Abstract
Purpose
This meta-analysis was conducted to evaluate the efficacy and safety of single-dose dexamethasone implantation for treating persistent DME (diabetic macular edema) refractory to anti-VEGF (anti-vascular endothelial growth factor) drugs over a period of 6 months.
Methods
All related clinical trials were reviewed by searching electronic databases of PubMed, Medline, Web of Science, Cochrane Library, and EMBASE. The primary outcome parameters were best-corrected visual acuity (BCVA) and central macular thickness (CMT). We performed this meta-analysis by using Stata15.0.
Results
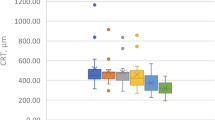
Ten clinical trials involving 362 eyes from 328 patients were eligible in the final analysis. After single-dose dexamethasone implantation, there was a significant improvement in BCVA from baseline to 1, 3, and 6 months with an average increase of − 0.15 logMAR (p < 0.001), − 0.14 logMAR (p < 0.001), and − 0.07 logMAR (p = 0.004), respectively. Further, mean CMT decreased significantly with an average reduction of 249.18 μm (p < 0.001), 217.66 μm (p < 0.001), and 91.56 μm (p < 0.001) at months 1, 3, and 6, respectively.
Conclusions
Our results indicate that switching to a dexamethasone implant could achieve significant anatomical and functional improvement among patients with refractory DME. Clinicians should be aware of this treatment option in refractory DME.




Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Browning DJ, Stewart MW, Lee C (2018) Diabetic macular edema: evidence-based management. Indian J of Ophthalmol 66(12):1736–1750. https://doi.org/10.4103/ijo.IJO_1240_18
Cavan D, Makaroff L, da Rocha FJ, Sylvanowicz M, Ackland P, Conlon J, Chaney D, Malhi A, Barratt J (2017) The Diabetic Retinopathy Barometer Study: global perspectives on access to and experiences of diabetic retinopathy screening and treatment. Diabetes Res Clin Pract 129:16–24. https://doi.org/10.1016/j.diabres.2017.03.023
Yau JW, Rogers SL, Kawasaki R, Lamoureux EL, Kowalski JW, Bek T, Chen SJ, Dekker JM, Fletcher A, Grauslund J, Haffner S, Hamman RF, Ikram MK, Kayama T, Klein BE, Klein R, Krishnaiah S, Mayurasakorn K, O’Hare JP, Orchard TJ, Porta M, Rema M, Roy MS, Sharma T, Shaw J, Taylor H, Tielsch JM, Varma R, Wang JJ, Wang N, West S, Xu L, Yasuda M, Zhang X, Mitchell P, Wong TY (2012) Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 35(3):556–564. https://doi.org/10.2337/dc11-1909
Varma R, Torres M, Peña F, Klein R, Azen SP (2004) Prevalence of diabetic retinopathy in adult Latinos: the Los Angeles Latino eye study. Ophthalmology 111(7):1298–1306. https://doi.org/10.1016/j.ophtha.2004.03.002
Varma R, Wen G, Jiang X, Hsu C, Torres M, Klein R, Azen SP, McKean-Cowdin R (2016) Prevalence of diabetic retinopathy in adult Chinese American individuals: the Chinese American eye study. JAMA Ophthalmol 134(5):563–569. https://doi.org/10.1001/jamaophthalmol.2016.0445
Schmidt-Erfurth U, Garcia-Arumi J, Bandello F, Berg K, Chakravarthy U, Gerendas BS, Jonas J, Larsen M, Tadayoni R, Loewenstein A (2017) Guidelines for the management of diabetic macular edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica 237(4):185–222. https://doi.org/10.1159/000458539
Nguyen QD, Brown DM, Marcus DM, Boyer DS, Patel S, Feiner L, Gibson A, Sy J, Rundle AC, Hopkins JJ, Rubio RG, Ehrlich JS (2012) Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology 119(4):789–801. https://doi.org/10.1016/j.ophtha.2011.12.039
Do DV, Nguyen QD, Boyer D, Schmidt-Erfurth U, Brown DM, Vitti R, Berliner AJ, Gao B, Zeitz O, Ruckert R, Schmelter T, Sandbrink R, Heier JS (2012) One-year outcomes of the da Vinci Study of VEGF trap-eye in eyes with diabetic macular edema. Ophthalmology 119(8):1658–1665. https://doi.org/10.1016/j.ophtha.2012.02.010
Ophir A (2017) Early and long-term responses to anti-vascular endothelial growth factor therapy in diabetic macular edema: analysis of protocol I data. Am J Ophthalmol 177:230–231. https://doi.org/10.1016/j.ajo.2016.11.025
Brown DM, Nguyen QD, Marcus DM, Boyer DS, Patel S, Feiner L, Schlottmann PG, Rundle AC, Zhang J, Rubio RG, Adamis AP, Ehrlich JS, Hopkins JJ (2013) Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology 120(10):2013–2022. https://doi.org/10.1016/j.ophtha.2013.02.034
Schmidt-Erfurth U, Lang GE, Holz FG, Schlingemann RO, Lanzetta P, Massin P, Gerstner O, Bouazza AS, Shen H, Osborne A, Mitchell P (2014) Three-year outcomes of individualized ranibizumab treatment in patients with diabetic macular edema: the RESTORE extension study. Ophthalmology 121(5):1045–1053. https://doi.org/10.1016/j.ophtha.2013.11.041
Iglicki M, Busch C, Zur D, Okada M, Mariussi M, Chhablani JK, Cebeci Z, Fraser-Bell S, Chaikitmongkol V, Couturier A, Giancipoli E, Lupidi M, Rodríguez-Valdés PJ, Rehak M, Fung AT, Goldstein M, Loewenstein A (2019) Dexamethasone implant for diabetic macular edema in naïve compared with refractory eyes: the International Retina Group Real-Life 24-Month Multicenter Study. The IRGREL-DEX Study. Retina (Philadelphia, Pa) 39(1):44–51. https://doi.org/10.1097/iae.0000000000002196
Zarranz-Ventura J, Romero-Núñez B, Bernal-Morales C, Velazquez-Villoria D, Sala-Puigdollers A, Figueras-Roca M, Copete S, Distefano L, Boixadera A, García-Arumi J, Adan A (2020) Differential response to intravitreal dexamethasone implant in naïve and previously treated diabetic macular edema eyes. BMC Ophthalmol 20(1):443. https://doi.org/10.1186/s12886-020-01716-2
Zhang X, Bao S, Lai D, Rapkins RW, Gillies MC (2008) Intravitreal triamcinolone acetonide inhibits breakdown of the blood-retinal barrier through differential regulation of VEGF-A and its receptors in early diabetic rat retinas. Diabetes 57(4):1026–1033. https://doi.org/10.2337/db07-0982
Chang-Lin JE, Attar M, Acheampong AA, Robinson MR, Whitcup SM, Kuppermann BD, Welty D (2011) Pharmacokinetics and pharmacodynamics of a sustained-release dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci 52(1):80–86. https://doi.org/10.1167/iovs.10-5285
Boyer DS, Yoon YH, Belfort R Jr, Bandello F, Maturi RK, Augustin AJ, Li XY, Cui H, Hashad Y, Whitcup SM (2014) Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology 121(10):1904–1914. https://doi.org/10.1016/j.ophtha.2014.04.024
Wecker T, Grundel B, Grundel M, Bründer MC, Trick S, Lange C, Böhringer D, Agostini H, Stahl A (2021) Real-life medium term follow-up data for intravitreal dexamethasone implant in retinal vein occlusion. Sci Rep 11(1):8303. https://doi.org/10.1038/s41598-021-87467-6
Lowder C, Belfort R Jr, Lightman S, Foster CS, Robinson MR, Schiffman RM, Li XY, Cui H, Whitcup SM (2011) Dexamethasone intravitreal implant for noninfectious intermediate or posterior uveitis. Arch Ophthalmol 129(5):545–553. https://doi.org/10.1001/archophthalmol.2010.339
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535. https://doi.org/10.1136/bmj.b2535
Downs SH, Black N (1998) The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health 52(6):377–384. https://doi.org/10.1136/jech.52.6.377
Hooper P, Jutai JW, Strong G, Russell-Minda E (2008) Age-related macular degeneration and low-vision rehabilitation: a systematic review. Can J Ophthalmol 43(2):180–187. https://doi.org/10.3129/i08-001
Castro-Navarro V, Cervera-Taulet E, Navarro-Palop C, Hernández-Bel L, Monferrer-Adsuara C, Mata-Moret L, Montero-Hernández J (2020) Analysis of anatomical biomarkers in subtypes of diabetic macular edema refractory to anti-vascular endothelial growth factor treated with dexamethasone implant. European J Ophthalmol 30(4):764–769. https://doi.org/10.1177/1120672119834182
Bayat AH, Elçioğlu MN (2020) Effects of dexamethasone treatment on serous retinal detachment in ranibizumab-resistant diabetic macular edema. Therapeutic Advances in Ophthalmology 12:2515841420971936https://doi.org/10.1177/2515841420971936
Özata K, Atum M, Çelik E, Doğan E, Alagöz G (2019) Efficacy of intravitreal dexamethasone implant in persistent diabetic macular edema after primary treatment with intravitreal ranibizumab. J Curr Ophthalmol 31(3):281–286. https://doi.org/10.1016/j.joco.2019.03.002
Nalçacı S, Akkın C, Afrashi F (2019) Dexamethasone implant in patients with diabetic macular edema resistant to anti-VEGF therapy. Turk J Ophthalmol 49(2):73–77. https://doi.org/10.4274/tjo.galenos.2018.84665
Yucel OE, Can E, Ozturk HE, Birinci H, Sullu Y (2017) Dexamethasone implant in chronic diabetic macular edema resistant to intravitreal ranibizumab treatment. Ophthalmic Res 57(3):161–165. https://doi.org/10.1159/000452422
Esen E, Sizmaz S, Demircan N (2017) Efficacy of dexamethasone intravitreal implant for the treatment of persistent diffuse diabetic macular edema. Int Ophthalmol 37(1):1–6. https://doi.org/10.1007/s10792-016-0219-2
Totan Y, Güler E, Gürağaç FB (2016) Dexamethasone intravitreal implant for chronic diabetic macular edema resistant to intravitreal bevacizumab treatment. Curr Eye Res 41(1):107–113. https://doi.org/10.3109/02713683.2014.1002048
Kim M, Cho YJ, Lee CH, Lee SC (2016) Effect of intravitreal dexamethasone implant on retinal and choroidal thickness in refractory diabetic macular oedema after multiple anti-VEGF injections. Eye (Lond) 30(5):718–725. https://doi.org/10.1038/eye.2016.23
Arıkan Yorgun M, Toklu Y, Mutlu M, Uysal BS, Çakmak HB (2016) Efficacy of single-dose dexamethasone implantation in patients with persistent diabetic macular edema. Int Ophthalmol 36(4):531–539. https://doi.org/10.1007/s10792-015-0155-6
Zhioua I, Semoun O, Lalloum F, Souied EH (2015) Intravitreal dexamethasone implant in patients with ranibizumab persistent diabetic macular edema. Retina (Philadelphia, Pa) 35(7):1429–1435. https://doi.org/10.1097/iae.0000000000000490
Pacella E, Vestri AR, Muscella R, Carbotti MR, Castellucci M, Coi L, Turchetti P, Pacella F (2013) Preliminary results of an intravitreal dexamethasone implant (Ozurdex®) in patients with persistent diabetic macular edema. Clin Ophthalmol (Auckland, NZ) 7:1423–1428. https://doi.org/10.2147/opth.S48364
Tamura H, Miyamoto K, Kiryu J, Miyahara S, Katsuta H, Hirose F, Musashi K, Yoshimura N (2005) Intravitreal injection of corticosteroid attenuates leukostasis and vascular leakage in experimental diabetic retina. Invest Ophthalmol Vis Sci 46(4):1440–1444. https://doi.org/10.1167/iovs.04-0905
He Y, Ren XJ, Hu BJ, Lam WC, Li XR (2018) A meta-analysis of the effect of a dexamethasone intravitreal implant versus intravitreal anti-vascular endothelial growth factor treatment for diabetic macular edema. BMC Ophthalmol 18(1):121. https://doi.org/10.1186/s12886-018-0779-1
Acknowledgements
The authors sincerely thank Editage (www.editage.cn) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuan, Q., Liu, Y., Xu, H. et al. Efficacy and safety of single-dose dexamethasone implantation for patients with persistent diabetic macular edema: a systematic review and meta-analysis. Graefes Arch Clin Exp Ophthalmol 260, 405–413 (2022). https://doi.org/10.1007/s00417-021-05369-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-021-05369-9