Abstract
Purpose
HSP90 targeting is a promising therapeutic approach in cancer. 17-AAG is an HSP90 inhibitor with completed Phase I trials in patients with advanced cancer and recently published Phase II trials. The aim of this work was to study the expression of HSP 90 and apoptotic proteins, the effects in culture of 17-AAG on cell survival and apoptosis and to compare Philadelphia-positive (Ph+) ALL to common B cell ALL, in ALL cell lines and in patients’ cells collected at ALL diagnosis.
Methods
We analysed 2 ALL cell lines and 63 leukaemic samples from patients treated in our institution (44 common B cell ALL and 19 Ph+ ALL). We performed flow cytometry analysis of bone marrow aspiration and cell lines with a combination of anti-HSP90, Bax, Bcl-2 and Bcl-xl antibodies. Apoptosis after cell culture (in presence or not of 17-AAG) was assessed using Annexin V and activated caspase-3 staining.
Results
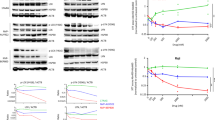
Ph+ ALL cells appeared to be more sensitive to 17-AAG cytotoxicity with a 100 % mortality rate after exposure to 10 μM for 24 h (vs. 62 % for B-common ALL). A high percentage of HSP90-positive cells (in Ph+ ALL samples) was associated with high sensitivity to 17-AAG. 17-AAG induced apoptosis in a dose-dependent manner and was associated with down-regulation of Bcl-2 and Bcl-Xl expression and up-regulation of Bax expression.
Conclusion
Considering that Bcr-Abl constitutes HSP 90 substrates, HSP 90 inhibition could be of particular interest for Ph+ ALL disease, even in patients harbouring resistance to tyrosine kinase inhibitor therapy.




Similar content being viewed by others
References
Bennet JM, Catovsky D, Daniel MT et al (1976) Proposals for the classification of the acute leukaemias: French-American-British (FAB) cooperative group. Br J Haematol 33:451–458
Flandrin P, Guyotat D, Duval A, Cornillon J, Tavernier E, Nadal N, Campos L (2008) Significance of heat-shock protein (HSP90) expression in acute myeloid leukemia cells. Cell Stress Chaperones 13(3):357–364
Gartner EM, Silverman P, Simon M, Flaherty L, Abrams J, Ivy P, Lorusso PM (2012) A phase II study of 17-allylamino-17-demethoxygeldanamycin in metastatic or locally advanced unresectable breast cancer. Breast Cancer Res Treat 131(3):933–937
Goetz MP, Toft DO, Reid JM, Ames M, Stensgard B, Safgren S, Adjei AA, Sloan J, Atherton P, Vasile V, Salazaar S, Adjei A, Croghan G, Erlichman C (2005) A phase I trial of 17-allylamino-17-demethoxygeldanamycin in patients with advanced cancer. J Clin Oncol 23:1078–1087
Hawkins LM, Jayanthan AA, Narendran A (2005) Effects of 17 AAG on pediatric acute lymphoblastic leukemia (ALL) with respect to Bcr-Abl status and imatinib mesylate sensitivity. Pediatr Res 57(3):430–437
Kaufmann SH, Karp JE, litzow MR, Mesa RA, Hogan W, Steensma DP, Flatten KS, Loegering DA, Schneider PA, Maurer MJ, Smith BD, Peterson KL, Greer J, Chen Y, Reid JM, Ivy Sp, Ames MM, Adjei AA, Erlichman C, Karnitz LM (2011) Phase I and pharmacological study of cytarabine and tanespimycin in relapsed and refractory leukaemia. Haematologica 96(11):1619–1626
Lancet JE, Gojo I, Burton M, Quinn M, Tighe SM, Kersey K, Zhong Z, Albitar MX, Bhalla K, Hannah AL, Baer MR (2010) Phase I study of the heat shock protein 90 inhibitor alvespimycin (KOS-1022,17-DMAG) administered intravenously twice weekly to patients with acute myeloid leukemia. Leukemia 24(4):699–705
Lindquist S, Craig EA (1988) The heat shock proteins. Annu Rev Genet 22:631–677
Lu Z, Jin Y, Qiu L, Lai Y, Pan J (2010) Celastrol, a novel HSP90 inhibitor, depletes Bcr-Abl and induces apoptosis in imatinib-resistant chronic myelogenous leukemia cells harboring T315I mutation. Cancer Lett 290(2):182–191
Oh WK, Galsky MD, Stadler WM, Srinivas S, Chu F, Bubley G, Goddard J, Dunbar J, Ross RW (2011) Multicenter phase II trial of the heat shock protein 90 inhibitor, retaspimycin hydrochloride (IPI-504), in patients with castration-resistant prostate cancer. Urology 78(3):626–630
Peng C, Brain J, Goodrich A, Kong L, Grayzel D, Pak R, Read M, Li S (2007) Inhibition of heat shock protein 90 prolongs survival of mice with Bcr-Abl-T315I-induced leukaemia and suppresses leukemic stem cells. Blood 110(2):678–685
Radujkovic A, Schad M, Topaly J, Veldwijk MR, Laufs S, Schultheis BS, Jauch A, Melo JV, Fruehauf S, Zeller WJ (2005) Synergistic activity of imatinib and 17-AAG in imatinib-resistant CML cells overexpressing BCR-ABL- inhibition of P-glycoprotein function by 17-AAG. Leukemia 19(7):1198–1206
Thomas X, Campos L, Mounier C, Cornillon J, Flandrin P, Le QH, Piselli S, Guyotat D (2005) Expression of heat-shock proteins is associated with major adverse prognostic factors in acute myeloid leukemia. Leuk Res 29(9):1049–1058
Tong WG, Estrov Z, Wang Y, O’Brien S, Faderl S, Harris DM, Van Pham Q, Hazan-Halevy I, Liu Z, Koch P, Kantarjian H, Keating MJ, Ferrajoli A (2010) The synthetic heat shock protein 90 (HSP90) inhibitor EC141 induces degradation of Bcr-Abl p190 protein and apoptosis of Ph-positive acute lymphoblastic leukaemia cells. Invest New Drugs 29(6):1206–1212
Tsukahara F, Maru Y (2010) Bag1 directly routes immature BCR-ABL for proteasomal degradation. Blood 116(18):3582–3592
Wu LX, Xu JH, Zhang KZ, Lin Q, Huang XW, Wen CX, Chen YZ (2008) Disruption of the Bcr-Abl/HSP90 protein complex: a possible mechanism to inhibit Bcr-Abl-positive human leukemic blasts by novobiocin. Leukemia 22(7):1402–1409
Young JC, Moarefi I, Hartl UF (2001) HSP 90: a specialized but essential protein-folding tool. J Cell Biol 154(2):267–273
Yufu Y, Nishimura J, Nawata H (1992) High constitutive expression of heat shock protein 90 α in human acute leukemia cells. Leuk Res 16(6–7):597–605
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tavernier, E., Flandrin-Gresta, P., Solly, F. et al. HSP90 inhibition results in apoptosis of Philadelphia acute lymphoblastic leukaemia cells: an attractive prospect of new targeted agents. J Cancer Res Clin Oncol 138, 1753–1758 (2012). https://doi.org/10.1007/s00432-012-1247-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-012-1247-6