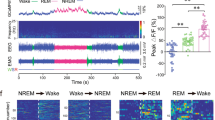
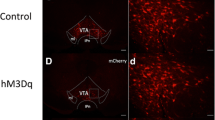
Abstract
Rapid eye movement (REM) sleep behavior disorder in humans is often accompanied by a reduced ability to smell and detect odors, and olfactory bulbectomized rats exhibit increased REM sleep, suggesting that the olfactory bulb (OB) is involved in REM-sleep regulation. However, the molecular mechanism of REM-sleep regulation by the OB is unknown. Adenosine promotes sleep and its A2A receptors (A2AR) are expressed in the OB. We hypothesized that A2AR in the OB regulate REM sleep. Bilateral microinjections of the A2AR antagonist SCH58261 into the rat OB increased REM sleep, whereas microinjections of the A2AR agonist CGS21680 decreased REM sleep. Similar to the A2AR antagonist, selective A2AR knockdown by adeno-associated virus carrying short-hairpin RNA for A2AR in the rat OB increased REM sleep. Using chemogenetics on the basis of designer receptors exclusively activated by designer drugs, we demonstrated that the inhibition of A2AR neurons increased REM sleep, whereas the activation of these neurons decreased REM sleep. Moreover, using a conditional anterograde axonal tract-tracing approach, we found that OB A2AR neurons innervate the piriform cortex and olfactory tubercle. These novel findings indicate that adenosine suppresses REM sleep via A2AR in the OB of rodents.






Similar content being viewed by others
References
Alexander GM, Rogan SC, Abbas AI, Armbruster BN, Pei Y, Allen JA, Nonneman RJ, Hartmann J, Moy SS, Nicolelis MA, McNamara JO, Roth BL (2009) Remote control of neuronal activity in transgenic mice expressing evolved G protein-coupled receptors. Neuron 63(1):27–39. doi:10.1016/j.neuron.2009.06.014
Anaclet C, Pedersen NP, Ferrari LL, Venner A, Bass CE, Arrigoni E, Fuller PM (2015) Basal forebrain control of wakefulness and cortical rhythms. Nature communications 6:8744. doi:10.1038/ncomms9744
Armbruster BN, Li X, Pausch MH, Herlitze S, Roth BL (2007) Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc Natl Acad Sci USA 104(12):5163–5168. doi:10.1073/pnas.0700293104
Basheer R, Strecker RE, Thakkar MM, McCarley RW (2004) Adenosine and sleep-wake regulation. Prog Neurobiol 73(6):379–396. doi:10.1016/j.pneurobio.2004.06.004
Boeve BF (2013) Idiopathic REM sleep behaviour disorder in the development of Parkinson’s disease. Lancet Neurol 12(5):469–482. doi:10.1016/S1474-4422(13)70054-1
Carskadon MA, Herz RS (2004) Minimal olfactory perception during sleep: why odor alarms will not work for humans. Sleep 27(3):402–405
Chang CH, Chen MC, Qiu MH, Lu J (2014) Ventromedial prefrontal cortex regulates depressive-like behavior and rapid eye movement sleep in the rat. Neuropharmacology 86:125–132. doi:10.1016/j.neuropharm.2014.07.005
Chieng B, Christie MJ (2010) Somatostatin and nociceptin inhibit neurons in the central nucleus of amygdala that project to the periaqueductal grey. Neuropharmacology 59(6):425–430. doi:10.1016/j.neuropharm.2010.06.001
Doengi M, Deitmer JW, Lohr C (2008) New evidence for purinergic signaling in the olfactory bulb: A2A and P2Y1 receptors mediate intracellular calcium release in astrocytes. FASEB J 22(7):2368–2378. doi:10.1096/fj.07-101782
Durieux PF, Bearzatto B, Guiducci S, Buch T, Waisman A, Zoli M, Schiffmann SN, de Kerchove d’Exaerde A (2009) D2R striatopallidal neurons inhibit both locomotor and drug reward processes. Nat Neurosci 12(4):393–395. doi:10.1038/nn.2286
Durieux PF, Schiffmann SN, de Kerchove d’Exaerde A (2012) Differential regulation of motor control and response to dopaminergic drugs by D1R and D2R neurons in distinct dorsal striatum subregions. EMBO J 31(3):640–653. doi:10.1038/emboj.2011.400
Ena SL, De Backer JF, Schiffmann SN, de Kerchove d’Exaerde A (2013) FACS array profiling identifies Ecto-5′ nucleotidase as a striatopallidal neuron-specific gene involved in striatal-dependent learning. J Neurosci 33(20):8794–8809. doi:10.1523/JNEUROSCI.2989-12.2013
Estabrooke IV, McCarthy MT, Ko E, Chou TC, Chemelli RM, Yanagisawa M, Saper CB, Scammell TE (2001) Fos expression in orexin neurons varies with behavioral state. J Neurosci 21(5):1656–1662
Fantini ML, Ferini-Strambi L, Montplaisir J (2005) Idiopathic REM sleep behavior disorder: toward a better nosologic definition. Neurology 64(5):780–786. doi:10.1212/01.WNL.0000152878.79429.00
Feldberg W, Sherwood SL (1954) Injections of drugs into the lateral ventricle of the cat. J Physiol 123(1):148–167
Fuller PM, Saper CB, Lu J (2007) The pontine REM switch: past and present. J Physiol 584(Pt 3):735–741. doi:10.1113/jphysiol.2007.140160
Gautron L, Lazarus M, Scott MM, Saper CB, Elmquist JK (2010) Identifying the efferent projections of leptin-responsive neurons in the dorsomedial hypothalamus using a novel conditional tracing approach. J Comp Neurol 518(11):2090–2108. doi:10.1002/cne.22323
Genzel L, Spoormaker VI, Konrad BN, Dresler M (2015) The role of rapid eye movement sleep for amygdala-related memory processing. Neurobiol Learn Mem 122:110–121. doi:10.1016/j.nlm.2015.01.008
Giessel AJ, Datta SR (2014) Olfactory maps, circuits and computations. Curr Opin Neurobiol 24(1):120–132. doi:10.1016/j.conb.2013.09.010
Gompf HS, Budygin EA, Fuller PM, Bass CE (2015) Targeted genetic manipulations of neuronal subtypes using promoter-specific combinatorial AAVs in wild-type animals. Front Behav Neurosci 9:152. doi:10.3389/fnbeh.2015.00152
Huang ZL, Qu WM, Eguchi N, Chen JF, Schwarzschild MA, Fredholm BB, Urade Y, Hayaishi O (2005) Adenosine A2A, but not A1, receptors mediate the arousal effect of caffeine. Nat Neurosci 8(7):858–859. doi:10.1038/nn1491
Huang ZL, Urade Y, Hayaishi O (2011) The role of adenosine in the regulation of sleep. Curr Top Med Chem 11(8):1047–1057
Huang ZL, Zhang Z, Qu WM (2014) Roles of adenosine and its receptors in sleep-wake regulation. Int Rev Neurobiol 119:349–371. doi:10.1016/B978-0-12-801022-8.00014-3
Iranzo A (2013) Parkinson disease and sleep: sleep-wake changes in the premotor stage of Parkinson disease; impaired olfaction and other prodromal features. Curr Neurol Neurosci Rep 13(9):373. doi:10.1007/s11910-013-0373-0
Jego S, Glasgow SD, Herrera CG, Ekstrand M, Reed SJ, Boyce R, Friedman J, Burdakov D, Adamantidis AR (2013) Optogenetic identification of a rapid eye movement sleep modulatory circuit in the hypothalamus. Nat Neurosci 16(11):1637–1643. doi:10.1038/nn.3522
Johansson B, Georgiev V, Fredholm BB (1997) Distribution and postnatal ontogeny of adenosine A2A receptors in rat brain: comparison with dopamine receptors. Neuroscience 80(4):1187–1207
Kaelin-Lang A, Lauterburg T, Burgunder JM (1999) Expression of adenosine A2a receptors gene in the olfactory bulb and spinal cord of rat and mouse. Neurosci Lett 261(3):189–191
Kalinchuk AV, Urrila AS, Alanko L, Heiskanen S, Wigren HK, Suomela M, Stenberg D, Porkka-Heiskanen T (2003) Local energy depletion in the basal forebrain increases sleep. Eur J Neurosci 17(4):863–869
Konadhode RR, Pelluru D, Blanco-Centurion C, Zayachkivsky A, Liu M, Uhde T, Glen WB Jr, van den Pol AN, Mulholland PJ, Shiromani PJ (2013) Optogenetic stimulation of MCH neurons increases sleep. J Neurosci 33(25):10257–10263. doi:10.1523/JNEUROSCI.1225-13.2013
Krashes MJ, Koda S, Ye C, Rogan SC, Adams AC, Cusher DS, Maratos-Flier E, Roth BL, Lowell BB (2011) Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J Clin Invest 121(4):1424–1428. doi:10.1172/JCI46229
Kugler S, Kilic E, Bahr M (2003) Human synapsin 1 gene promoter confers highly neuron-specific long-term transgene expression from an adenoviral vector in the adult rat brain depending on the transduced area. Gene Ther 10(4):337–347. doi:10.1038/sj.gt.3301905
Lazarus M, Shen HY, Cherasse Y, Qu WM, Huang ZL, Bass CE, Winsky-Sommerer R, Semba K, Fredholm BB, Boison D, Hayaishi O, Urade Y, Chen JF (2011) Arousal effect of caffeine depends on adenosine A2A receptors in the shell of the nucleus accumbens. J Neurosci 31(27):10067–10075. doi:10.1523/JNEUROSCI.6730-10.2011
Lazarus M, Chen JF, Urade Y, Huang ZL (2013) Role of the basal ganglia in the control of sleep and wakefulness. Curr Opin Neurobiol 23(5):780–785. doi:10.1016/j.conb.2013.02.001
Leite-Panissi CR, Coimbra NC, Menescal-de-Oliveira L (2003) The cholinergic stimulation of the central amygdala modifying the tonic immobility response and antinociception in guinea pigs depends on the ventrolateral periaqueductal gray. Brain Res Bull 60(1–2):167–178
Lu J, Sherman D, Devor M, Saper CB (2006) A putative flip-flop switch for control of REM sleep. Nature 441(7093):589–594. doi:10.1038/nature04767
Mavanji V, Siwek DF, Patterson EH, Spoley EE, Datta S (2003) Effects of passive-avoidance training on sleep-wake state-specific activity in the basolateral and central nuclei of the amygdala. Behav Neurosci 117(4):751–759
Miyamichi K, Amat F, Moussavi F, Wang C, Wickersham I, Wall NR, Taniguchi H, Tasic B, Huang ZJ, He Z, Callaway EM, Horowitz MA, Luo L (2011) Cortical representations of olfactory input by trans-synaptic tracing. Nature 472(7342):191–196. doi:10.1038/nature09714
Miyamoto T, Miyamoto M, Iwanami M, Hirata K, Kobayashi M, Nakamura M, Inoue Y (2010) Olfactory dysfunction in idiopathic REM sleep behavior disorder. Sleep Med 11(5):458–461. doi:10.1016/j.sleep.2009.09.013
Niu JG, Yokota S, Tsumori T, Oka T, Yasui Y (2012) Projections from the anterior basomedial and anterior cortical amygdaloid nuclei to melanin-concentrating hormone-containing neurons in the lateral hypothalamus of the rat. Brain Res 1479:31–43. doi:10.1016/j.brainres.2012.08.011
Oishi Y, Huang ZL, Fredholm BB, Urade Y, Hayaishi O (2008) Adenosine in the tuberomammillary nucleus inhibits the histaminergic system via A1 receptors and promotes non-rapid eye movement sleep. Proc Natl Acad Sci USA 105(50):19992–19997. doi:10.1073/pnas.0810926105
Oishi Y, Takata Y, Taguchi Y, Kohtoh S, Urade Y, Lazarus M (2016) Polygraphic recording procedure for measuring sleep in mice. J Vis Exp 107:e53678. doi:10.3791/53678
Pawlyk AC, Sanford LD, Brennan FX, Morrison AR, Ross RJ (2006) Corticotropin-releasing factor microinjection into the central nucleus of the amygdala alters REM sleep. Pharmacol Rep 58(1):125–130
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates. Elsevier, Boston
Porkka-Heiskanen T, Strecker RE, Thakkar M, Bjorkum AA, Greene RW, McCarley RW (1997) Adenosine: a mediator of the sleep-inducing effects of prolonged wakefulness. Science 276(5316):1265–1268
Postuma RB, Gagnon JF, Vendette M, Montplaisir JY (2009) Idiopathic REM sleep behavior disorder in the transition to degenerative disease. Mov Disord 24(15):2225–2232. doi:10.1002/mds.22757
Prediger RD, Batista LC, Takahashi RN (2005) Caffeine reverses age-related deficits in olfactory discrimination and social recognition memory in rats. Involvement of adenosine A1 and A2A receptors. Neurobiol Aging 26(6):957–964. doi:10.1016/j.neurobiolaging.2004.08.012
Qu WM, Xu XH, Yan MM, Wang YQ, Urade Y, Huang ZL (2010) Essential role of dopamine D2 receptor in the maintenance of wakefulness, but not in homeostatic regulation of sleep, in mice. J Neurosci 30(12):4382–4389. doi:10.1523/JNEUROSCI.4936-09.2010
Root CM, Denny CA, Hen R, Axel R (2014) The participation of cortical amygdala in innate, odour-driven behaviour. Nature 515(7526):269–273. doi:10.1038/nature13897
Scammell TE, Gerashchenko DY, Mochizuki T, McCarthy MT, Estabrooke IV, Sears CA, Saper CB, Urade Y, Hayaishi O (2001) An adenosine A2a agonist increases sleep and induces Fos in ventrolateral preoptic neurons. Neuroscience 107(4):653–663
Schredl M, Atanasova D, Hormann K, Maurer JT, Hummel T, Stuck BA (2009) Information processing during sleep: the effect of olfactory stimuli on dream content and dream emotions. J Sleep Res 18(3):285–290. doi:10.1111/j.1365-2869.2009.00737.x
Song C, Leonard BE (2005) The olfactory bulbectomised rat as a model of depression. Neurosci Biobehav Rev 29(4–5):627–647. doi:10.1016/j.neubiorev.2005.03.010
Stark ME, Safir I, Wisco JJ (2014) Probabilistic mapping of the cervical sympathetic trunk ganglia. Auton Neurosci 181:79–84. doi:10.1016/j.autneu.2014.01.007
Stiasny-Kolster K, Clever SC, Moller JC, Oertel WH, Mayer G (2007) Olfactory dysfunction in patients with narcolepsy with and without REM sleep behaviour disorder. Brain 130(Pt 2):442–449. doi:10.1093/brain/awl343
Treloar HB, Miller AM, Ray A, Greer CA (2010) Development of the olfactory system. In: Menini A (ed) The neurobiology of olfaction. Frontiers in Neuroscience. CRC Press, Boca Raton
Tsunematsu T, Ueno T, Tabuchi S, Inutsuka A, Tanaka KF, Hasuwa H, Kilduff TS, Terao A, Yamanaka A (2014) Optogenetic manipulation of activity and temporally controlled cell-specific ablation reveal a role for MCH neurons in sleep/wake regulation. J Neurosci 34(20):6896–6909. doi:10.1523/JNEUROSCI.5344-13.2014
Tupal S, Faingold CL (2012) The amygdala to periaqueductal gray pathway: plastic changes induced by audiogenic kindling and reversal by gabapentin. Brain Res 1475:71–79. doi:10.1016/j.brainres.2012.07.044
Urade Y, Eguchi N, Qu WM, Sakata M, Huang ZL, Chen JF, Schwarzschild MA, Fink JS, Hayaishi O (2003) Sleep regulation in adenosine A2A receptor-deficient mice. Neurology 61(11 Suppl 6):S94–S96
Urban DJ, Roth BL (2015) DREADDs (designer receptors exclusively activated by designer drugs): chemogenetic tools with therapeutic utility. Annu Rev Pharmacol Toxicol 55:399–417. doi:10.1146/annurev-pharmtox-010814-124803
Wang YQ, Tu ZC, Xu XY, Li R, Qu WM, Urade Y, Huang ZL (2012) Acute administration of fluoxetine normalizes rapid eye movement sleep abnormality, but not depressive behaviors in olfactory bulbectomized rats. J Neurochem 120(2):314–324. doi:10.1111/j.1471-4159.2011.07558.x
Wang YQ, Takata Y, Li R, Zhang Z, Zhang MQ, Urade Y, Qu WM, Huang ZL (2015) Doxepin and diphenhydramine increased non-rapid eye movement sleep through blockade of histamine H1 receptors. Pharmacol Biochem Behav 129:56–64. doi:10.1016/j.pbb.2014.12.002
Weaver DR (1993) A2a adenosine receptor gene expression in developing rat brain. Brain Res Mol Brain Res 20(4):313–327
Zhang JP, Xu Q, Yuan XS, Cherasse Y, Schiffmann SN, de Kerchove d’Exaerde A, Qu WM, Urade Y, Lazarus M, Huang ZL, Li RX (2013) Projections of nucleus accumbens adenosine A2A receptor neurons in the mouse brain and their implications in mediating sleep-wake regulation. Front Neuroanat 7:43. doi:10.3389/fnana.2013.00043
Acknowledgments
This study was supported in part by grants-in-aid for scientific research from the National Natural Science Foundation of China (81571295, 31530035, 31471064, 31271164, and 81420108015); the National Basic Research Program of China (2015CB856401 and 2011CB711000); a key laboratory program of the Education Commission of Shanghai Municipality (ZDSYS14005); the Shanghai Committee of Science and Technology (14JC1400900); the Japan Society for the Promotion of Science (KAKENHI Grant 2604762); the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan (Grant for Scientific Research on Innovative Areas “Living in Space”, 16H01629); the World Premier International Research Center Initiative (WPI) from MEXT; FMRE-Belgium; FRS–FNRS, Belgium; Interuniversity Attraction Pole (IUAP—P7/10) from Belgian Federal Scientific Affairs, and Action de Recherche Concertée (FWB).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
Y.-Q. Wang, R. Li and D.-R. Wang contributed equally.
Rights and permissions
About this article
Cite this article
Wang, YQ., Li, R., Wang, DR. et al. Adenosine A2A receptors in the olfactory bulb suppress rapid eye movement sleep in rodents. Brain Struct Funct 222, 1351–1366 (2017). https://doi.org/10.1007/s00429-016-1281-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-016-1281-2