOVA against violations of the normal distribution assumption. Methodology 6:147–151. https://doi.org/10.1027/1614-2241/a000016
Senko J, Koch V, Megill WM, Carthy RR, Templeton RP, Nichols WJ (2010) Fine scale daily movements and habitat use of East Pacific green turtles at a shallow coastal lagoon in Baja California Sur, Mexico. J Exp Mar Biol Ecol 391:92–100. https://doi.org/10.1016/j.jembe.2010.06.017
Settles WH, Hoyt RD (1976) Age structure, growth patterns, and food habits of the southern redbelly dace Chrosomus erythrogaster in Kentucky. Trans Ky Acad Sci 37:1–10
Skalski GT, Gilliam JF (2000) Modeling diffusive spread in a heterogeneous population: a movement study with stream fish. Ecology 81:1685–1700. https://doi.org/10.1890/0012-9658(2000)081[1685:MDSIAH]2.0.CO;2
Usio N, Nakano S (1998) Influences of microhabitat use and foraging mode similarities on intra- and interspecific aggressive interactions in a size-structured stream fish assemblage. Ichthyol Res 45:19–28. https://doi.org/10.1007/BF02678571
Vernes K (2003) Fine-scale habitat preferences and habitat partitioning by three mycophagous mammals in tropical wet sclerophyll forest, north-eastern Australia. Austral Ecol 28:471–479. https://doi.org/10.1046/j.1442-9993.2003.01303.x
Waters TF (1962) Diurnal periodicity in the drift of stream invertebrates. Ecology 43:316–320. https://doi.org/10.2307/1931988
Weber N, Bouwes N, Jordan CE (2014) Estimation of salmonid habitat growth potential through the measurements of invertebrate food abundance and temperature. Can J Fish Aquat Sci 71:1158–1170. https://doi.org/10.1139/cjfas-2013-0390
Wells WG, Johnson TC, Gebhard AE, Paine RTR, Hix LA, Ferrell HN, Engle AN, Perkin JS (2017) March of the sculpin: measuring and predicting short-term movement of banded sculpin Cottus carolinae. Ecol Freshw Fish 26:280–291. https://doi.org/10.1111/eff.12274
Wentworth CK (1922) A scale of grade and class terms for clastic sediments. J Geol 30:377–392. https://doi.org/10.1086/622910
Winemiller KO, Rose KA (1992) Patterns of life-history diversification in North American fishes: implications for population regulation. Can J Fish Aquat Sci 49:2196–2218. https://doi.org/10.1139/f92-242
Winemiller KO, Flecker AS, Hoeinghaus DJ (2010) Patch dynamics and environmental heterogeneity in lotic ecosystems. J N Am Benthol Soc 29:84–99. https://doi.org/10.1899/08-048.1
Wolf JB, Trillmich F (2007) Beyond habitat requirements: individual fine-scale site fidelity in a colony of the Galapagos sea lion (Zalophus wollebaeki) creates conditions for social structuring. Oecologia 152:553–567. https://doi.org/10.1007/s00442-007-0665-7
Young MK (1996) Summer movements and habitat use by Colorado River cutthroat trout (Oncorhynchus clarki pleuriticus) in small, montane streams. Can J Fish Aquat Sci 53:1403–1408. https://doi.org/10.1139/f96-054
Zaret TM, Rand AS (1971) Competition in tropical stream fishes: support for the competitive exclusion principle. Ecology 52:336–342. https://doi.org/10.2307/1934593
Acknowledgements
We would like to thank G. Hopper, B. Frenette, L. Bruckerhoff, A. Earl, J. Grill, and T. Fanshier for assistance in the field. This manuscript was greatly improved by comments and discussions with J. Perkin, J. Wellemeyer, J. Whitney, J. Trexler, and two anonymous reviewers. This study was approved by the Kansas State University Institutional Animal Care and Use Committee (protocol #3676). The Kansas Department of Wildlife, Parks and Tourism and College of Arts and Sciences, Kansas State University provided financial support for this project.
Author information
Authors and Affiliations
Contributions
CAP led writing the manuscript. REW, CNC, and KBG conceived and designed the first study. CAP and KBG conceived and designed the follow-up study. CAP, REW, CNC, SCH, and KBG conducted fieldwork. CAP, REW, and SCH analyzed the data. All authors contributed to the writing of the manuscript.
Corresponding author
Additional information
Communicated by Joel Trexler.
Rights and permissions
About this article
Cite this article
Pennock, C.A., Nathan Cathcart, C., Hedden, S.C. et al. Fine-scale movement and habitat use of a prairie stream fish assemblage. Oecologia 186, 831–842 (2018). https://doi.org/10.1007/s00442-018-4073-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-018-4073-y