Abstract
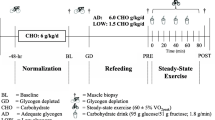
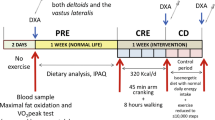
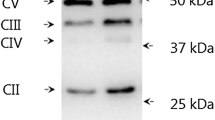
The present study aimed at comparing the responses of myogenic regulatory factors and signaling pathways involved in muscle protein synthesis after a resistance training session performed in either the fasted or fed state. According to a randomized crossover study design, six young male subjects participated in two experimental sessions separated by 3 weeks. In each session, they performed a standardized resistance training. After the sessions, they received during a 4-h recovery period 6 ml/kg b.w. h of a solution containing carbohydrates (50 g/l), protein hydrolysate (33 g/l), and leucine (16.6 g/l). On one occasion, the resistance exercise session was performed after the intake of a carbohydrate-rich breakfast (B), whereas in the other session they remained fasted (F). Needle biopsies from m. vastus lateralis were obtained before (Rest), and 1 h (+1h) and 4 h (+4h) after exercise. Myogenin, MRF4, and MyoD1 mRNA contents were determined by RT-PCR. Phosphorylation of PKB (protein kinase B), GSK3, p70s6k (p70 ribosomal S6 kinase), eIF2B, eEF2 (eukaryotic elongation factor 2), ERK1/2, and p38 was measured via western blotting. Compared with F, the pre-exercise phosphorylation states of PKB and p70s6k were higher in B, whereas those of eIF2B and eEF2 were lower. During recovery, the phosphorylation state of p70s6k was lower in B than in F (p = 0.02). There were no differences in basal mRNA contents between B and F. However, compared with F at +1h, MyoD1 and MRF4 mRNA contents were lower in B (p < 0.05). Our results indicate that prior fasting may stimulate the intramyocellular anabolic response to ingestion of a carbohydrate/protein/leucine mixture following a heavy resistance training session.






Similar content being viewed by others
References
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, Hemmings BA (1996) Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 15:6541–6551
Baar K, Esser K (1999) Phosphorylation of p70(S6k) correlates with increased skeletal muscle mass following resistance exercise. Am J Physiol 276:C120–C127
Biolo G, Declan Fleming RY, Wolfe RR (1995) Physiologic hyperinsulinemia stimulates protein synthesis and enhances transport of selected amino acids in human skeletal muscle. J Clin Invest 95:811–819
Borsheim E, Cree MG, Tipton KD, Elliott TA, Aarsland A, Wolfe RR (2004) Effect of carbohydrate intake on net muscle protein synthesis during recovery from resistance exercise. J Appl Physiol 96:674–678
Byfield MP, Murray JT, Backer JM (2005) hVps34 is a nutrient-regulated lipid kinase required for activation of p70 S6 kinase. J Biol Chem 280:33076–33082
Civitarese AE, Hesselink MK, Russell AP, Ravussin E, Schrauwen P (2005) Glucose ingestion during exercise blunts exercise-induced gene expression of skeletal muscle fat oxidative genes. Am J Physiol Endocrinol Metab 289:E1023–E1029
Cluberton LJ, McGee SL, Murphy RM, Hargreaves M (2005) Effect of carbohydrate ingestion on exercise-induced alterations in metabolic gene expression. J Appl Physiol 99:1359–1363
Cuthbertson DJ, Babraj J, Smith K, Wilkes E, Fedele MJ, Esser K, Rennie M (2006) Anabolic signaling and protein synthesis in human skeletal muscle after dynamic shortening or lengthening exercise. Am J Physiol Endocrinol Metab 290:E731–E738
De Bock K, Richter EA, Russell AP, Eijnde BO, Derave W, Ramaekers M, Koninckx E, Leger B, Verhaeghe J, Hespel P (2005) Exercise in the fasted state facilitates fibre type-specific intramyocellular lipid breakdown and stimulates glycogen resynthesis in humans. J Physiol 564:649–660
Deldicque L, Atherton P, Patel R, Theisen D, Nielens H, Rennie MJ, Francaux M (2008) Decrease in Akt/PKB signalling in human skeletal muscle by resistance exercise. Eur J Appl Physiol 104:57–65
Dreyer HC, Fujita S, Cadenas JG, Chinkes DL, Volpi E, Rasmussen BB (2006) Resistance exercise increases AMPK activity and reduces 4E-BP1 phosphorylation and protein synthesis in human skeletal muscle. J Physiol 576:613–624
Fujita S, Rasmussen BB, Cadenas JG, Grady JJ, Volpi E (2006) Effect of insulin on human skeletal muscle protein synthesis is modulated by insulin-induced changes in muscle blood flow and amino acid availability. Am J Physiol Endocrinol Metab 291:E745–E754
Fujita S, Dreyer HC, Drummond MJ, Glynn EL, Volpi E, Rasmussen BB (2009) Essential amino acid and carbohydrate ingestion before resistance exercise does not enhance postexercise muscle protein synthesis. J Appl Physiol 106:1730–1739
Furuya N, Yu J, Byfield M, Pattingre S, Levine B (2005) The evolutionarily conserved domain of Beclin 1 is required for Vps34 binding, autophagy and tumor suppressor function. Autophagy 1:46–52
Greenhaff PL, Karagounis LG, Peirce N, Simpson EJ, Hazell M, Layfield R, Wackerhage H, Smith K, Atherton P, Selby A, Rennie MJ (2008) Disassociation between the effects of amino acids and insulin on signaling, ubiquitin ligases, and protein turnover in human muscle. Am J Physiol Endocrinol Metab 295:E595–E604
Hesselink MK, Minnaard R, Schrauwen P (2006) Eat the meat or feed the meat: protein turnover in remodeling muscle. Curr Opin Clin Nutr Metab Care 9:672–676
Ishido M, Kami K, Masuhara M (2004) Localization of MyoD, myogenin and cell cycle regulatory factors in hypertrophying rat skeletal muscles. Acta Physiol Scand 180:281–289
Jacobs-El J, Zhou MY, Russell B (1995) MRF4, Myf-5, and myogenin mRNAs in the adaptive responses of mature rat muscle. Am J Physiol 268:C1045–C1052
Judelson DA, Maresh CM, Yamamoto LM, Farrell MJ, Armstrong LE, Kraemer WJ, Volek JS, Spiering BA, Casa DJ, Anderson JM (2008) Effect of hydration state on resistance exercise-induced endocrine markers of anabolism, catabolism, and metabolism. J Appl Physiol 105:816–824
Karlsson HK, Nilsson PA, Nilsson J, Chibalin AV, Zierath JR, Blomstrand E (2004) Branched-chain amino acids increase p70S6k phosphorylation in human skeletal muscle after resistance exercise. Am J Physiol Endocrinol Metab 287:E1–E7
Koopman R, Wagenmakers AJ, Manders RJ, Zorenc AH, Senden JM, Gorselink M, Keizer HA, van Loon LJ (2005) Combined ingestion of protein and free leucine with carbohydrate increases postexercise muscle protein synthesis in vivo in male subjects. Am J Physiol Endocrinol Metab 288:E645–E653
Koopman R, Pennings B, Zorenc AH, van Loon LJ (2007) Protein ingestion further augments S6K1 phosphorylation in skeletal muscle following resistance type exercise in males. J Nutr 137:1880–1886
Larsen AE, Tunstall RJ, Carey KA, Nicholas G, Kambadur R, Crowe TC, Cameron-Smith D (2006) Actions of short-term fasting on human skeletal muscle myogenic and atrogenic gene expression. Ann Nutr Metab 50:476–481
Li H, Capetanaki Y (1993) Regulation of the mouse desmin gene: transactivated by MyoD, myogenin, MRF4 and Myf5. Nucleic Acids Res 21:335–343
Lin H, Yutzey KE, Konieczny SF (1991) Muscle-specific expression of the troponin I gene requires interactions between helix-loop-helix muscle regulatory factors and ubiquitous transcription factors. Mol Cell Biol 11:267–280
MacKenzie MG, Hamilton DL, Murray JT, Taylor PM, Baar K (2009) mVps34 is activated following high-resistance contractions. J Physiol 587:253–260
Megeney LA, Rudnicki MA (1995) Determination versus differentiation and the MyoD family of transcription factors. Biochem Cell Biol 73:723–732
Miller BF, Olesen JL, Hansen M, Dossing S, Crameri RM, Welling RJ, Langberg H, Flyvbjerg A, Kjaer M, Babraj JA, Smith K, Rennie MJ (2005) Coordinated collagen and muscle protein synthesis in human patella tendon and quadriceps muscle after exercise. J Physiol 567:1021–1033
Moore DR, Robinson MJ, Fry JL, Tang JE, Glover EI, Wilkinson SB, Prior T, Tarnopolsky MA, Phillips SM (2009) Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am J Clin Nutr 89:161–168
Morck C, Pilon M (2007) Caloric restriction and autophagy in Caenorhabditis elegans. Autophagy 3:51–53
Muscat GE, Dressel U (2000) Not a minute to waste. Nat Med 6:1216–1217
Psilander N, Damsgaard R, Pilegaard H (2003) Resistance exercise alters MRF and IGF-I mRNA content in human skeletal muscle. J Appl Physiol 95:1038–1044
Rasmussen BB, Tipton KD, Miller SL, Wolf SE, Wolfe RR (2000) An oral essential amino acid–carbohydrate supplement enhances muscle protein anabolism after resistance exercise. J Appl Physiol 88:386–392
Rennie MJ, Bohe J, Wolfe RR (2002) Latency, duration and dose response relationships of amino acid effects on human muscle protein synthesis. J Nutr 132:3225S–3227S
Schneiter P, Di Vetta V, Jequier E, Tappy L (1995) Effect of physical exercise on glycogen turnover and net substrate utilization according to the nutritional state. Am J Physiol 269:E1031–E1036
Stewart CE, Rittweger J (2006) Adaptive processes in skeletal muscle: molecular regulators and genetic influences. J Musculoskelet Neuronal Interact 6:73–86
Tang JE, Perco JG, Moore DR, Wilkinson SB, Phillips SM (2008) Resistance training alters the response of fed state mixed muscle protein synthesis in young men. Am J Physiol Regul Integr Comp Physiol 294:R172–R178
Terzis G, Georgiadis G, Stratakos G, Vogiatzis I, Kavouras S, Manta P, Mascher H, Blomstrand E (2008) Resistance exercise-induced increase in muscle mass correlates with p70S6 kinase phosphorylation in human subjects. Eur J Appl Physiol 102:145–152
Tipton KD, Jeukendrup AE, Hespel P (2007) Nutrition for the sprinter. J Sports Sci 25(Suppl 1):S5–S15
van Loon LJ, Kruijshoop M, Verhagen H, Saris WH, Wagenmakers AJ (2000) Ingestion of protein hydrolysate and amino acid–carbohydrate mixtures increases postexercise plasma insulin responses in men. J Nutr 130:2508–2513
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Vissing K, Andersen JL, Schjerling P (2005) Are exercise-induced genes induced by exercise? FASEB J 19:94–96
Welle S, Bhatt K, Thornton CA (1999) Stimulation of myofibrillar synthesis by exercise is mediated by more efficient translation of mRNA. J Appl Physiol 86:1220–1225
Wilkinson SB, Phillips SM, Atherton PJ, Patel R, Yarasheski KE, Tarnopolsky MA, Rennie MJ (2008) Differential effects of resistance and endurance exercise in the fed state on signalling molecule phosphorylation and protein synthesis in human muscle. J Physiol 586:3701–3717
Williamson D, Gallagher P, Harber M, Hollon C, Trappe S (2003) Mitogen-activated protein kinase (MAPK) pathway activation: effects of age and acute exercise on human skeletal muscle. J Physiol 547:977–987
Witard OC, Tieland M, Beelen M, Tipton KD, van Loon LJ, Koopman R (2009) Resistance exercise increases postprandial muscle protein synthesis in humans. Med Sci Sports Exerc 41:144–154
Yang Y, Creer A, Jemiolo B, Trappe S (2005) Time course of myogenic and metabolic gene expression in response to acute exercise in human skeletal muscle. J Appl Physiol 98:1745–1752
Acknowledgments
This study was supported by grants from the Onderzoeksraad K.U.-Leuven (grant # OT04/45), the Fonds voor Wetenschappelijk Onderzoek Vlaanderen (grant # G.0233.05) and the Fonds de la Recherche Scientifique Médicale (grant # 3.4574.03). Hyprol 4107 was kindly provided by Kerry Bio-Science, Utrecht, the Netherlands. Louise Deldicque is supported by the «Fonds National de la Recherche Scientifique (FNRS)».
Conflict of interest statement
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Håkan Westerblad.
L. Deldicque and K. De Bock contributed equally to this work.
Rights and permissions
About this article
Cite this article
Deldicque, L., De Bock, K., Maris, M. et al. Increased p70s6k phosphorylation during intake of a protein–carbohydrate drink following resistance exercise in the fasted state. Eur J Appl Physiol 108, 791–800 (2010). https://doi.org/10.1007/s00421-009-1289-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-009-1289-x