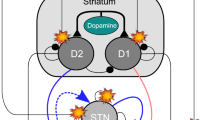
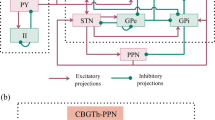
Abstract
This paper develops a new closed-loop firing rate regulation strategy for a population of neurons in the subthalamic nucleus, derived using a model-based analysis of the basal ganglia. The system is described using a firing rate model, in order to analyse the generation of beta-band oscillations. On this system, a proportional regulation of the firing rate reduces the gain of the subthalamo-pallidal loop in the parkinsonian case, thus impeding pathological oscillation generation. A filter with a well-chosen frequency is added to this proportional scheme, in order to avoid a potential instability of the feedback loop due to actuation and measurement delays. Our main result is a set of conditions on the parameters of the stimulation strategy that guarantee both its stability and a prescribed delay margin. A discussion on the applicability of the proposed method and a complete set of mathematical proofs is included.





Similar content being viewed by others
References
Agarwal R, Sarma SV (2010) Restoring the basal ganglia in Parkinson’s disease to normal via multi-input phase-shifted deep brain stimulation. In: IEEE/EMBS conference on neural engineering (NER)
Aström KJ, Murray RM (2010) Feedback systems: an introduction for scientists and engineers. Princeton University Press, Princeton
Benabid AL, Pollak P, Gervason C, Hoffmann D, Gao DM, Hommel M, Perret JE, de Rougemont J (1991) Long-term suppression of tremor by chronic stimulation of the ventral intermediate thalamic nucleus. Lancet 337:403–406
Bergman H, Wichmann T, Delong MR (1990) Reversal of experimental parkinsonism by lesions of the subthalamic nucleus. Science 249:1436–1438
Boyden ES (2011) A history of optogenetics: the development of tools for controlling brain circuits with light. F1000 Biol Rep 3:1–12. Art ID 11
Bragin A, Wilson CL, Staba RJ, Reddick M, Fried I, Engel J (2002) Interictal high-frequency oscillations (80–50 Hz) in the human epileptic brain: entorhinal cortex. Ann Neurol 52(4):407–415
Carron R, Chaillet A, Filipchuk A, Pasillas-Lépine W, Hammond C (2013) Closing the loop of deep brain stimulation. Front Syst Neurosci 7:1–18. Art ID 112
Carron R, Filipchuk A, Nardou R, Singh A, Michel FJ, Humphries MD, Hammond C (2014) Early hypersynchrony in juvenile pink1(-)/(-) motor cortex is rescued by antidromic stimulation. Front Syst Neurosci 8:1–12. Art ID 95
Coombes S, Laing C (2009) Delays in activity-based neural networks. Philos Trans R Soc A Math Phys Eng Sci 367(1891):1117–1129
Curtain RF, Zwart H (1995) An introduction to infinite-dimensional linear systems theory, volume 21 of texts in applied mathematics, 1st edn. Springer, New York
Davie CA (2008) A review of Parkinson’s disease. Br Med Bull 86(1):109–127
Dayan P, Abbott LF (2001) Theoretical neuroscience: computational and mathematical modeling of neural systems. The MIT Press, Cambridge
Dovzhenok A, Park C, Worth RM, Rubchinsky LL (2013) Failure of delayed feedback deep brain stimulation for intermittent pathological synchronization in Parkinson’s disease. PLoS ONE 8(3):e58264
Doyle J, Francis B, Tannenbaum A (1990) Feedback control theory. Macmillan Publishing Co., London
Doyle JC, Glover K, Khargonekar PP, Francis B (1989) State-space solutions to standard \(h_2\) and \(h_\infty \) control problems. IEEE Trans Autom Control 34(8):831–847
Dunn EM, Lowery MM (2013) Simulation of PID control schemes for closed-loop deep brain stimulation. In: IEEE/EMBS conference on neural engineering (NER), San Diego (California)
Feng X, Greenwald B, Rabitz H, Shea-Brown E, Kosut R (2007) Toward closed-loop optimization of deep brain stimulation for parkinson’s disease: concepts and lessons from a computational model. J Neural Eng 4:L14
Franci A, Chaillet A, Pasillas-Lépine W (2011) Existence and robustness of phase-locking in coupled Kuramoto oscillators under mean-field feedback. Automatica 47(6):1193–1202
Fridman E, Shaked U (2002) An improved stabilization method for linear time-delay systems. IEEE Trans Autom Control 47(11):1931–1937
Gahinet P, Nemirovskii A, Laub AJ, Chilali M (1994) The LMI control toolbox. In: Proceedings of the IEEE conference on decision and control, pp 2038–2041
Gonzalez-Burgos G, Lewis DA (2008) GABA neurons and the mechanisms of network oscillations: implications for understanding cortical dysfunction in schizophrenia. Schizophr Bull 34(5):944–961
Gradinaru V, Mogri M, Thompson KR, Henderson JM, Deisseroth K (2009) Optical deconstruction of parkinsonian neural circuitry. Science 324(5925):354–359
Gray CM, König P, Engel AK, Singer W (1989) Oscillatory responses in cat visual cortex exhibit inter-columnar synchronization which reflects global stimulus properties. Nature 338(6213):334–337
Haidar I, Pasillas-Lépine W, Panteley E, Chaillet A, Palfi S, Senova S (2014) Analysis of delay-induced basal ganglia oscillations: the role of external excitatory nuclei. Int J Control 87(9):1936–1956
Hammond C, Bergman H, Brown P (2007) Pathological synchronization in Parkinson’s disease: networks, models and treatments. Trends Neurosci 30(7):357–364 INMED/TINS special issue–Physiogenic and pathogenic oscillations: the beauty and the beast
Han X, Boyden ES (2007) Multiple-color optical activation, silencing, and desynchronization of neural activity, with single-spike temporal resolution. PLoS ONE 2(3):299
Han X, Qian X, Stern P, Chuong AS, Boyden ES (2009) Informational lesions: optical perturbation of spike timing and neural synchrony via microbial opsin gene fusions. Front Mol Neurosci 2:1–9. Art ID 12
Han X, Chow BY, Zhou H, Klapoetke NC, Chuong A, Rajimehr R, Yang A, Baratta MV, Winkle J, Desimone R et al (2011) A high-light sensitivity optical neural silencer: development and application to optogenetic control of non-human primate cortex. Front Syst Neurosci 5:18
Hauptmann C, Popovych O, Tass PA (2005a) Delayed feedback control of synchronization in locally coupled neuronal networks. Neurocomputing 65:759–767
Hauptmann C, Popovych O, Tass PA (2005b) Effectively desynchronizing deep brain stimulation based on a coordinated delayed feedback stimulation via several sites: a computational study. Biol Cybern 93(6):463–470
He Y, Wang QG, Lin C, Wu M (2007) Delay-range-dependent stability for systems with time-varying delay. Automatica 43(2):371–376
Jenkinson N, Brown P (2011) New insights into the relationship between dopamine, beta oscillations and motor function. Trends Neurosci 34(12):611–618
Kumar R, Lozano AM, Sime E, Lang AE (2003) Long-term follow-up of thalamic deep brain stimulation for essential and parkinsonian tremor. Neurology 61(11):1601–1604
Leblois A, Boraud T, Meissner W, Bergman H, Hansel D (2006) Competition between feedback loops underlies normal and pathological dynamics in the basal ganglia. J Neurosci 26(13):3567–3583
Legatt AD, Arezzo J, Vaughan HG Jr (1980) Averaged multiple unit activity as an estimate of phasic changes in local neuronal activity: effects of volume-conducted potentials. J Neurosci Methods 2(2):203–217
Limousin P, Pollak P, Benazzouz A, Hoffmann D, Le Bas J-F, Perret J-E, Benabid A-L, Broussolle E (1995) Effect on parkinsonian signs and symptoms of bilateral subthalamic nucleus stimulation. Lancet 345(8942):91–95
Little S, Pogosyan A, Kuhn AA, Brown P (2012) Beta band stability over time correlates with Parkinsonian rigidity and bradykinesia. Exp Neurol 236(2):383–388
Liu J, Oweiss KG, Khalil HK (2010) Feedback control of the spatiotemporal firing patterns of neural microcircuits. In: IEEE conference on decision and control
Liu J, Khalil HK, Oweiss KG (2011) Model-based spatiotemporal analysis and control of a network of spiking basal ganglia neurons. In: IEEE/EMBS conference on neural engineering (NER)
Lysyansky B, Popovych OV, Tass PA (2011) Desynchronizing anti-resonance effect of m: n on-off coordinated reset stimulation. J Neural Eng 8:036019
McIntyre C, Savasta M, Kerkerian-Le Goff L, Vitek JL (2004a) Uncovering the mechanism(s) of action of deep brain stimulation: activation, inhibition, or both. Clin Neurophysiol 115(6):1239–1248
McIntyre CC, Grill WM, Sherman DL, Thakor NV (2004b) Cellular effects of deep brain stimulation: model-based analysis of activation and inhibition. J Neurophysiol 91(4):1457–1469
Michmizos KP, Sakas D, Nikita KS (2012) Prediction of the timing and the rhythm of the parkinsonian subthalamic nucleus neural spikes using the local field potentials. IEEE Trans Inf Technol Biomed 16(2):190–197
Middleton RH, Miller DE (2007) On the achievable delay margin using LTI control for unstable plants. IEEE Trans Autom Control 52(7):1194–1207
Mitzdorf U (1987) Properties of the evoked potential generators: current source-density analysis of visually evoked potentials in the cat cortex. Int J Neurosci 33(1–2):33
Modolo J, Henry J, Beuter A (2008) Dynamics of the subthalamo-pallidal complex in parkinson’s disease during deep brain stimulation. J Biol Phys 34(3–4):251–266
Montaseri G, Yazdanpanah MJ, Pikovsky A, Rosenblum M (2013) Synchrony suppression in ensembles of coupled oscillators via adaptive vanishing feedback. Chaos: an interdisciplinary. J Nonlinear Sci 23(3):033122
Nambu A (2011) Somatotopic organization of the primate basal ganglia. Front Neuroanat 5:1–9. Art ID 26
Nevado-Holgado AL, Terry JR, Bogacz R (2010) Conditions for the generation of beta oscillations in the subthalamic nucleus–globus pallidus network. J Neurosci 30(37):12340–12352
Nini A, Feingold A, Slovin H, Bergman H (1995) Neurons in the globus pallidus do not show correlated activity in the normal monkey, but phase-locked oscillations appear in the MPTP model of parkinsonism. J Neurophysiol 74(4):1800–1805
Omel’chenko OE, Hauptmann C, Maistrenko YL, Tass PA (2008) Collective dynamics of globally coupled phase oscillators under multisite delayed feedback stimulation. Phys D 237(3):365–384
Park C, Worth RM, Rubchinsky LL (2010) Fine temporal structure of beta oscillations synchronization in subthalamic nucleus in Parkinson’s disease. J Neurophysiol 103(5):2707–2716
Pascual A, Modolo J, Beuter A (2006) Is a computational model useful to understand the effect of deep brain stimulation in Parkinson’s disease? J Integr Neurosci 5(04):541–559
Pasillas-Lépine W (2013) Delay-induced oscillations in Wilson and Cowan’s model: an analysis of the subthalamo-pallidal feedback loop in healthy and parkinsonian subjects. Biol Cybern 107(3):289–308
Pavlides A, Hogan J, Bogacz R (2012) Improved conditions for the generation of beta oscillations in the subthalamic nucleus–globus pallidus network. Eur J Neurosci 36(2):2229–2239
Plenz D, Kital ST (1999) A basal ganglia pacemaker formed by the subthalamic nucleus and external globus pallidus. Nature 400(6745):677–682
Pogosyan A, Yoshida F, Chen CC, Martinez-Torres I, Foltynie T, Limousin P, Zrinzo L, Hariz MI, Brown P (2010) Parkinsonian impairment correlates with spatially extensive subthalamic oscillatory synchronization. Neuroscience 171(1):245–257
Pyragas K, Popovych OV, Tass PA (2007) Controlling synchrony in oscillatory networks with a separate stimulation-registration setup. Europhys Lett 80(4):40002-p1–40002-p6
Rosin B, Slovik M, Mitelman R, Rivlin-Etzion M, Haber SN, Israel Z, Vaadia E, Bergman H (2011) Closed-loop deep brain stimulation is superior in ameliorating parkinsonism. Neuron 72(2):370–384
Rubin JE, Terman D (2004) High frequency stimulation of the subthalamic nucleus eliminates pathological thalamic rhythmicity in a computational model. J Comput Neurosci 16(3):211–235
Santaniello S, Fiengo G, Glielmo L, Grill WM (2011) Closed-loop control of deep brain stimulation: a simulation study. IEEE Trans Neural Syst Rehabil Eng 19(1):15–24
Schiff SJ (2010) Towards model-based control of Parkinson’s disease. Philos Trans R Soc A Math Phys Eng Sci 368(1918):2269–2308
Seuret A, Gouaisbaut F, Fridman E (2013) Stability of systems with fast-varying delay using improved Wirtinger’s inequality. In: IEEE conference on decision and control (CDC), pp 946–951, Florence (Italy)
Sharott A, Magill PJ, Harnack D, Kupsch A, Meissner W, Brown P (2005) Dopamine depletion increases the power and coherence of \(\beta \)-oscillations in the cerebral cortex and subthalamic nucleus of the awake rat. Eur J Neurosci 21(5):1413–1422
Sipahi R, Niculescu SI, Abdallah CT, Michiels W, Gu K (2011) Stability and stabilization of systems with time delay. IEEE Control Syst Mag 31(1):38–65
Strafella AP, Vanderwerf Y, Sadikot AF (2004) Transcranial magnetic stimulation of the human motor cortex influences the neuronal activity of subthalamic nucleus. Eur J Neurosci 20(8):2245–2249
Teleńczuk B, Destexhe A (2014) Local field potential, relationship to unit activity. In: Jaeger D, Jung R (eds) Encyclopedia of computational neuroscience. Springer, New York, pp 1–6
Tufail Y, Matyushov A, Baldwin N, Tauchmann ML, Georges J, Yoshihiro A, Tillery SIH, Tyler WJ (2010) Transcranial pulsed ultrasound stimulates intact brain circuits. Neuron 66(5):681–694
Tukhlina N, Rosenblum M, Pikovsky A, Kurths J (2007) Feedback suppression of neural synchrony by vanishing stimulation. Phys Rev E 75(1):011918
Tye KM, Deisseroth K (2012) Optogenetic investigation of neural circuits underlying brain disease in animal models. Nat Rev Neurosci 13(4):251–266
Wagenaar DA, Madhavan R, Pine J, Potter SM (2005) Controlling bursting in cortical cultures with closed-loop multi-electrode stimulation. J Neurosci 25(3):680–688
Zaidel A, Spivak A, Grieb B, Bergman H, Israel Z (2010) Subthalamic span of oscillations predicts deep brain stimulation efficacy for patients with Parkinson’s disease. Brain 133(7):2007–2021
Zhang F, Wang L-P, Brauner M, Liewald JF, Kay K, Watzke N, Wood PG, Bamberg E, Nagel G, Gottschalk A et al (2007) Multimodal fast optical interrogation of neural circuitry. Nature 446(7136):633–639
Acknowledgments
This work was financially supported by the European Commission through the FP7 NoE HYCON2 and by the region Ile-de-France through the Neurosynch project (RTRA Digiteo). The work of the fourth author was supported by the Government of the Russian Federation (grant 074-U01).
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: The Nyquist criterion
Consider a transfer function G(s), and in order to keep the discussion simple, assume that it is rational and that it has no poles on the imaginary axis. Note that the poles of a rational transfer function are the roots of its denominator, while its zeros are the roots of its numerator, and that such a transfer function is stable if all its poles have a strictly negative real part. A pole with strictly positive real part is called unstable. A standard question, in control theory, is to determine whether the closed-loop transfer function
associated with the feedback interconnection in Fig. 6, is stable. Observe that \(y(s) = H(s) u(s)\).
An answer to this question is given by Nyquist’s stability theorem. Consider the Nyquist contour \(\varGamma \) (see Fig. 7), composed of a straight line on the imaginary axis, going from \(-iR\) to \(+iR\), and a half-circle of radius R. It is assumed that R is big enough so that all unstable poles of G(s) are contained in the interior of \(\varGamma \). The Nyquist plot of the open-loop transfer G(s) is the image of \(\varGamma \) under the complex function G (see Fig. 8).
Nyquist plots of the open-loop transfer \(G(s) = 1/\left( (s+1) ( s^2 + 2 \zeta s + 1 ) \right) \). The solid and dashed parts of the plot correspond, respectively, to the positive and negative frequencies on the imaginary axis of the Nyquist contour. The dotted part of the contour does not appear in the plot, when R is big enough, because the image of the circle at infinity by a strictly proper rational function is a single point at the origin. Left when \(\zeta =1/2\), the Nyquist locus does not encircle the critical point \(-1\). Since G(s) is stable (\(P=0\)), the closed-loop transfer H(s) is then also stable. Right when \(\zeta =1/6\), the Nyquist locus encircles \(-1\), and thus H(s) is unstable
Theorem 1
(Nyquist’s stability theorem) Assume that the open-loop transfer function G(s) has P unstable poles and that they are all contained in the interior of the Nyquist contour \(\varGamma \). The closed-loop transfer function H(s) is stable if and only if the total number of clockwise encirclements of the critical point \(-1\) by G(s), when s follows \(\varGamma \) in the clockwise direction, is equal to \(-P\). In particular, if G(s) is stable, then H(s) is stable if and only if the Nyquist plot of G(s) does not encircle the critical point.
In fact, this result is directly related to the argument principle of Cauchy, which applies to any meromorphic function. For this reason, the assumption that G(s) is rational is not essential and this result can also be applied to transfer functions that contain delays. We refer the reader to Curtain and Zwart (1995) for a detailed account on this question, and to Haidar et al. (2014) for an example on how such extended results can be applied in the case of the basal ganglia dynamics. A particularly interesting case, for which the application of the previous criterion is trivial, is the small-gain case.
Corollary 1
(Small-gain theorem) Assuming that the open-loop transfer function G(s) is stable and that \(\Vert G \Vert _\infty < 1\), then the closed-loop transfer function H(s) is also stable.
Indeed, when the gain of G is smaller than 1, the Nyquist locus is contained inside the unit circle and thus cannot encircle the critical point. The interested reader will find in (Aström and Murray 2010, Chap. 9) an accessible introduction to the two previous classical results of control theory, and in Curtain and Zwart (1995) a detail mathematical account that in particular, covers the case of non-rational transfer functions. In order to illustrate them with a simple example, we consider the following transfer function
When \(\zeta >0\), this transfer function does not have any unstable pole. Since \(P=0\), the closed-loop function H(s) will be stable if and only if the Nyquist plot of G(s) does not encircle the critical point. The curves obtained for \(\zeta =1/2\) and \(\zeta =1/6\) are shown in Fig. 8. In the first case H(s) is stable, while in the second it is unstable.
One of the main strengths of the Nyquist criterion is that in addition to the information it provides about whether a system is stable, it also gives an indication on the robustness of the stability, with respect to different perturbations. In order to explain this point, one has to introduce two additional concepts. To any transfer function G, one can associate its gain \(\gamma _G(\omega )\) and phase \(\varphi _G(\omega )\), at a given frequency \(\omega \ge 0\), which are defined by the relations
In control theory, the gain and phase of the system are often represented graphically either as a Bode diagram (the gain and the phase are plotted using a logarithmic scale for the frequencies) or as a Nyquist diagram (the gain and the phase are represented in polar coordinates, like in Fig. 8). In our approach, the case where the function \(\gamma _G\) is strictly decreasing is of a particular interest. Indeed, in this case, to any strictly proper transfer function G such that \(\gamma _G(0) > 0\) we can associate its (gain) crossover frequency \(\omega _G\), which is defined as the only frequency such that
This frequency can be used to define the delay margin \(\varDelta (G)\) by the relation
If \(\gamma _G\) is strictly decreasing but \(\gamma _G(0) \le 0\), if G is stable, then we can still define \(\omega _G = +\infty \). The case of non-monotonic gains is more complicated. Indeed, in this case, the delay margin might not be unique. We refer the interested reader the survey paper of Sipahi et al. (2011) for a more detailed introduction to systems with time-delays.
In order to illustrate the application of the Nyquist criterion for quantifying the robustness of stability, we consider again the transfer function G(s) of Eq. (12) but, this time, with a delay d in the loop (see Fig. 9). When \(\zeta =1/3\), the system is stable when \(d=0\). Nevertheless, there is a single strictly positive frequency \(\omega _G\) such that \(\gamma _G(\omega _G)=0\). This frequency defines the phase margin \(\pi +\varphi _G\left( \omega _G\right) \) and the delay margin \(\varDelta (G)\) defined above. When the delay is such that \(d \ge \varDelta (G)\), the system becomes unstable (see Fig. 10).
Appendix 2: Mathematical proofs
Proof of Proposition 1
We recall that the transfer function \(H_s\) is given by
where
Remark that reducing \(\Vert H_s\Vert _{\infty }\) amounts to increasing \(\Vert D\Vert _{\infty }\). The computation of \(\Vert D\Vert _{\infty }\) is complicated because of the presence of delay in D(s). For this reason, we introduce the function
The proof is decomposed in five steps. The first step consists in comparing the magnitude of the transfer functions D and \(D_0\). This is done with the help of a function \(D_1\), introduced below. In the second step, we compute the magnitudes of \(D_0\) and \(D_1\). The third step consists in providing a lower bound and an upper bound on \(\left| D_0(s)\right| \) and \(\left| D_1(s)\right| \), respectively. In the fourth step, we give an estimate of \(\Vert D\Vert _{\infty }\) when the filter frequency \(\omega _p\) is sufficiently close to zero. By the last step, we conclude the proof.
Step i Let \(s=j\omega \), where \(\omega \in \mathbb {R}\). Using the triangle inequality, we have
from which we obtain that
From (Doyle et al. 1990, Sect. 4), we know that for each \(d>0\), there exist two real positive numbers \(\alpha \) and \(\beta \), such that the following inequality holds
for all \(\omega \in \mathbb {R}\). Thanks to this property, the delay d will not be involved in our estimation of \(\Vert H_s\Vert _{\infty }\). Now, introduce the function
Inequality (16) then implies that
Step ii A direct computation shows that the magnitude of \(D_0(j\omega )\) and \(D_1(j\omega )\) are given by
and
Thus, the expressions of \(|D_0(j\omega )|^2\) and \(|D_1(j\omega )|^2\) can be rewritten as
where \(h_0(\cdot )\) and \(h_1(\cdot )\) are given by
Step iii The functions \(h_0\) and \(h_1\) have the same expression of h, with parameters summarized in Table 2. To compute their extrema, we rely on the following fact:
Consider a rational function \(h:\mathbb {R}^+\rightarrow \mathbb {R}^+\), defined as
where \(a_1,a_3, a_4\) and \(a_5\) are positive constants and \(a_2\ge 0\). If \(a_2a_4<a_1a_5\) then \(\max _{x\ge 0}h(x)=h(\bar{x})\) with
Now, let
One can verify that if \(\omega _p<\omega _a\), then the condition \(a_2a_4<a_1a_5\) is well satisfied in each case. This implies that we have \(\max _{x\ge 0} h_0(x)=h_0(\bar{x}_0)\) and \(\max _{x\ge 0} h_1(x)=h_1(\bar{x}_1)\), where \(\bar{x}_0\) and \(\bar{x}_1\) are calculated using formula (19), and they are given by
and
By replacing \(\bar{x}_0\) and \(\bar{x}_1\) by their values in \(h_0(\cdot )\) and \(h_1(\cdot )\), we obtain
and
where
Therefore, from Eq. (18), we have
Step iv Let
Equation (17) together with (23) implies that
One can check that
which imply that
Step v Recalling that \(\Vert H_s\Vert _{\infty }=\dfrac{\sigma _s^{\star }}{\Vert D\Vert _{\infty }}\), we deduce that for each delay \(d>0\), each firing rate reference \(\bar{x}_s \in [0,M_s]\), and each gain \(k_p>0\), there exists \(\omega _a>0\) such that for every \(\omega _p<\omega _a\), Eq. (9) holds, which concludes the proof.\(\square \)
Proof of Proposition 2
We recall that the transfer function \(H_g\) is given by
where
We want to estimate \(\Vert H_g\Vert _{\infty }\). Remark that reducing \(\Vert H_g\Vert _{\infty }\) amounts to increasing \(\Vert E\Vert _{\infty }\). As in Proposition 1, we introduce the auxiliary function
Let \(s=j\omega \), where \(\omega \in \mathbb {R}\). Using the triangle inequality, we have
One can check that for all \(\omega \in \mathbb {R}\), we have \(\left| E_0(j\omega )\right| \ge 1\). Equation (25) together with the fact that \(\sigma _g\) is the maximal slope of the activation function \(F_g\) leads to the following
This implies that \(\Vert E\Vert _{\infty }\ge 1-\sigma _g w_{gg}\). Recalling that \(\Vert H_g\Vert _{\infty }=\dfrac{\sigma _g^{\star }}{\Vert E\Vert _{\infty }} \le \dfrac{\sigma _g}{\Vert E\Vert _{\infty }}\), we deduce that for each firing rate reference \(\bar{x}_s \in [0,M_s]\) and each gain \(k_p>0\), Eq. (10) holds if \(\sigma _g w_{gg}<1\), which concludes the proof.\(\square \)
Proof of Proposition 3
The proof is split into two steps. In the first step, we prove that for sufficiently large values of \(k_p\), the equilibrium point \(x_s^\star \) is close to the reference \(\bar{x}_s\). The second step consists in showing that reducing the value of \(\sigma ^{\star }_s\) can be made as small as we want by picking \(\bar{x}_s\) near to 0 or \(M_s\), provided that \(k_p\) is sufficiently large.
Step i Following the lines of the proof of [Pasillas-Lépine 2013, Theorem 1], we affirm that system (1) has a unique equilibrium point \((x^{\star }_s,x^{\star }_g)\in [0,M_s]\times [0,M_g]\) solution of
We see clearly, from Eq. (26), that this equilibrium depends on \(k_p\). Let \(\bar{x}_s\in [0,M_s]\). We have that \(\lim \nolimits _{k_p\rightarrow +\infty }x_s^{\star }(k_p)=\bar{x}_s.\) To see this, it is sufficient to prove that \(\limsup \nolimits _{k_p \rightarrow {+\infty }}x_s^{\star }(k_p)=\liminf \nolimits _{k_p \rightarrow {+\infty }}x_s^{\star }(k_p)=\bar{x}_s\).
Suppose, by contradiction, that \(\limsup \nolimits _{k_p \rightarrow {+\infty }}x_s^{\star }(k_p)<\bar{x}_s\), i.e. \(\limsup \nolimits _{k_p \rightarrow {+\infty }}k_p(\bar{x}_s-x_s^{\star }(k_p))=+\infty \).
Recalling that the activation function \(F_s\) is increasing with a supremum equal to \(M_s\), we obtain, from (26), that \(\limsup \nolimits _{k_p \rightarrow {+\infty }}x_s^{\star }(k_p)=M_s\). This leads to conclude that \(\bar{x}_s>M_s\). In a similar way, if we suppose that \(\limsup \nolimits _{k_p \rightarrow {+\infty }}x_s^{\star }(k_p)>\bar{x}_s\), we obtain that \(\bar{x}_s<0\), which leads to a contradiction.
Thus, we have \(\lim \nolimits _{k_p \rightarrow {+\infty }}x_s^{\star }(k_p)=\bar{x}_s\), as claimed.
Step ii In view of (7), the expression of \(\sigma ^{\star }_s\) is given by
We are going to show that if \(\bar{x}_s\) is sufficiently close to either 0 or \(M_s\), then \(\left| u^{\star }\right| \) tends to \(+\infty \) when \(k_p\) tends to \(+\infty \). This will imply that \(\sigma ^{\star }_s\) tends to zero since (2) guaranties that
From Eq. (26), we have
where \(F^{-1}_s\) denotes the inverse function of \(F_s\). Adding and subtracting \(F^{-1}_s(\bar{x}_s)\) to Eq. (30) and applying the triangle inequality, we obtain that
In view of (29), for every \(\epsilon >0\) there exists a \(\delta _\epsilon >0\) such that \(u_1>\dfrac{1}{\epsilon }\) as soon as either \(0 < \bar{x}_s < \delta _\epsilon \) or \(M_s-\delta _\epsilon < \bar{x}_s < M_s\). Furthermore, knowing that \(F^{-1}_s\) is continuous and using Step (i), we state that for this \(\epsilon >0 \), there exists \(k_{\epsilon }>0\) such that if \(k_p>k_\epsilon \), then \(u_2<\epsilon .\) Noticing that \(u_3\) is bounded, we conclude [in view of (28)] that for every \(\epsilon >0 \), there exist \(\bar{x}_\epsilon >0\) and \(k_{\epsilon }>0\) such that if \(k_p>k_\epsilon \) and \(\bar{x}_s\in \left( 0,\bar{x}_\epsilon \right) \cup \left( M_s-\bar{x}_\epsilon ,M_s\right) \), then \(\sigma _s^\star <\epsilon \), which concludes the proof.\(\square \)
Proof of Proposition 4
The proof is split into two steps. In the first step, we give explicitly the delay margin of stability of the transfer function \(H_s\). In the second step, we prove that this delay margin is near \(+\infty \) when \(\omega _p\) is close to zero.
Step i The transfer function \(H_s\) can be reformulated as
where
The stability of the closed-loop transfer function \(H_s(s)\) can be determined from the characteristics of its open-loop transfer function \(G(s):=C(s)G_s(s)\) (see, for example, Curtain and Zwart 1995). Considering the expression of G(s), which is given by
we see clearly that its gain is strictly decreasing. The latter means that for each firing rate reference \(\bar{x}_s\), each constant \(k_p\), and each filter frequency \(\omega _p\), there exists a unique delay margin \(\varDelta \) (calculated from the expression of G) such that \(H_s\) is stable if and only if \(d<\varDelta \) (see, for example, Middleton and Miller 2007). This delay margin can be computed analytically.
For simplicity of further calculations, we introduce the following notations:
and
We can distinguish, from (Pasillas-Lépine 2013, Corollary 1), two different cases:
-
If \(k_p\sigma _s^{\star }<1\), then \(\varDelta =+\infty \).
-
If \(k_p\sigma _s^{\star }\ge 1\), then the delay margin of the transfer function \(H_s\) is given by
$$\begin{aligned} \varDelta =\dfrac{\pi +\phi _0}{\omega _0}, \end{aligned}$$with (observe that \(c_0\le 0\))
$$\begin{aligned} \omega _0=\sqrt{\dfrac{-b_0+\sqrt{b_0^2-4a_0c_0}}{2a_0}}, \end{aligned}$$and
$$\begin{aligned} \phi _0=\arctan (-\tau _s\omega _0)+\arctan \left( -\dfrac{\omega _0}{\omega _p}\right) . \end{aligned}$$(32)
Step ii In the case when \(k_p\sigma _s^{\star }\ge 1\), one can show that
To see this, it is enough to estimate the limit when \(\omega _p\) tends to \(+\infty \) of \(\omega _0\) and \(\dfrac{\omega _0}{\omega _p}\). We have
Noticing that \(\lim \nolimits _{\omega _p\rightarrow 0}b_0=+\infty \) and that \(c_0\) does not depend on \(\omega _p\), we obtain that
On the other hand, we have
Observing that
we thus have
from which we conclude that
Thus, in view of (32), (34), and (35), we conclude that Eq. (33) is satisfied. Then, for each delay \(d>0\), each firing rate reference \(\bar{x}_s\), and each gain \(k_p>0\), there exists a frequency \(\omega _b>0\) such that the transfer function \(H_{s}\) is stable for every filter frequency \(\omega _p>0\) such that \(\omega _p<\omega _b\), which concludes the proof.\(\square \)
Rights and permissions
About this article
Cite this article
Haidar, I., Pasillas-Lépine, W., Chaillet, A. et al. Closed-loop firing rate regulation of two interacting excitatory and inhibitory neural populations of the basal ganglia. Biol Cybern 110, 55–71 (2016). https://doi.org/10.1007/s00422-015-0678-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-015-0678-y