Abstract
Purpose
To investigate peripapillary vessel density at various spatial locations and layers in healthy and normal tension glaucoma eyes using optical coherence tomography angiography (OCTA).
Methods
A commercial OCTA device (AngioPlex; Carl Zeiss Meditec) was used to image microvasculature in a 6 × 6-mm optic disc region. Vessel densities of superficial and deep retinal layers were calculated using an automatic thresholding algorithm. Vessel density maps were plotted by averaging individual angiogram images. The spatial characteristics of vessel densities were analyzed at clock-hour sectors and in five 0.7-mm-thick concentric circles from a diameter of 2.0 to 5.5 mm. Areas under the receiver operating characteristics curves (AUCs) assessed the glaucoma diagnostic ability.
Results
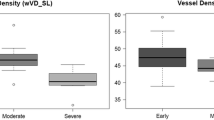
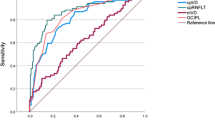
Vessel density maps of superficial and deep retinal layers were significantly reduced at the 7 and 11 o’clock positions in glaucomatous eyes. In superficial layer, vessel density significantly decreased as the distance from the optic disc margin increased, except in the innermost circle (2.0–2.7-mm). There were significant differences in AUCs of superficial vessel density between innermost circle and the other outer circles. In the deep layer, the innermost circle showed significantly higher vessel density than the outer circles. Vessel density at 7 o’clock showed the best diagnostic performance (AUCs, 0.898 and 0.789) both in the superficial and deep layers. The innermost circle showed eccentric feature compared to the outer circles in terms of spatial characteristics and diagnostic ability.
Conclusions
Understanding of the spatial characteristics of peripapillary vasculature may be helpful in clinical practice and determining the optimal measurement area of vessel density.




Similar content being viewed by others
References
Flammer J, Orgul S, Costa VP, Orzalesi N, Krieglstein GK, Serra LM, Renard JP, Stefansson E (2002) The impact of ocular blood flow in glaucoma. Prog Retin Eye Res 21:359–393
Chung HS, Harris A, Kagemann L, Martin B (1999) Peripapillary retinal blood flow in normal tension glaucoma. Br J Ophthalmol 83:466–469
Harris A, Sergott RC, Spaeth GL, Katz JL, Shoemaker JA, Martin BJ (1994) Color Doppler analysis of ocular vessel blood velocity in normal-tension glaucoma. Am J Ophthalmol 118:642–649
Mitchell P, Leung H, Wang JJ, Rochtchina E, Lee AJ, Wong TY, Klein R (2005) Retinal vessel diameter and open-angle glaucoma: the Blue Mountains eye study. Ophthalmology 112:245–250
Wang S, Xu L, Wang Y, Wang Y, Jonas JB (2007) Retinal vessel diameter in normal and glaucomatous eyes: the Beijing eye study. Clin Experiment Ophthalmol 35:800–807
Yu DY, Cringle SJ, Balaratnasingam C, Morgan WH, Yu PK, Su EN (2013) Retinal ganglion cells: energetics, compartmentation, axonal transport, cytoskeletons and vulnerability. Prog Retin Eye Res 36:217–246
Zhang HR (1994) Scanning electron-microscopic study of corrosion casts on retinal and choroidal angioarchitecture in man and animals. Prog Retin Eye Res 13:243–270
Alterman M, Henkind P (1968) Radial peripapillary capillaries of the retina. II. Possible role in Bjerrum scotoma. Br J Ophthalmol 52:26–31
Kornzweig AL, Eliasoph I, Feldstein M (1968) Selective atrophy of the radial peripapillary capillaries in chronic glaucoma. Arch Ophthalmol 80:696–702
Spaide RF, Klancnik JM Jr, Cooney MJ (2015) Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol 133:45–50
Mendis KR, Balaratnasingam C, Yu P, Barry CJ, McAllister IL, Cringle SJ, Yu DY (2010) Correlation of histologic and clinical images to determine the diagnostic value of fluorescein angiography for studying retinal capillary detail. Invest Ophthalmol Vis Sci 51:5864–5869
Akagi T, Iida Y, Nakanishi H, Terada N, Morooka S, Yamada H, Hasegawa T, Yokota S, Yoshikawa M, Yoshimura N (2016) Microvascular density in glaucomatous eyes with Hemifield visual field defects: an optical coherence tomography angiography study. Am J Ophthalmol 168:237–249
Liu L, Jia Y, Takusagawa HL, Pechauer AD, Edmunds B, Lombardi L, Davis E, Morrison JC, Huang D (2015) Optical coherence tomography angiography of the Peripapillary retina in glaucoma. JAMA Ophthalmol 133:1045–1052
Bojikian KD, Chen CL, Wen JC, Zhang Q, Xin C, Gupta D, Mudumbai RC, Johnstone MA, Wang RK, Chen PP (2016) Optic disc perfusion in primary open angle and normal tension glaucoma eyes using optical coherence tomography-based Microangiography. PLoS One 11:e0154691
Yarmohammadi A, Zangwill LM, Diniz-Filho A, Suh MH, Manalastas PI, Fatehee N, Yousefi S, Belghith A, Saunders LJ, Medeiros FA, Huang D, Weinreb RN (2016) Optical coherence tomography angiography vessel density in healthy, glaucoma suspect, and glaucoma eyes. Invest Ophthalmol Vis Sci 57:451–459
Hollo G (2016) Vessel density calculated from OCT angiography in 3 peripapillary sectors in normal, ocular hypertensive, and glaucoma eyes. Eur J Ophthalmol 26:e42–e45
Yu PK, Cringle SJ, Yu DY (2014) Correlation between the radial peripapillary capillaries and the retinal nerve fibre layer in the normal human retina. Exp Eye Res 129:83–92
An L, Johnstone M, Wang RK (2012) Optical microangiography provides correlation between microstructure and microvasculature of optic nerve head in human subjects. J Biomed Opt 17:116018
Zhang A, Zhang Q, Chen CL, Wang RK (2015) Methods and algorithms for optical coherence tomography-based angiography: a review and comparison. J Biomed Opt 20:100901
Shahlaee A, Samara WA, Hsu J, Say EA, Khan MA, Sridhar J, Hong BK, Shields CL, Ho AC (2016) In vivo assessment of macular vascular density in healthy human eyes using optical coherence tomography angiography. Am J Ophthalmol 165:39–46
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Wang Q, Chan S, Yang JY, You B, Wang YX, Jonas JB, Wei WB (2016) Vascular density in retina and choriocapillaris as measured by optical coherence tomography angiography. Am J Ophthalmol
Jia Y, Wei E, Wang X, Zhang X, Morrison JC, Parikh M, Lombardi LH, Gattey DM, Armour RL, Edmunds B, Kraus MF, Fujimoto JG, Huang D (2014) Optical coherence tomography angiography of optic disc perfusion in glaucoma. Ophthalmology 121:1322–1332
Wang X, Jiang C, Ko T, Kong X, Yu X, Min W, Shi G, Sun X (2015) Correlation between optic disc perfusion and glaucomatous severity in patients with open-angle glaucoma: an optical coherence tomography angiography study. Graefes Arch Clin Exp Ophthalmol 253:1557–1564
Henkind P (1967) Radial peripapillary capillaries of the retina. I. Anatomy: human and comparative. Br J Ophthalmol 51:115–123
Scoles D, Gray DC, Hunter JJ, Wolfe R, Gee BP, Geng Y, Masella BD, Libby RT, Russell S, Williams DR, Merigan WH (2009) In-vivo imaging of retinal nerve fiber layer vasculature: imaging histology comparison. BMC Ophthalmol 9:9
Garcia-Valenzuela E, Mori M, Edward DP, Shahidi M (2000) Thickness of the peripapillary retina in healthy subjects with different degrees of ametropia. Ophthalmology 107:1321–1327
Budenz DL, Anderson DR, Varma R, Schuman J, Cantor L, Savell J, Greenfield DS, Patella VM, Quigley HA, Tielsch J (2007) Determinants of normal retinal nerve fiber layer thickness measured by stratus OCT. Ophthalmology 114:1046–1052
Hayreh SS (2004) Posterior ciliary artery circulation in health and disease: the Weisenfeld lecture. Invest Ophthalmol Vis Sci 45:749–757 748
Hayreh SS (1990) In vivo choroidal circulation and its watershed zones. Eye (Lond) 4(Pt 2):273–289
Ross RD, Barofsky JM, Cohen G, Baber WB, Palao SW, Gitter KA (1998) Presumed macular choroidal watershed vascular filling, choroidal neovascularization, and systemic vascular disease in patients with age-related macular degeneration. Am J Ophthalmol 125:71–80
Takahashi K, Muraoka K, Kishi S, Shimizu K (1996) Watershed zone in the human peripheral choroid. Ophthalmology 103:336–342
Mammo Z, Heisler M, Balaratnasingam C, Lee S, Yu DY, Mackenzie P, Schendel S, Merkur A, Kirker A, Albiani D, Navajas E, Beg MF, Morgan W, Sarunic MV (2016) Quantitative optical coherence tomography angiography of radial Peripapillary capillaries in glaucoma, glaucoma suspect, and normal eyes. Am J Ophthalmol 170:41–49
Zhang M, Hwang TS, Campbell JP, Bailey ST, Wilson DJ, Huang D, Jia Y (2016) Projection-resolved optical coherence tomographic angiography. Biomed Opt Express 7:816–828
Zhang A, Zhang Q, Wang RK (2015) Minimizing projection artifacts for accurate presentation of choroidal neovascularization in OCT micro-angiography. Biomed Opt Express 6:4130–4143
Mitchell P, Hourihan F, Sandbach J, Wang JJ (1999) The relationship between glaucoma and myopia: the Blue Mountains eye study. Ophthalmology 106:2010–2015
Perera SA, Wong TY, Tay WT, Foster PJ, Saw SM, Aung T (2010) Refractive error, axial dimensions, and primary open-angle glaucoma: the Singapore Malay eye study. Arch Ophthalmol 128:900–905
Sommer A, Tielsch JM (1996) Risk factors for open-angle glaucoma: the Barbados eye study. Arch Ophthalmol 114:235
Wong TY, Klein BE, Klein R, Knudtson M, Lee KE (2003) Refractive errors, intraocular pressure, and glaucoma in a white population. Ophthalmology 110:211–217
Xu L, Wang Y, Wang S, Wang Y, Jonas JB (2007) High myopia and glaucoma susceptibility the Beijing eye study. Ophthalmology 114:216–220
Shin JW, Shin YU, Lee BR (2012) Choroidal thickness and volume mapping by a six radial scan protocol on spectral-domain optical coherence tomography. Ophthalmology 119:1017–1023
Ho M, Liu DT, Chan VC, Lam DS (2013) Choroidal thickness measurement in myopic eyes by enhanced depth optical coherence tomography. Ophthalmology 120:1909–1914
Bussel II, Wollstein G, Schuman JS (2014) OCT for glaucoma diagnosis, screening and detection of glaucoma progression. Br J Ophthalmol 98(Suppl 2):ii15-19
Mwanza JC, Oakley JD, Budenz DL, Anderson DR (2011) Ability of cirrus HD-OCT optic nerve head parameters to discriminate normal from glaucomatous eyes. Ophthalmology 118(241–248):e241
Bengtsson B, Andersson S, Heijl A (2012) Performance of time-domain and spectral-domain optical coherence tomography for glaucoma screening. Acta Ophthalmol 90:310–315
Kim CS, Seong GJ, Lee NH, Song KC, Namil Study Group KGS (2011) Prevalence of primary open-angle glaucoma in central South Korea the Namil study. Ophthalmology 118:1024–1030
Suh W, Kee C, Namil Study G, Korean Glaucoma S (2012) The distribution of intraocular pressure in urban and in rural populations: the Namil study in South Korea. Am J Ophthalmol 154:99–106
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this research.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Shin, J.W., Sung, K.R., Lee, J.Y. et al. Optical coherence tomography angiography vessel density mapping at various retinal layers in healthy and normal tension glaucoma eyes. Graefes Arch Clin Exp Ophthalmol 255, 1193–1202 (2017). https://doi.org/10.1007/s00417-017-3671-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-017-3671-4