Abstract
Purpose
To evaluate whether oral myo-inositol supplementation (MI) is able to reduce the amount of gonadotropins (GA) and the length of controlled ovarian hyperstimulation (SL) in both Polycystic Ovarian Syndrome (PCOS) and non-PCOS women undergoing in vitro fertilization (IVF).
Methods
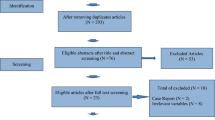
We performed a systematic review (PROSPERO ID: CRD42017069439) of randomized controlled trials (RCTs). We searched articles published in English between January 1985 to August 2017, using the combination of the Medical Subject Headings “Inositol” with “Ovulation Induction”, “follicle-stimulating hormone, human, with HCG C-terminal peptide”, “Reproductive Techniques, Assisted”, and “Fertilization in Vitro”. We collected data about GA and SL comparing MI to no treatment or d-Chiro-Inositol (DCI) supplementation (controls). A subgroup analysis was performed to evaluate selected outcomes in PCOS and non-PCOS women.
Results
We included 8 studies embedding 812 participants. We found a reduction in GA (p < 0.00001) and SL (p = 0.0007) in patients receiving MI with respect to controls. MI was effective in both PCOS (p < 0.00001) and non-PCOS women (p = 0.02) in reducing GA; conversely, MI supplementation decreased the SL only in PCOS women (p < 0.00001).
Conclusion
During IVF, MI is effective in both PCOS and non-PCOS women in saving gonadotropins, but reduces efficiently SL only in PCOS women.
Similar content being viewed by others
References
Bizzarri M, Fuso A, Dinicola S, Cucina A, Bevilacqua A (2016) Pharmacodynamics and pharmacokinetics of inositol(s) in health and disease. Expert Opin Drug Metab Toxicol 12:1181–1196. https://doi.org/10.1080/17425255.2016.1206887
Hatch AJ, York JD (2010) SnapShot: inositol phosphates. Cell 143(1030–1030):e1. https://doi.org/10.1016/j.cell.2010.11.045
Di Paolo G, De Camilli P (2006) Phosphoinositides in cell regulation and membrane dynamics. Nature 443:651–657. https://doi.org/10.1038/nature05185
Sortino MA, Salomone S, Carruba MO, Drago F (2017) Polycystic ovary syndrome: insights into the therapeutic approach with inositols. Front Pharmacol 8:341. https://doi.org/10.3389/fphar.2017.00341
Chakraborty A, Kim S, Snyder SH (2011) Inositol pyrophosphates as mammalian cell signals. Sci Signal 4:re1. https://doi.org/10.1126/scisignal.2001958
Noventa M, Vitagliano A, Quaranta M, Borgato S, Abdulrahim B, Gizzo S (2016) Preventive and therapeutic role of dietary inositol supplementation in periconceptional period and during pregnancy: a summary of evidences and future applications. Reprod Sci 23:278–288. https://doi.org/10.1177/1933719115594018
Paul C, Laganà AS, Maniglio P, Triolo O, Brady DM (2016) Inositol’s and other nutraceuticals’ synergistic actions counteract insulin resistance in polycystic ovarian syndrome and metabolic syndrome: state-of-the-art and future perspectives. Gynecol Endocrinol 32:431–438. https://doi.org/10.3109/09513590.2016
Vitagliano A, Noventa M, Gizzo S (2015) Is it time to consider patients suffering from endometriosis-related infertility as “novel candidates” for targeted peri-conceptional d-chiro inositol supplementation? Hypothesis, rationale and some considerations. J Assist Reprod Genet 32:407–408. https://doi.org/10.1007/s10815-014-0412-z
Pizzo A, Laganà AS, Barbaro L (2014) Comparison between effects of myo-inositol and d-chiro-inositol on ovarian function and metabolic factors in women with PCOS. Gynecol Endocrinol 30:205–208. https://doi.org/10.3109/09513590.2013.860120
Papaleo E, Unfer V, Baillargeon JP, Fusi F, Occhi F, De Santis L (2009) Myo-inositol may improve oocyte quality in intracytoplasmic sperm injection cycles. A prospective, controlled, randomized trial. Fertil Steril 91:1750–1754. https://doi.org/10.1016/j.fertnstert.2008.01.088
Colazingari S, Treglia M, Najjar R, Bevilacqua A (2013) The combined therapy myo-inositol plus d-chiro-inositol, rather than d-chiro-inositol, is able to improve IVF outcomes: results from a randomized controlled trial. Arch Gynecol Obstet 288:1405–1411. https://doi.org/10.1007/s00404-013-2855-3
Pacchiarotti A, Carlomagno G, Antonini G, Pacchiarotti A (2016) Effect of myo-inositol and melatonin versus myo-inositol, in a randomized controlled trial, for improving in vitro fertilization of patients with polycystic ovarian syndrome. Gynecol Endocrinol 32:69–73. https://doi.org/10.3109/09513590.2015.1101444
Mendoza N, Pérez L, Simoncini T, Genazzani A (2017) Inositol supplementation in women with polycystic ovary syndrome undergoing intracytoplasmic sperm injection: a systematic review and meta-analysis of randomized controlled trials. Reprod Biomed Online 35:529–535. https://doi.org/10.1016/j.rbmo.2017.07.005
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Higgins JPT, Green S (eds) (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration. Available from http://handbook.cochrane.org
Rizzo P, Raffone E, Benedetto V (2010) Effect of the treatment with myo-inositol plus folic acid plus melatonin in comparison with a treatment with myo-inositol plus folic acid on oocyte quality and pregnancy outcome in IVF cycles. A prospective, clinical trial. Eur Rev Med Pharmacol 14:555–561
Ciotta L, Stracquadanio M, Pagano I, Carbonaro A, Palumbo M, Gulino F (2011) Effects of Myo-Inositol supplementation on oocyte’s quality in PCOS patients: a double blind trial. Eur Rev Med Pharmacol Sci 15:509–514
Unfer V, Carlomagno G, Rizzo P, Raffone E, Roseff S (2011) Myo-inositol rather than d-chiro-inositol is able to improve oocyte quality in intracytoplasmic sperm injection cycles. A prospective, controlled, randomized trial. Eur Rev Med Pharmacol Sci 15:452–457
Carlomagno G, Oliva MM, Roseff SJ, Unfer V (2012) Myo-inositol: ovarian stimulation and IVF outcomes. Fertil Steril 98:S74–S75. https://doi.org/10.1016/j.fertnstert.2012.07.270
Lisi F, Carfagna P, Oliva MM, Rago R, Lisi R, Poverini R, Manna C, Vaquero E, Caserta D, Raparelli V, Marci R, Moscarini M (2012) Pretreatment with myo-inositol in non polycystic ovary syndrome patients undergoing multiple follicular stimulation for IVF: a pilot study. Reprod Biol Endocrinol 10:52. https://doi.org/10.1186/1477-7827-10-52
Schillaci R, Mangione D, Lo Monte G, Vassiliadis A (2012) Inositol supplementation and IVF outcome: preliminary data. Ital J Gynaecol Obstet 21:38–44
Artini PG, Di Berardino OM, Papini F, Genazzani AD, Simi G, Ruggiero M, Cela V (2013) Endocrine and clinical effects of myo-inositol administration in polycystic ovary syndrome. A randomized study. Gynecol Endocrinol 29:375–379. https://doi.org/10.3109/09513590.2012.743020
Caprio F, D’Eufemia MD, Trotta C, Campitiello MR, Ianniello R, Mele D, Colacurci N (2015) Myo-inositol therapy for poor-responders during IVF: a prospective controlled observational trial. J Ovarian Res 8:37. https://doi.org/10.1186/s13048-015-0167-x
Lesoine B, Regidor PA (2016) Prospective randomized study on the influence of myoinositol in PCOS women undergoing IVF in the improvement of oocyte quality, fertilization rate, and embryo quality. Int J Endocrinol 2016:4378507. https://doi.org/10.1155/2016/4378507
The Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group (2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 81:19–25
Unfer V, Carlomagno G, Dante G, Facchinetti F (2012) Effects of myo-inositol in women with PCOS: a systematic review of randomized controlled trials. Gynecol Endocrinol 28:509–515. https://doi.org/10.3109/09513590.2011.650660
Unfer V, Nestler JE, Kamenov ZA, Prapas N, Facchinetti F (2016) Effects of Inositol(s) in women with PCOS: a systematic review of randomized controlled trials. Int J Endocrinol 2016:1849162. https://doi.org/10.1155/2016/1849162
Zheng X, Lin D, Zhang Y, Lin Y, Song J, Li S, Sun Y (2017) Inositol supplement improves clinical pregnancy rate in infertile women undergoing ovulation induction for ICSI or IVF-ET. Medicine (Baltimore) 96:e8842. https://doi.org/10.1097/MD.0000000000008842
Busnelli A, Papaleo E, Del Prato D, La Vecchia I, Iachini E, Paffoni A, Candiani M, Somigliana E (2015) A retrospective evaluation of prognosis and cost-effectiveness of IVF in poor responders according to the Bologna criteria. Hum Reprod 30:315–322. https://doi.org/10.1093/humrep/deu319
Zivi E, Simon A, Laufer N (2010) Ovarian hyperstimulation syndrome: definition, incidence, and classification. Semin Reprod Med 28:441–447. https://doi.org/10.1055/s-0030-1265669
Csokmay JM, Yauger BJ, Henne MB, Armstrong AY, Queenan JT, Segars JH (2010) Cost analysis model of outpatient management of ovarian hyperstimulation syndrome with paracentesis: “Tap early and often” versus hospitalization. Fertil Steril 93:167–173. https://doi.org/10.1016/j.fertnstert.2008.09.054
Funding
The work was not supported by any grant/fund.
Author information
Authors and Affiliations
Contributions
ASL: designed the systematic review and lead its development. AV and MN: performed the meta-analysis of retrieved data. GA: edited the manuscript. RD: supervised the development of the systematic review and meta-analysis and gave the final approval. All the authors concurred to screen the literature, include relevant data and write the manuscript. All the authors fulfil the International Committee of Medical Journal Editors (ICMJE) criteria.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no proprietary, financial, professional, or other personal interest of any nature in any product, service, or company. The authors alone are responsible for the content and writing of the paper.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Laganà, A.S., Vitagliano, A., Noventa, M. et al. Myo-inositol supplementation reduces the amount of gonadotropins and length of ovarian stimulation in women undergoing IVF: a systematic review and meta-analysis of randomized controlled trials. Arch Gynecol Obstet 298, 675–684 (2018). https://doi.org/10.1007/s00404-018-4861-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-018-4861-y