Abstract
Background
Cancer patients commonly use dietary supplements to “boost immune function”. A polysaccharide extract from Grifola frondosa (Maitake extract) showed immunomodulatory effects in preclinical studies and therefore the potential for clinical use. Whether oral administration in human produces measurable immunologic effects, however, is unknown.
Methods
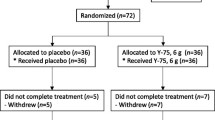
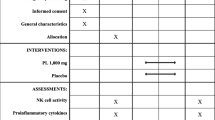
In a phase I/II dose escalation trial, 34 postmenopausal breast cancer patients, free of disease after initial treatment, were enrolled sequentially in five cohorts. Maitake liquid extract was taken orally at 0.1, 0.5, 1.5, 3, or 5 mg/kg twice daily for 3 weeks. Peripheral blood was collected at days −7, 0 (prior to the first dosing), 7, 14, and 21 for ex vivo analyses. The primary endpoints were safety and tolerability.
Results
No dose-limiting toxicity was encountered. Two patients withdrew prior to completion of the study due to grade I possibly related side effects: nausea and joint swelling in one patient; rash and pruritus in the second. There was a statistically significant association between Maitake and immunologic function (p < 0.0005). Increasing doses of Maitake increased some immunologic parameters and depressed others; the dose–response curves for many endpoints were non-monotonic with intermediate doses having either immune enhancing or immune suppressant effects compared with both high and low doses.
Conclusions
Oral administration of a polysaccharide extract from Maitake mushroom is associated with both immunologically stimulatory and inhibitory measurable effects in peripheral blood. Cancer patients should be made aware of the fact that botanical agents produce more complex effects than assumed, and may depress as well as enhance immune function.





Similar content being viewed by others
References
Brown GD, Taylor PR, Reid DM, Willment JA, Williams DL, Martinez-Pomares L et al (2002) Dectin-1 is a major beta-glucan receptor on macrophages. J Exp Med 196(3):407–412. doi:10.1084/jem.20020470
Cheung NK, Modak S, Vickers A, Knuckles B (2002) Orally administered beta-glucans enhance anti-tumor effects of monoclonal antibodies. Cancer Immunol Immunother 51(10):557–564
Couper KN, Blount DG, Riley EM (2008) IL-10: the master regulator of immunity to infection. J Immunol 180(9):5771–5777
Harada N, Kodama N, Nanba H (2003) Relationship between dendritic cells and the D-fraction-induced Th-1 dominant response in BALB/c tumor-bearing mice. Cancer Lett 192(2):181–187. doi:10.1016/S0304-3835(02)00716-4
Hawrylowicz CM (2005) Regulatory T cells and IL-10 in allergic inflammation. J Exp Med 202(11):1459–1463. doi:10.1084/jem.20052211
Herre J, Gordon S, Brown GD (2004) Dectin-1 and its role in the recognition of beta-glucans by macrophages. Mol Immunol 40(12):869–876. doi:10.1016/j.molimm.2003.10.007
Kennedy AD, Willment JA, Dorward DW, Williams DL, Brown GD, DeLeo FR (2007) Dectin-1 promotes fungicidal activity of human neutrophils. Eur J Immunol 37(2):467–478. doi:10.1002/eji.200636653
Kodama N, Komuta K, Sakai N, Nanba H (2002a) Effects of D-Fraction, a polysaccharide from Grifola frondosa on tumor growth involve activation of NK cells. Biol Pharm Bull 25(12):1647–1650. doi:10.1248/bpb.25.1647
Kodama N, Harada N, Nanba H (2002b) A polysaccharide, extract from Grifola frondosa, induces Th-1 dominant responses in carcinoma-bearing BALB/c mice. Jpn J Pharmacol 90(4):357–360. doi:10.1254/jjp.90.357
Kodama N, Komuta K, Nanba H (2003) Effect of Maitake (Grifola frondosa) D-Fraction on the activation of NK cells in cancer patients. J Med Food 6(4):371–377. doi:10.1089/109662003772519949
Kodama N, Murata Y, Asakawa A, Inui A, Hayashi M, Sakai N et al (2005) Maitake D-Fraction enhances antitumor effects and reduces immunosuppression by mitomycin-C in tumor-bearing mice. Nutrition 21(5):624–629. doi:10.1016/j.nut.2004.09.021
Lin H, She YH, Cassileth BR, Sirotnak F, Cunningham Rundles S (2004) Maitake beta-glucan MD-fraction enhances bone marrow colony formation and reduces doxorubicin toxicity in vitro. Int Immunopharmacol 4(1):91–99. doi:10.1016/j.intimp.2003.10.012
Mayell M (2001) Maitake extracts and their therapeutic potential. Altern Med Rev 6(1):48–60
Nanba H, Hamaguchi A, Kuroda H (1987) The chemical structure of an antitumor polysaccharide in fruit bodies of Grifola frondosa (maitake). Chem Pharm Bull (Tokyo) 35(3):1162–1168
O’Garra A, Vieira PL, Vieira P, Goldfeld AE (2004) IL-10-producing and naturally occurring CD4+ Tregs: limiting collateral damage. J Clin Invest 114(10):1372–1378
Penna C, Dean PA, Nelson H (1996) Pulmonary metastases neutralization and tumor rejection by in vivo administration of beta glucan and bispecific antibody. Int J Cancer 65(3):377–382. doi:10.1002/(SICI)1097-0215(19960126)65:3<377::AID-IJC17>3.0.CO;2-7
Ross GD, Vetvicka V, Yan J, Xia Y, Vetvickova J (1999) Therapeutic intervention with complement and beta-glucan in cancer. Immunopharmacology 42(1–3):61–74. doi:10.1016/S0162-3109(99)00013-2
Suzuki I, Itani T, Ohno N, Oikawa S, Sato K, Miyazaki T et al (1985) Effect of a polysaccharide fraction from Grifola frondosa on immune response in mice. J Pharmacobiodyn 8(3):217–226
Suzuki I, Hashimoto K, Oikawa S, Sato K, Osawa M, Yadomae T (1989) Antitumor and immunomodulating activities of a beta-glucan obtained from liquid-cultured Grifola frondosa. Chem Pharm Bull (Tokyo) 37(2):410–413
Takeyama T, Suzuki I, Ohno N, Oikawa S, Sato K, Ohsawa M et al (1987) Host-mediated antitumor effect of grifolan NMF-5N, a polysaccharide obtained from Grifola frondosa. J Pharmacobiodyn 10(11):644–651
Taylor PR, Tsoni SV, Willment JA, Dennehy KM, Rosas M, Findon H et al (2007) Dectin-1 is required for beta-glucan recognition and control of fungal infection. Nat Immunol 8(1):31–38. doi:10.1038/ni1408
Vickers AJ (2006) How to design a phase I trial of an anticancer botanical. J Soc Integr Oncol 4(1):46–51
Willment JA, Marshall AS, Reid DM, Williams DL, Wong SY, Gordon S et al (2005) The human beta-glucan receptor is widely expressed and functionally equivalent to murine Dectin-1 on primary cells. Eur J Immunol 35(5):1539–1547. doi:10.1002/eji.200425725
Wu K, Bi Y, Sun K, Wang C (2007) IL-10-producing type 1 regulatory T cells and allergy. Cell Mol Immunol 4(4):269–275
Acknowledgments
We would like to thank Tina Chuck, Kristine Brown and Carrie Trevisan (Research Study Assistants) for their work in this study. The study was supported by the Memorial Sloan-Kettering Cancer Center Translational Research Grant and Grant Number P50 AT002779 from the National Center for Complementary and Alternative Medicine (NCCAM) and the Office of Dietary Supplements (ODS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deng, G., Lin, H., Seidman, A. et al. A phase I/II trial of a polysaccharide extract from Grifola frondosa (Maitake mushroom) in breast cancer patients: immunological effects. J Cancer Res Clin Oncol 135, 1215–1221 (2009). https://doi.org/10.1007/s00432-009-0562-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-009-0562-z