Abstract
Purpose
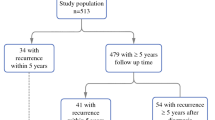
The recommended therapy for type I FIGO IB endometrial cancer (EC) is hysterectomy and adnexectomy, but the therapeutic benefits of additional pelvic and paraaortic lymph node dissection (LND) are still under discussion. In this study, we retrospectively evaluated overall survival (OAS) and recurrence-free survival (RFS) among patients with type I FIGO IB EC who did undergo systematic or elective lymphadenectomy or none at all.
Methods
We selected 299 individuals from the database of the German Tumor Centre Regensburg who were diagnosed between 1998 and 2015 with endometrial adenocarcinoma of the uterus type I FIGO IB. We applied multivariable Cox regression to the selected patient data and estimated hazard ratios for OAS and RFS against the performed intervention. Further, we carried out risk adjustments with respect to clinicopathological parameters, and performed model selection using conditional stepwise forward selection.
Results
We observed significant benefits of LND in the unadjusted survival analysis; however, we did not confirm this effect in multivariable regression analysis upon risk adjustment. In this case, hazard ratio (HR) for OAS in patients without LND versus patients with LND is reduced to 1.214 (95% CI 0.771–1.911; p = 0.402), HR for RFS is 1.059 (95% CI 0.689–1.626; p = 0.795). Similarly, we were also able to eliminate the statistical benefit of systematic versus elective LND by risk adjustment.
Conclusions
In contrast to previous observations in high-grade EC, our study provides compelling evidence that LND, in particular systematic lymphadenectomy, is not beneficial for patients with type I FIGO IB EC in terms of long-term OAS and RFS.



Similar content being viewed by others
References
Achouri A, Huchon C, Bats AS, Bensaid C, Nos C, Lécuru F (2013) Complications of lymphadenectomy for gynecologic cancer. Eur J Surg Oncol 39(1):81–86
Andersen SL, Terry DF, Wilcox MA, Babineau T, Malek K, Perls TT (2005) Cancer in the oldest old. Mech Ageing Dev 126(2):263–267
Benedetti-Panici P, Basile S, Maneschi F, Alberto Lissoni A, Signorelli M, Scambia G et al (2008) Systematic pelvic lymphadenectomy vs. no lymphadenectomy in early-stage endometrial carcinoma: randomized clinical trial. J Natl Cancer Inst 100(23):1707–1716
Bokhman JV (1983) Two pathogenetic types of endometrial carcinoma. Gynecol Oncol 15(1):10–17
Charlson ME, Pompei P, Ales KL, MacKenzie (1987) CR a new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Cragun JM, Havrilesky LJ, Calingaert B, Synan I, Secord AA, Soper JT et al (2005) Retrospective analysis of selective LND in apparent early-stage endometrial cancer. J Clin Oncol 23(16):3668–3675
Cramer DW (2012) The epidemiology of endometrial and ovarian cancer. Hematol Oncol Clin N Am 26(1):1–12
Creasman W (2009) Revised FIGO staging for carcinoma of the endometrium. Int J Gynaecol Obstet 105(2):109
Creasman W, Morrow CP, Bundy BN, Homesley HD, Graham JE, Heller PB (1987) Surgical pathologic spread patterns of endometrial cancer. A Gynecologic Oncology Group Study. Cancer 60(8 suppl):2035–2041
Denschlag D, Ulrich U, Emons G (2010) The diagnosis and treatment of endometrial cancer: progress and controversies. Dtsch Ärzteblatt Int 108(34–35):571–577
Endometrial Cancer Lymphadenectomy Trial (ECLAT) (2008) https://clinicaltrials.gov/ct2/show/NCT03438474. Accessed 01 Apr 2018
Frost JA, Webster KE, Bryant A, Morrison J (2017) Lymphadenectomy for the management of endometrial cancer. Cochrane Database Syst Rev 10:CD007585
Fujimoto T, Nanjyo H, Nakamura A, Yokoyama Y, Takano T, Shoji T et al (2007) Para-aortic lymphadenectomy may improve disease-related survival in patients with multipositive pelvic lymph node stage IIIc endometrial cancer. Gynecol Oncol 107(2):253–259
Gottwald L (2010) Long-term survival of endometrioid endometrial cancer patients. Arch Med Sci 6(6):937–944
Kitchener H, Swart AM, Qian Q, Amos C, Parmar MK (2009) Efficacy of systematic pelvic lymphadenectomy in endometrial cancer (MRC ASTEC trial): a randomised study. Lancet 373(9658):125–136
Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF) (2018) Diagnostik, Therapie und Nachsorge der Patientinnen mit Endometriumkarzinom Leitlinienreport 0.1, 2017, AWMF Registernummer: 032/034-OL. http://www.leitlinienprogramm-onkologie.de/leitlinien/endometriumkarzinom/. Accessed 28 Mar 2018
Mariani A, Webb MJ, Galli L, Podratz KC (2000) Potential therapeutic role of para-aortic LND in node-positive endometrial cancer. Gynecol Oncol 76(3):348–356
May K, Bryant A, Dickinson HO, Kehoe S, Morrison J (2010) Lymphadenectomy for the management of endometrial cancer. Cochrane Database Syst Rev 1:CD007585
Morice P, Leary A, Creutzberg C, Abu-Rustum N, Darai E (2016) Endometrial cancer. Lancet 387(10023):1094–1108
Papathemelis (2017) Survival benefit of pelvic and paraaortic lymphadenectomy in high-grade endometrial carcinoma: a retrospective population-based cohort analysis. J Cancer Res Clin Oncol 143(12):2555–2562
Rani PR, Begum J, Reddy KS (2017) Controversies in the management of early endometrial carcinoma: an update. Int J Reprod Contracept Obstet Gynecol 6(6):2124–2131
Saso S, Chatterjee J, Georgiou E, Ditri AM, Smith JR, Ghaem- Maghami S (2011) Endometrial cancer. BMJ 343(2):d3954–d3954
Thaker PH, Sood AK (2017) Molecular oncology in gynecologic cancer. In: Lentz GM, Lobo RA, Gershenson DM, Katz VL (eds) Comprehensive gynecology (6th edn). Mosby, St. Louis. ISBN 978-0-323-06986-1
Todo Y, Kato H, Kaneuchi M, Watari H, Takeda M, Sakuragi N (2010) Survival effect of para-aortic LND in endometrial cancer (SEPAL study): a retrospective cohort analysis. Lancet 375(9721):1165–1172
Tumorzentrum Regensburg (2018) http://www.tumorzentrum-regensburg.de. Accessed 01 Apr 2018
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Papathemelis, T., Hassas, D., Gerken, M. et al. Is there a benefit of lymphadenectomy for overall and recurrence-free survival in type I FIGO IB G1-2 endometrial carcinoma? A retrospective population-based cohort analysis. J Cancer Res Clin Oncol 144, 2019–2027 (2018). https://doi.org/10.1007/s00432-018-2715-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2715-4