Abstract
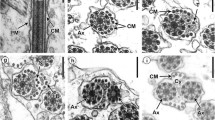
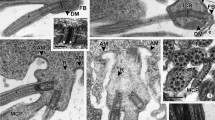
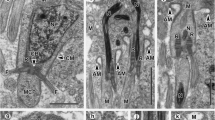
Spermiogenesis and the spermatozoon ultrastructure of the cestode Nippotaenia mogurndae Yamaguti et Myiata, 1940 (Nippotaeniidea), a parasite of Perccottus glenii Dubowski, 1877 (Perciformes: Odontobutidae), have been investigated by means of transmission electron microscopy, cytochemical staining with periodic acid-thiosemicarbazide-silver proteinate (PA-TSC-SP) for glycogen, and electron tomography. The process of spermatozoon formation is characterised by the presence of (1) two centrioles without typical striated rootlets, (2) a single intercentriolar body, (3) a flagellar rotation (free flagellum plus flagellar bud), and (4) a complete proximodistal fusion. The mature spermatozoon of N. mogurndae contains a single helicoidal crested body, one axoneme of the 9 + “1” trepaxonematan structure, parallel cortical microtubules arranged in a ring in the anterior region of the cell, and a spiraled nucleus encircling the axoneme. Intracellular components are situated in a moderately electron-dense cytoplasm, containing glycogen in the principal regions (II, III, IV) of the spermatozoon. Application of electron tomography has revealed a helicoidal nature of the central electron-dense core in the central cylinder of the axoneme in parasitic cestodes for the first time. The patterns of spermiogenesis and spermatozoon ultrastructure resemble most closely those in mesocestoidids and may reflect the relationships between Nippotaeniidea and Mesocestoididae.







Similar content being viewed by others
References
Azzouz-Draoui N, Mokhtar-Maamouri F (1986/88) Ultrastructure comparée de la spermiogenèse et du spermatozoïde de Echinobothrium affine Diesing, 1863 et E. harfordi Mac Vicar, 1976 (Cestoda, Diphyllidea). Bull Soc Sci Nat Tuniesie 18:9–12
Bâ CT, Marchand B (1995) Spermiogenesis, spermatozoa and phyletic affinities in the Cestoda. In: Jamieson, BGM, Ausie J, Justine JL (eds) Advances in spermatozoal phylogeny and taxonomy. Mém Mus natn Hist nat 166:87–95
Bray RA (1994) Order Nippotaeniidea Yamaguti, 1939. In: Khalil LF, Jones A, Bray RA (eds) Keys to the cestode parasites of vertebrates. CAB International, Wallingford, pp 253–255
Bruňanská M (2009) Spermatological characters of the caryophyllidean cestode Khawia sinensis Hsü, 1935, a carp parasite. Parasitol Res 105:1603–1610
Bruňanská M (2010) Recent insights into spermatozoa development and ultrastructure in the Eucestoda. In: Lejeune T, Delvaux P (eds) Human spermatozoa: maturation, capacitation and abnormalities. Nova Science Publishers, Inc., New York, pp 327–354
Bruňanská M, Kostič B (2012) Revisiting caryophyllidean type of spermiogenesis in the Eucestoda based on spermatozoon differentiation and ultrastructure of Caryophyllaeus laticeps (Pallas, 1781). Parasitol Res 110:141–149
Bruňanská M, Poddubnaya LG (2006) Spermiogenesis in the caryophyllidean cestode Khawia armeniaca (Cholodkovski, 1915). Parasitol Res 99:449–454
Bruňanská M, Nebesářová J, Scholz T (2003) Spermiogenesis in the proteocephalidean cestode Proteocephalus torulosus (Batsch, 1786). Parasitol Res 90:318–324
Bruňanská M, Scholz T, Nebesářová J (2004) Reinvestigation of spermiogenesis in the proteocephalidean cestode Proteocephalus longicollis (Zeder, 1800). J Parasitol 90:23–29
Bruňanská M, Scholz T, Ibraheem MH (2005) Spermiogenesis in the cestode Corallobothrium solidum Fritsch, 1886 (Proteocephalidea: Corallobothriinae). Acta Zool 86:55–61
Davydov VG, Korneva JV (2000) Differentiation and structure of a uterus for Nippotaenia mogurndae Yamaguti et Myiata, 1940 (Cestoda: Nippotaeniidea). Helminthologia 37:77–82
Eira C, Miquel J, Vingada J, Torres J (2006) Spermiogenesis and spermatozoon ultrastructure of the cestode Mosgovoyia ctenoides (Cyclophyllidea: Anoplocephalidae), an intestinal parasite of Oryctolagus cuniculus (Lagomorpha: Leporidae). J Parasitol 92:708–718
Euzet L, Świderski Z, Mokhtar-Maamouri F (1981) Ultrastructure comparée du spermatozoïde des cestodes. Relations avec la phylogénèse. Ann Parasitol Hum Comp 56:247–259
Hoberg EP, Mariaux J, Brooks DR (2001) Phylogeny among the orders of the Eucestoda (Cercomeromorphae): integrating morphology, molecules and total evidence. In: Littlewood DTJ, Bray RA (eds) Interrelationships of the Platyhelminthes. Taylor and Francis, London, pp 112–126
Justine JL (1998) Spermatozoa as phylogenetic characters for the Eucestoda. J Parasitol 84:385–408
Justine JL (2001) Spermatozoa as phylogenetic characters for the Platyhelminthes. In: Littlewood DTJ, Bray RA (eds) Interrelationships of the Platyhelminthes. Taylor and Francis, London, pp 231–238
Justine J-L (2003) Ultrastructure des spermatozoïdes et phylogénie des Neodermata. In: Combes C, Jourdane J (eds) Taxonomy, ecology and evolution of metazoan parasites. PUP, Perpignan, pp 359–380
Korneva JV (2002) Fine structure of reproductive system in Nippotaenia mogurndae (Cestoda: Nippotaeniidea). Zool Zhurnal 81:266–275
Košuthová L, Letková V, Koščo J, Košuth P (2004) First record of Nippotaenia mogurndae Yamaguti et Myiata, 1940 (Cestoda: Nippotaeniidea), a parasite of Perccottus glenii Dubowski, 1877, from Europe. Helminthologia 41:55–57
Kremer JR, Mastronarde DN, McIntosh JR (1996) Computer visualization of three-dimensional image data using IMOD. J Struct Biol 116:71–76
Kvach Y, Drobiniak O, Kutsokon Y, Hoch I (2013) The parasites of the invasive Chinese sleeper Perccottus glenii (Fam. Odontobutidae), with the first report of Nippotaenia mogurndae in Ukraine. Knowl Manag Aquat Ecosyst 409:05
Levron C, Miquel J, Oros M, Scholz T (2010) Spermatozoa of tapeworms (Platyhelminthes, Eucestoda): advances in ultrastructural and phylogenetic studies. Biol Rev 85:523–543
Li H-Y, Brennan JP, Halton DW (2003) Spermatogenesis, spermiogenesis and spermatozoon in the cestode (Moniesia expansa) (Cyclophyllidea: Anoplocephalidae). Acta Zool Sin 49:370–379
Marigo AM, Levron C, Bâ CT, Miquel J (2012a) Ultrastructural study of spermiogenesis and the spermatozoon of the proteocephalidean cestode Barsonella lafoni de Chambrier et al., 2009, a parasite of the catfish Clarias gariepinus (Burchell, 1822) (Siluriformes, Clariidae). Zool Anz 251:147–159
Marigo AM, Delgado E, Torres J, Bâ CT, Miquel J (2012b) Spermiogenesis and spermatozoon ultrastructure of the bothriocephalidean cestode Clestobothrium crassiceps (Rudolphi, 1819), a parasite of the teleost fish Merluccius merluccius (Gadiformes: Merlucciidae). Parasitol Res 110:19–30
Miquel J, Marchand B (1998) Ultrastructure of the spermatozoon of the bank vole tapeworm, Paranoplocephala omphalodes (Cestoda, Cyclophyllidea, Anoplocephalidae). Parasitol Res 84:239–245
Miquel J, Feliu C, Marchand B (1999) Ultrastructure of spermiogenesis and the spermatozoon of Mesocestoides lineatus (Cestoda, Mesocestoididae). Int J Parasitol 29:499–510
Miquel J, Świderski Z, Marchand B (2005a) Spermatological characters in the Dipylidiidae Stiles, 1896 (Cestoda, Cyclophyllidea). Acta Parasitol 50:65–73
Miquel J, Świderski Z, Młocicki D, Eira C, Marchand B (2005b) Spermatogenesis in the anoplocephalid cestode Gallegoides arfaai (Mobedi et Ghadirian, 1977) Tenora et Mas-Coma, 1978. Acta Parasitol 50:132–144
Miquel J, Eira C, Świderski Z, Conn DB (2007) Mesocestoides lineatus (Goeze, 1782) (Mesocestoididae): new data on sperm ultrastructure. J Parasitol 93:545–552
Miquel J, Świderski Z, Mackiewicz JS, Ibraheem MH (2008) Ultrastructure of spermiogenesis in the caryophyllidean cestode Wenyonia virilis Woodland, 1923, with re-assessment of flagellar rotation in Glaridacris catostomi Cooper, 1920. Acta Parasitol 53:19–29
Miquel J, Świderski Z, Foronda P, Torres J, Feliu C (2009) Ultrastructure of spermatogenesis of Taenia taeniaeformis (Batsch, 1786) (Cestoda, Cyclophyllidea, Taeniidae) and comparison of spermatological characters in the family Taeniidae Ludwig, 1886. Acta Parasitol 54:230–243
Mokhtar-Maamouri F (1979) Étude en microscopie électronique de la spermatogénèse et du spermatozoïde de Phyllobothrium gracile Wedl, 1855 (Cestoda, Tetraphyllidea, Phyllobothriidae). Z Parasitenkd 59:245–258
Ndiaye PI, Quilichini Y, Bâ A, Bâ CT, Marchand B (2012) Ultrastructural study of the male gamete of Glossobothrium sp, (Cestoda: Bothriocephalidea: Triaenophoridae) a parasite of Schedophilus velami (Perciformes: Centrolophidae) in Senegal. Tissue Cell 44:296–300
Reshetnikov AN (2010) The current range of Amur sleeper Perccottus glenii Dubowski, 1877 (Odontobutidae, Pisces) in Euroasia. Russ J Biol Invasions 1:119–126
Sène A, Bâ CT, Marchand B (1997) Ultrastructure of spermiogenesis and the spermatozoon of Nomimoscolex sp. (Cestoda, Proteocephalidea) intestinal parasite of Clarotes laticeps (fish, teleost) in Senegal. J Submicrosc Cytol Pathol 29:1–6
Silveira M (1969) Ultrastructural studies on a “nine plus one” flagellum 1. J Ultrastruct Res 26:274–288
Stoitsova SR, Georgiev BB, Dacheva RB (1995) Ultrastructure of spermiogenesis and the mature spermatozoon of Tetrabothrius erostris Loennberg, 1896 (Cestoda, Tetrabothriidae). Int J Parasitol 25:1427–1436
Świderski Z (1986) Three types of spermiogenesis in cestodes. In: Imura T, Maruse S, Suzuki T (eds) Proceedings of 11th International Congress on Electron Microscopy, Kyoto: 2959–2960
Thiéry J-P (1967) Mise en évidence des polysaccharides sur coupes fines en microscopie électronique. J Microsc 6:987–1018
Waeschenbach A, Webster BL, Littlewood DTJ (2012) Adding resolution to ordinal level relationships of tapeworms (Platyhelminthes: Cestoda) with large fragments of mtDNA. Mol Phylogen Evol 63:834–847
Yoneva A, Georgieva K, Mizinska Y, Georgiev BB, Stoitsova SR (2006) Ultrastructure of spermiogenesis and mature spermatozoon of Skrjabinoporus merops (Cyclophyllidea, Metadilepididae). Acta Parasitol 51:200–208
Yoneva A, Levron C, Oros M, Orosová M, Scholz T (2011) Ultrastructure of spermiogenesis and mature spermatozoon of Breviscolex orientalis (Cestoda: Caryophyllidea). Parasitol Res 108:997–1005
Acknowledgments
We would like to thank Céline Levron (France) and Mikuláš Oros (Slovakia) for invaluable technical help with collecting parasites. We are grateful to the staff of the Laboratory of Electron Microscopy, Institute of Parasitology AS CR, České Budějovice, Czech Republic, for technical assistance. Financial support was provided by the Grant Agency of the Slovak Republic VEGA (project no. 2/0047/11) and the Technology Agency of the Czech Republic (project no. TE01020118 (programme Competence Centre: Electron Microscopy)). The work was realised within a frame of Centre of Excellence for Parasitology (Code ITMS 26220120022) based on the support of the Operational Programme “Research & Development” funded from the European Regional Development Fund (rate 0.1).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
The movie shows a 3D colored model and Z walkthrough the axoneme section volume with detail of of the central electrondense core in red colour (correspond to Figures 6 and 7). Single axis tomogram was reconstructed from ∼79 nm thick section and divided to 231 slices and subsequently was applied binning by factor of 2. (MOV 24241 kb)
Rights and permissions
About this article
Cite this article
Bruňanská, M., Bílý, T. & Nebesářová, J. Nippotaenia mogurndae Yamaguti et Myiata, 1940 (Cestoda, Nippotaeniidea): first data on spermiogenesis and sperm ultrastructure. Parasitol Res 114, 1443–1453 (2015). https://doi.org/10.1007/s00436-015-4327-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4327-0