Abstract
Background
The R2 component of blink reflex recovery cycle (R2BRrc) is a simple neurophysiological tool to detect the brainstem hyperexcitability commonly occurring in several neurological diseases such as Parkinson’s disease and atypical parkinsonisms. In our study, we investigated for the first time the usefulness of R2BRrc to assess brainstem excitability in patients with idiopathic Normal Pressure Hydrocephalus (iNPH) in comparison with healthy subjects.
Methods
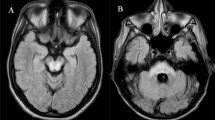
Eighteen iNPH patients and 25 age-matched control subjects were enrolled. R2BRrc was bilaterally evaluated at interstimulus intervals (ISIs) of 100, 150, 200, 300, 400, 500 and 750 ms in all participants. We investigated the diagnostic performance of R2BRrc in differentiating iNPH patients from control subjects using ROC analysis. Midbrain area and Magnetic Resonance Hydrocephalic Index (MRHI), an MRI biomarker for the diagnosis of iNPH, were measured on T1-weighted MR images, and correlations between R2BRrc values and MRI measurements were investigated.
Results
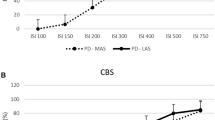
Fourteen (78%) of 18 iNPH patients showed an enhanced R2BRrc at ISIs 100–150–200 ms, while no control subjects had abnormal R2BRrc. The mean amplitude of bilateral R2BRrc at the shortest ISIs (100–150–200 ms) showed high accuracy in differentiating iNPH patients from controls (AUC = 0.89). R2BRrc values significantly correlated with midbrain area and MRHI values.
Conclusions
This study represents the first evidence of brainstem hyperexcitability in iNPH patients. Given its low cost and wide availability, R2BRrc could be a useful tool for selecting elderly subjects with mild gait and urinary dysfunction who should undergo an extensive diagnostic workup for the diagnosis of NPH.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Code availability
Not applicable.
References
Mori E, Ishikawa M, Kato T, Kazui H, Miyake H, Miyajima M, Nakajima M, Hashimoto M, Kuriyama N, Tokuda T, Ishii K, Kaijima M, Hirata Y, Saito M, Arai H, Japanese Society of Normal Pressure H (2012) Guidelines for management of idiopathic normal pressure hydrocephalus: second edition. Neurol Med Chir (Tokyo) 52(11):775–809. https://doi.org/10.2176/nmc.52.775
Graff-Radford NR, Jones DT (2019) Normal pressure hydrocephalus. Continuum (Minneap Minn) 25(1):165–186. https://doi.org/10.1212/CON.0000000000000689
Anger JT, Saigal CS, Stothers L, Thom DH, Rodriguez LV, Litwin MS, Urologic Diseases of America P (2006) The prevalence of urinary incontinence among community dwelling men: results from the National Health and Nutrition Examination survey. J Urol 176(5):2103–2108. https://doi.org/10.1016/j.juro.2006.07.029 (discussion 2108)
Stothers L, Thom D, Calhoun E (2005) Urologic diseases in America project: urinary incontinence in males–demographics and economic burden. J Urol 173(4):1302–1308. https://doi.org/10.1097/01.ju.0000155503.12545.4e
Kimura J (1983) Clinical uses of the electrically elicited blink reflex. Adv Neurol 39:773–786
Basso MA, Evinger C (1996) An explanation for reflex blink hyperexcitability in Parkinson’s disease. II. Nucleus raphe magnus. J Neurosci 16(22):7318–7330
Arabia G, Lupo A, Manfredini LI, Vescio B, Nistico R, Barbagallo G, Salsone M, Morelli M, Novellino F, Nicoletti G, Quattrone A, Cascini GL, Louis ED, Quattrone A (2018) Clinical, electrophysiological, and imaging study in essential tremor-Parkinson’s disease syndrome. Parkinsonism Relat Disord 56:20–26. https://doi.org/10.1016/j.parkreldis.2018.06.005
Sciacca G, Nicoletti A, Mostile G, Luca A, Raciti L, Dibilio V, Drago F, Salomone S, Zappia M (2018) Blink reflex recovery cycle to differentiate progressive supranuclear palsy from corticobasal syndrome. Eur J Neurol 25(8):1100-e1185. https://doi.org/10.1111/ene.13673
Schwingenschuh P, Katschnig P, Edwards MJ, Teo JT, Korlipara LV, Rothwell JC, Bhatia KP (2011) The blink reflex recovery cycle differs between essential and presumed psychogenic blepharospasm. Neurology 76(7):610–614. https://doi.org/10.1212/WNL.0b013e31820c3074
Nistico R, Pirritano D, Salsone M, Valentino P, Novellino F, Condino F, Bono F, Quattrone A (2012) Blink reflex recovery cycle in patients with dystonic tremor: a cross-sectional study. Neurology 78(17):1363–1365. https://doi.org/10.1212/WNL.0b013e3182518316
Nistico R, Salsone M, Vescio B, Morelli M, Trotta M, Barbagallo G, Arabia G, Quattrone A (2014) Blink reflex recovery cycle distinguishes essential tremor with resting tremor from de novo Parkinson’s disease: an exploratory study. Parkinsonism Relat Disord 20(2):153–156. https://doi.org/10.1016/j.parkreldis.2013.10.006
Basso MA, Powers AS, Evinger C (1996) An explanation for reflex blink hyperexcitability in Parkinson’s disease. I. Superior colliculus. J Neurosci 16(22):7308–7317
Sciacca G, Mostile G, Disilvestro I, Donzuso G, Manna R, Portaro G, Rascuna C, Salomone S, Drago F, Nicoletti A, Zappia M (2020) Asymmetry index of Blink Reflex Recovery Cycle differentiates Parkinson’s disease from atypical Parkinsonian syndromes. J Neurol 267(6):1859–1863. https://doi.org/10.1007/s00415-020-09900-6
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12(3):189–198. https://doi.org/10.1016/0022-3956(75)90026-6
Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, Poewe W, Sampaio C, Stern MB, Dodel R, Dubois B, Holloway R, Jankovic J, Kulisevsky J, Lang AE, Lees A, Leurgans S, LeWitt PA, Nyenhuis D, Olanow CW, Rascol O, Schrag A, Teresi JA, van Hilten JJ, LaPelle N, Movement Disorder Society URTF (2008) Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 23(15):2129–2170. https://doi.org/10.1002/mds.22340
Kubo Y, Kazui H, Yoshida T, Kito Y, Kimura N, Tokunaga H, Ogino A, Miyake H, Ishikawa M, Takeda M (2008) Validation of grading scale for evaluating symptoms of idiopathic normal-pressure hydrocephalus. Dement Geriatr Cogn Disord 25(1):37–45. https://doi.org/10.1159/000111149
van Hedel HJ, Wirz M, Dietz V (2005) Assessing walking ability in subjects with spinal cord injury: validity and reliability of 3 walking tests. Arch Phys Med Rehabil 86(2):190–196. https://doi.org/10.1016/j.apmr.2004.02.010
van Swieten JC, Koudstaal PJ, Visser MC, Schouten HJ, van Gijn J (1988) Interobserver agreement for the assessment of handicap in stroke patients. Stroke 19(5):604–607. https://doi.org/10.1161/01.str.19.5.604
Quattrone A, Sarica A, La Torre D, Morelli M, Vescio B, Nigro S, Barbagallo G, Nistico R, Salsone M, Arcuri PP, Novellino F, Bianco MG, Arabia G, Cascini G, Quattrone A (2020) Magnetic resonance imaging biomarkers distinguish normal pressure hydrocephalus from progressive supranuclear palsy. Mov Disord 35(8):1406–1415. https://doi.org/10.1002/mds.28087
Miskin N, Patel H, Franceschi AM, Ades-Aron B, Le A, Damadian BE, Stanton C, Serulle Y, Golomb J, Gonen O, Rusinek H, George AE, Alzheimer’s Disease Neuroimaging I (2017) Diagnosis of normal-pressure hydrocephalus: use of traditional measures in the era of volumetric MR imaging. Radiology 285(1):197–205. https://doi.org/10.1148/radiol.2017161216
Ishii K, Kanda T, Harada A, Miyamoto N, Kawaguchi T, Shimada K, Ohkawa S, Uemura T, Yoshikawa T, Mori E (2008) Clinical impact of the callosal angle in the diagnosis of idiopathic normal pressure hydrocephalus. Eur Radiol 18(11):2678–2683. https://doi.org/10.1007/s00330-008-1044-4
Quattrone A, Nicoletti G, Messina D, Fera F, Condino F, Pugliese P, Lanza P, Barone P, Morgante L, Zappia M, Aguglia U, Gallo O (2008) MR imaging index for differentiation of progressive supranuclear palsy from Parkinson disease and the Parkinson variant of multiple system atrophy. Radiology 246(1):214–221. https://doi.org/10.1148/radiol.2453061703
Nigro S, Antonini A, Vaillancourt DE, Seppi K, Ceravolo R, Strafella AP, Augimeri A, Quattrone A, Morelli M, Weis L, Fiorenzato E, Biundo R, Burciu RG, Krismer F, McFarland NR, Mueller C, Gizewski ER, Cosottini M, Del Prete E, Mazzucchi S, Quattrone A (2020) Automated MRI classification in progressive supranuclear palsy: a large international cohort study. Mov Disord 35(6):976–983. https://doi.org/10.1002/mds.28007
Brogley JE (2019) DaTQUANT: the future of diagnosing Parkinson disease. J Nucl Med Technol 47(1):21–26. https://doi.org/10.2967/jnmt.118.222349
Peterson DA, Sejnowski TJ (2017) A dynamic circuit hypothesis for the pathogenesis of Blepharospasm. Front Comput Neurosci 11:11. https://doi.org/10.3389/fncom.2017.00011
Kimura J, Wilkinson JT, Damasio H, Adams HR Jr, Shivapour E, Yamada T (1985) Blink reflex in patients with hemispheric cerebrovascular accident (CVA). Blink reflex in CVA. J Neurol Sci 67(1):15–28. https://doi.org/10.1016/0022-510x(85)90018-8
Spissu A, Cossu G, Cannas A (2004) Late blink reflex changes in patients with pure sensory stroke due to geniculo-thalamic infarct: a contribution to the long loop theory. J Clin Neurophysiol 21(2):105–109. https://doi.org/10.1097/00004691-200403000-00005
Chia LG (1997) Late blink reflex changes in lesions of thalamus and internal capsule. Neurology 49(3):874–876. https://doi.org/10.1212/wnl.49.3.874
Esteban A (1999) A neurophysiological approach to brainstem reflexes Blink reflex. Neurophysiol Clin 29(1):7–38. https://doi.org/10.1016/S0987-7053(99)80039-2
Mocco J, Tomey MI, Komotar RJ, Mack WJ, Frucht SJ, Goodman RR, McKhann GM 2nd (2006) Ventriculoperitoneal shunting of idiopathic normal pressure hydrocephalus increases midbrain size: a potential mechanism for gait improvement. Neurosurgery 59(4):847–850. https://doi.org/10.1227/01.NEU.0000232655.78335.D5 (discussion 850–841)
Constantinides VC, Paraskevas GP, Velonakis G, Toulas P, Stefanis L, Kapaki E (2020) Midbrain morphology in idiopathic normal pressure hydrocephalus: a progressive supranuclear palsy mimic. Acta Neurol Scand 141(4):328–334. https://doi.org/10.1111/ane.13205
Lee PH, Yong SW, Ahn YH, Huh K (2005) Correlation of midbrain diameter and gait disturbance in patients with idiopathic normal pressure hydrocephalus. J Neurol 252(8):958–963. https://doi.org/10.1007/s00415-005-0791-2
Sarica A, Quattrone A, Quarantelli M, Arcuri PP, Mechelli A, La Torre D, Vaccaro MG, Cascini GL, Quattrone A (2021) Reduced striatal DAT uptake normalizes after shunt in normal-pressure hydrocephalus. Mov Disord 36(1):261–262. https://doi.org/10.1002/mds.28336
Todisco M, Zangaglia R, Minafra B, Pisano P, Trifiro G, Bossert I, Pozzi NG, Brumberg J, Ceravolo R, Isaias IU, Fasano A, Pacchetti C (2021) Clinical outcome and striatal dopaminergic function after shunt surgery in patients with idiopathic normal pressure hydrocephalus. Neurology. https://doi.org/10.1212/WNL.0000000000012064
Practice Guideline (2016) Idiopathic normal pressure hydrocephalus: response to shunting and predictors of response: report of the guideline development, dissemination, and implementation subcommittee of the American Academy of Neurology. Neurology 86(8):793. https://doi.org/10.1212/WNL.0000000000002432
Williams MA, Malm J (2016) Diagnosis and Treatment of Idiopathic Normal Pressure Hydrocephalus. Continuum (Minneap Minn) 22(2 Dementia):579–599. https://doi.org/10.1212/CON.0000000000000305
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: AM, AQ, and AQ; methodology: AM, AQ, RN, MC, DLT, and BV; formal analysis and investigation: AM, AQ, RN, MC, DLT, and BV; writing—original draft preparation: AM, and AQ; writing—review and editing: AQ; supervision: AQ.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no financial or other conflict of interest.
Ethics approval
All the study procedures and ethical aspects were approved by the institutional review board of Magna Graecia University, Catanzaro, Italy.
Consent to participate
Written informed consent was obtained from each participant included in the study.
Consent for publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Figure 1.
a Correlation graph between midbrain area (mm2) and mean R2BRrc at ISIs 100–200 ms in the 14 iNPH patients with abnormal blink reflex recovery cycle. b Correlation graph between Magnetic Resonance Hydrocephalic Index (MRHI) and mean R2BRrc at ISIs 100–200 ms in the 14 iNPH patients with abnormal blink reflex recovery cycle. r = correlation coefficient, p = statistical significance (TIFF 9873 KB)
Rights and permissions
About this article
Cite this article
Mechelli, A., Quattrone, A., Nisticò, R. et al. Blink reflex recovery cycle distinguishes patients with idiopathic normal pressure hydrocephalus from elderly subjects. J Neurol 269, 1007–1012 (2022). https://doi.org/10.1007/s00415-021-10687-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10687-3