Abstract
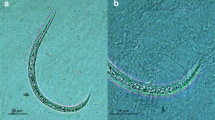
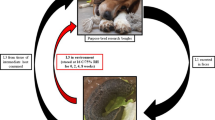
Biomphalaria glabrata snails were experimentally infected with Angiostrongylus vasorum first-stage larvae and divided into four groups of 30 snails. To assess the shedding of third-stage larvae (L3), the snails were maintained under different stimuli: group 1 60 W light bulb for 24 h, group 2 37 °C water bath for 24 h, group 3 room temperature (23–25 °C) for 24 h, Group 4 room temperature (23–25 °C) for up to 15 days. After 24 h, a total of 512 A. vasorum L3, alive and active, were released by snails from group 1, while 2,446 L3 were released from group 2 and five L3 from group 3. After 15 days, snails from group 4 released a total of 44 L3. To evaluate the infectivity of A. vasorum L3, two mongrel dogs were successfully infected with L3 released by snails from groups 1 and 2, confirming that the infection of dogs with A. vasorum L3 was possible, independently of ingestion of the mollusk intermediate host. The results shown in these experiments suggest that angiostrongylosis could be directly transmitted to the definitive hosts, with implications for the parasite′s life cycle.


Similar content being viewed by others
References
Ash LR (1970) Diagnostic morphology of the third-stage larvae of Angiostrongylus cantonensis, Angiostrongylus vasorum, Aelurostrongylus abstrusus and Anafilaroides rotratus (Nematoda: Metastrongyloidea). J Parasitol 56:249–253
Baillet CC (1866) Strongle des vaisseaus et du coeur du chein Strongylus vasorum. N Dict Paract Med Vet 8:587–588
Barçante JMP, Barçante TA, Dias SRC, Vieira LQ, Lima WS, Negrão-Corrêa D (2003) A method to obtain axenic Angiostrongylus vasorum first-stage larvae from dog feces. Parasitol Res 89:89–93
Bolt G, Monrad J, Frandsen F, Henrikesen P, Dietz HH (1993) The common frog (Rana temporaria) as a potential paratenic and intermediate host for Angiostrongylus vasorum. Parasitol Res 79:428–430
Bwangamoi O (1974) Renal lymphoid and pulmonary lesions in naturally acquired canine angiostrongylosis in Uganda. Bull Epizoot Dis Afr 22:55–68
Cheng TC, Alicata JE (1964) Possible role of water in the transmission of Angiostrongylus cantonensis (Nematoda: Metastrongylidae). J Parasitol 50:39–40
Conejo ME, Morera P (1988) Influencia de la edad de los veronicelideos en la infeccion com Angiostrongylus costaricensis. Rev Biol Trop 36:519–526
Dodd K (1973) Angiostrongylus vasorum (Baillet, 1866) infestation in a greyhound kennels. Vet Rec 92:195–197
Edwards NJ (1995) ECG manual for the veterinary technician. Saunders, Philadelphia
Guilhon J, Afghahi A (1969) Évolution larvaire d′Angiostrongylus vasorum (Baillet, 1866) dans l′organisme de diverses espéces de mollusques terrestres. C R Acad Sci 268:434–436
Guilhon J, Cens B (1973) Angiostrongylus vasorum (Baillet, 1866): Étude biologique et morfologique. Ann Parasitol Hum Comp 48:567–596
Heyneman D, Lym BL (1967) Angiostrongylus cantonensis: proof of direct transmission with its epidemiological implications. Science 158:1057–1058
Kamensky SN (1905) Sistematichescoe poloz henieradov Metastrongylus wots in Protostrongylus g.n. sredi drugikh Strongylidae (in Russian). Sb Tr Khardk Vet Inst 7:17–20
Lima WS, Costa HMA, Guimarães MP, Leite ACR (1985) Angiostrongylus vasorum (Baillet, 1866) Nematoda: Protostrongylidae em cães de Minas Gerais, Brasil. Mem Inst Oswaldo Cruz 80:233–235
Lima WS, Guimarães MP, Lemos IS (1994) Occurrence of Angiostrongylus vasorum in the lungs of Brazilian fox Dusicyon vetulus. J Helminthol 68:87
Morera P, Céspedes R (1971) Angiostrongylosis abdominal. Una nueva parasitosis humana. Acta Med Costar 14:159–173
Ribeiro VM, Lima WS (2001) Larval production of cats infected and re-infected with Aelurostrongylus abstrusus (Nematoda:Protostrongylidae). Rev Med Vet 152:815–820
Richards CS, Merritt JW (1967) Studies on Angiostrongylus cantonensis in molluscan intermediate host. J Parasitol 53:382–388
Richinitti LMZ, Fonseca NA, Graeff-Teixeira C (1999) The effect of temperature on mobility of Angiostrongylus costaricensis third-stage larvae. Inst Med Trop Sao Paulo 41:225–228
Rosen L, Ash LR, Wallace GD (1970) Life history of the canine lungworm Angiostrongylus vasorum (Baillet, 1866). Am J Vet Res 31:131–143
Ubelaker JE, Bullick GR, Caruso J (1980) Emergence of third-stage larvae of Angiostrongylus costaricensis Morera and Céspedes 1971 from Biomphalaria glabrata (Say). J Parasitol 66:856–857
Acknowledgements
The authors are grateful to Dra. Leda Quercia Vieira for useful discussions and English review. The authors wish to thank the technical staff of the Schistosomiasis Laboratory (GIDE-UFMG) for supplying the Biomphalaria snails used in the experiments. The authors also thank Edna Maia for technical advice. The experimentation protocols complied with the current ethics laws of the Colégio Brasileiro de Experimentação Animal.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barçante, T.A., Barçante, J.M.d.P., Dias, S.R.C. et al. Angiostrongylus vasorum (Baillet, 1866) Kamensky, 1905: emergence of third-stage larvae from infected Biomphalaria glabrata snails. Parasitol Res 91, 471–475 (2003). https://doi.org/10.1007/s00436-003-1000-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-003-1000-9