Abstract
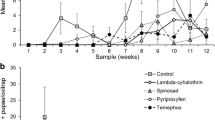
The efficacy of an ultralow volume formulation (ULV) and fumigant canister, containing both permethrin and pyriproxyfen, was compared with that of standard permethrin applications in a field assay conducted in Banda del Río Salí, Tucumán (north-western Argentina). Five treatment areas were established: first area was sprayed with a ULV formulation of 10 % permethrin, a second area was treated using a fumigant canister containing 10 % permethrin and 3 % pyriproxyfen, the third area was sprayed with a ULV formulation of 10 % permethrin and 3 % pyriproxyfen, the fourth area with ULV formulation of 10 % permethrin using a portable aerosol generator and the fifth area was a left untreated area. Immature and adult Aedes aegypti individuals placed in containers and sentinel cages were positioned within the treated and control areas. The effects of treatment and time on larval, pupal and adult survival were tested. We also investigated the effects of treatment and time on the numbers of larval and pupal deaths, on the proportion of larvae that metamorphosed into pupae and adults, and on the proportion of dead adults. Larval A. aegypti survivorship in 250 mL containers revealed a significant treatment effect and significant treatment × time interaction 2 and 24 h after the application of the ULV treatment with 10 % permethrin using the portable aerosol generator. The number of dead larvae in 20 L containers differed significantly by treatment and by time. ULV treatment with 10 % permethrin and 10 % permethrin plus 3 % pyriproxyfen using the cold fogger truck mount ULV resulted in the greatest numbers of dead larvae; most larvae died 2 weeks after application. Adult A. aegypti mortality in all treatments did not differ significantly 2 and 24 h after application. In addition, we found no significant differences in adult mortality between cages exposed at 3 m and those at 6 m from the ULV application line. However, there was a significant difference in adult mortality between the 10 % permethrin treatment applied with cold fogger truck mount ULV and that applied using the portable aerosol generator (P < 0.001). The larval index known as Breteau index (BI) was higher before treatment than after treatment in different areas. After the treatments, the lowest value of BI was observed in the area treated with the fumigant canister formulation, and a long-lasting effect was observed with the formulation of 10 % permethrin and 3 % pyriproxyfen.









Similar content being viewed by others

References
Aviles G, Rangeon G, Vorndam V, Briones A, Baroni P, Enria D, Sabattini M (1999) Dengue reemergence in Argentina. Emerg Infect Dis 5:1–4
Bang YH, Pant CP (1972) A field trial of Abate larvicide for the control of Aedes aegypti in Bangkok, Thailand. Bull World Health Organ 46:416–425
Boletín Epidemiológico (2011) Dirección Nacional de Epidemiología. Departamento de Vigilancia Epidemiológica. Ministerio de Salud de la Nación (Argentina). Dengue. Pp. 25–26
Boffi R (2002) Erradicación y reinfestación por el Aedes aegypti en la Republica Argentina Master dissertation. Universidad Nacional General San Martin, Argentina
Braga Ima A, Lima JBP, Soares SS, Valle D (2004) Aedes aegypti resistance to temephos during 2001 in several municipalities in the states of Rio de Janeiro, Sergipe and Alagoass, Brazil. Mem Inst Oswaldo Cruz 99:199–203
Brooks GD, Shoof HF, Smith EA (1966) Evaluation of five formulations of Abate against Aedes aegypti, Savannah, Georgia, 1965. Mosq News 26:580–582
Carbajo AE, Schweigmann N, Curto SI, Garin A, Bejaran R (2001) Dengue transmission risk maps of Argentina. Trop Med Int Health 6:170–183
Chadee DD (1984) An evaluation of temephos in water drums in Trinidad, WI. Mosq News 44:51–53
Chadee DD (1985) An evaluation of malathion ULV spraying against caged and natural populations of Aedes aegypti in Trinidad, West Indies. Cahiers ORSTOM Series Entomol and Med Parasitol 23:71–74
Chavasse DC, Yap HH (1997) Chemical methods for the control of vectors and pests of public health importance Document WHO/CTD/WHOPES/97.2. World Health Organization, Geneva
Dantur Juri MJ (2009) Brote de dengue en el NOA: situación epidemiológica en la provincia de Tucumán. Bol Soc Entomol Arg 20(1–2):6–7
Donalisio MRC, Leite OF, Mayo RC, Alves M, de Souza A, Rangel O, Andrade VR, Oliveira SS, Matias VLM (2002) Use of temephos for control of field population of Aedes aegypti in Americana São Paulo, Brazil. Dengue Bull 26:173–177
Garelli FM, Espinosa MO, Weinberg D, Trinelli MA, Gürtler RE (2011) Water Use Practices Limit the Effectiveness of a Temephos-Based Aedes aegypti Larval Control Program in Northern Argentina. PLoS Negl Trop Dis 5(3):e991
Goma LK (1964) Laboratory observations on the oviposition habits of Aedes aegypti (Linnaeus). Ann Trop Med Parasitol 58:347–349
Gonzalez Audino P, Licastro S, Zerba E (1999) Thermal behaviour and biological activity of pyrethroids in smoke-generating formulations. Pestic Sci 55:1187–1193
Gratz N, Knudsen B (1996) The rise and spread of dengue, dengue hemorrhagic fever and its vectors. A Historical Review (up to 1995). CTD/FIL (DEN) 96.7, WHO
Harburguer LV, Seccacini E, Masuh H, González Audino P, Zerba E, Licastro S (2009) Thermal behaviour and biological activity against Aedes aegypti (Diptera: Culicidae) of permethrin and pyriproxyfen in a smoke-generating formulation. PestManag Sci 65:1208–1214
Itaya N (1987) Insect juvenile hormone analogue as an insect growth regulator Sumitomo Pyrethroid World 8:2–4
Koehler PG, Patterson RJ (1991) Incorporation of pyriproxyfen in a German cockroach management program. J Econ Entomol 84:917–921
Kourí GI, Pelegrino JL, Munster BM, Guzmán MG (2007) Sociedad, economía, inequidades y dengue. Rev Cubana Med Trop 59
Kroeger A, Nathan MB (2006) Dengue: setting the global research agenda. Lancet 368:2193–2195
Kyle JL, Harris E (2008) Global spread and persistence of dengue. Ann Rev Microbiol 62:71–92
Lee YW, Gregorio ER, Khadri MS, Seleena F (1996) Ultra low volume application of Bacillus thuringiensis ssp. israeliensis for the control of mosquitoes. J Am Mosq Cont Assoc 12:651–655
Lucia A, Harburguer L, Licastro S, Zerba E, Masuh H (2009) Efficacy of a new combined larvicidal–adulticidal ultralow volume formulation against Aedes aegypti (Diptera: Culicidae), vector of dengue. Parasitol Res 104:1101–1107
Macoris MLG, Andrighetti MTM, Takaku L, Glasser CM, Garbeloto VC, Bracco JC (2003) Resistance of Aedes aegypti from the state of S. Paulo, Brazil, to organophosphates insecticides. Mem Inst Oswaldo Cruz 98:703–708
Mani TR, Arunachalam N, Rajendran R, Satyanarayana K, Dash AP (2005) Efficacy of thermal fog application of deltacide, a synergized mixture of pyrethroids, against Aedes aegypti, the vector of dengue. Trop Med Internat Health 10:1298–1304
Masuh H, Arnstein de Licastro S, Lopez PA, Vega C, Zerba E (2003) Field evaluation of a smoke generating formulation containing beta- cypermethrin against the dengue vector in Argentina. J Am Mosq Cont Assoc 19:53–57
Masuh H, Seccacini E, Zerba E, Licastro SA (2008) Aedes aegypti (Diptera: Culicidae): monitoring of populations to improve control strategies in Argentina. Parasitol Res 103:167–170
Mulla MS, Darwazeh HA, Norland RL (1974) Insect growth regulators: evaluation procedures and activity against mosquitoes. J Econ Entomol 67:329–332
PAHO (1994) Dengue and dengue hemorrhagic fever in the Americas: Guidelines for prevention and control. Scientific Publication 548:28–29
PAHO (2011) Alerta epidemiológica: Actualización sobre la situación de Dengue en las Américas. Organización Panamericana de la Salud. 1–5 pp
Palomino M, Solari L, León W, Vega R, Vergaray M et al (2006) Evaluación del efecto residual del temephos en larvas de Aedes aegypti en Lima, Perú. Rev Peru Med Exp Salud Publica 23:158–162
Perich MJ, Tidwell MA, Williams DC, Sardelis MR, Pena CJ, Mandeville D et al (1990) Comparison of ground and aerial ultra-low volume applications of malathion against Aedes aegypti in Santo Domingo, Dominican Republic. J Am Mosq Cont Assoc 6:1–6
Perich MJ, Davila G, Turner A, Garcia A, Nelson M (2000) Behavior of resting Aedes aegypti (Culicidae: Diptera) and its relation to ultra-low volume adulticide efficacy in Panama City, Panama. J Med Entomol 37:541–546
Perich MJ, Sherman C, Burge R, Gill E, Quintana M, Wirtz RA (2001) Evaluation of the efficacy of lambda-cyhalothrin applied as ultralow volume and thermal fog for emergency control of Aedes aegypti in Honduras. J Am Mosq Control Assoc 17:221–224
Pontes RJS, Regazzi ACF, Lima JWO, Kerr-Pontes LRS (2005) Residual effect of commercial applications of larvicides temefos and Bacillus thuringiensis israelensis on Aedes aegypti larvae in recipients with water renewal. Rev Soc Bras Med Trop 38:316–321
Raudenbush S, Bryk T, Congdon R (2000) HLM 6 Hierarchical Linear and Nonlinear Modeling. Scientific Software International, Inc. (c). http://www.ssicentral.com. Accessed 17 October 2011
Reiter P, Nathan MB (2003) World Health Organization. Report of the WHO Informal Consultation on the evaluation and testing of insecticidas—Guía para la evaluación de la eficacia del rociado espacial de insecticidas para el control del vector del dengue. (WHO/CTD/WHOPES/IC/96.1). Geneva, Switzerland
Seccacini E, Lucia A, Zerba E, Licastro MH (2008) Aedes aegypti (L.) resistance to temephos in Argentina. J Am Mosq Control Assoc 24:608–609
Sistema Nacional de Vigilancia de la Salud (SNVS) (2012) Dengue. Ministerio de Salud de la Nación. http://www.msal.gov.ar/dengue/index.php. Accessed 12 March 2012
Tawatsin A, Thavara U, Chompoosri J, Bhakdeenuan P, Asavadachanukorn P (2007) Larvicidal efficacy of new formulations of temephos in non-woven sachets against larvae of Aedes aegypti (L.) (Diptera: Culicidae) in water-storage containers. Southeast Asian J Trop Med Public Health 38:641–645
Thavara U, Tawatsin A, Srithommarat R, Zaim M, Mulla MS (2005) Sequential release and residual activity of temephos applied as sand granules to water storage jars for the control of Aedes aegypti larvae (Diptera: Culicidae). J Vector Ecol 30:62–72
Tidwell MA, Williams DC, Gwinn GA, Pena CJ, Tedders SH, Gonzalvez GE, Mekuria Y (1994) Emergency control of Aedes aegypti in the Dominican Republic using the Scorpion TM ULV forced-air generator. J Am Mosq Control Assoc 10:403–406
WHO (World Health Organization) (1999) Bacillus thuringiensis. Environmental Health Criteria 217, Geneva
WHO (World Health Organization) (2005) Report of the Eighth WHOPES Working Group Meeting. Geneva. Guidelines for laboratory and field testing of mosquito larvicides (WHO/CDS/WHOPES/GCDPP/2005.13). Geneva, Switzerland
WHO (World Health Organization) (2007a) Pyriproxyfen in drinking water: use for vector control in drinking water sources and containers. Background document for development of WHO guidelines for drinking-water quality. Document WHO/SDE/WSH/07.01.9. Geneva, Switzerland, 13 pp
WHO (World Health Organization) (2007b) Pyriproxyfen in drinking water. Background document for development of WHO guidelines for drinking-water quality. Document WHO/SDE/WSH/07.01.10. Geneva, Switzerland, 13 pp
Yap HH, Chong ASC, Adanan R, Chong NL, Rohaizat B, Abdul Malik Y, Lim SY (1997) Performance of ULV formulations (Pestguard® 102/Vectobac® 12AS) against three mosquito species. J Am Mosq Cont Assoc 13:384–388
Zerba E (1995) Fumigant canisters and other novel insecticide formulations. Public Health Magazine (Bayer) 12:63–67
Zerba E, Melgar Suarez F, Licastro S, Wood EJ, Picollo MI, Malkenson N (1996) Un dispositivo fumigeno insecticida. Argentine Patent 248872
Acknowledgements
Authors are grateful to the technicians of the National Coordination of Vector Control of NOA region (National Ministry of Health) who participate during the field essay. This research received financial support from Chemotécnica S.A. (Argentina).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dantur Juri, M.J., Zaidenberg, M. & Santana, M. The efficacy of a combined larvicide–adulticide in ultralow volume and fumigant canister formulations in controlling the dengue vector Aedes aegypti (Diptera: Culicidae) in Northwest of Argentina. Parasitol Res 112, 1237–1246 (2013). https://doi.org/10.1007/s00436-012-3256-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-3256-4