Abstract
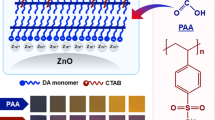
Polydiacetylenes (PDAs) and PDA/ZnO nanocomposites based on the monomers 10,12-pentacosadiynoic acid (PCDA), 10,12-tricosadiynoic acid (TCDA), and 10,12-docosadiynedioic acid (DCDA) monomers have been investigated for chromatic chemical sensing of a number of organic liquids. Chromatic sensitivity is associated with the interaction of the organic liquid with the PDA side chain to give rise to the strain-induced blue to red colorimetric transition. Attenuated total reflection (ATR) Fourier transform infrared (FTIR) spectroscopy demonstrated that in the PDA/ZnO nanocomposites, the PDA side chains form chelates with ZnO. The chromatic properties of PDAs and PDA/ZnO composites in organic liquids, to certain extent, depend on the side-chain length and the number of carboxylic head groups. Pure PDAs and PDA/ZnO nanocomposites in different organic liquids studied by Raman spectroscopy show that the chromatic selectivity of PDAs for certain organic liquids with respect to the blue to red phase transition is closely related to the side-chain structure of the PDAs. Moreover, the interactions are stronger with those PDAs where the blue to red transition is irreversible. Density functional theory (DFT) simulations show that the chromatic sensitivity of the PDAs toward a particular organic correlates with the C–C bond torsion angle of the PDA backbone.









Similar content being viewed by others
References
Descalzo AB, Dolores Marcos M, Monte C, Martinez-Manez R, Rurack K (2007) Mesoporous silica materials with covalently anchored phenoxazinone dyes as fluorescent hybrid materials for vapour sensing. J Mater Chem 17:4716–4723
Janzen MC, Ponder JB, Bailey DP, Ingison CK, Suslick KS (2006) Colorimetric sensor arrays for volatile organic compounds. Anal Chem 78:3591–3600
Lu Y, Yang Y, Sellinger A, Lu M, Huang J, Fan H et al (2001) Self-assembly of mesoscopically ordered chromatic polydiacetylene/silica nanocomposites. Nature 410:913–917
Muro ML, Daws CA, Castellano FN (2008) Microarray pattern recognition based on PtII terpyridyl chloride complexes: vapochromic and vapoluminescent response. Chem Commun (46):6134-6. doi: 10.1039/b812634h
Rakow NA, Suslick KS (2000) A colorimetric sensor array for odour visualization. Nature 406:710–713
Champaiboon T, Tumcharern G, Potisatityuenyong A, Wacharasindhu S, Sukwattanasinitt M (2009) A polydiacetylene multilayer film for naked eye detection of aromatic compounds. Sensors Actuators B Chem 139:532–537
Jiang H, Wang Y, Ye Q, Zou G, Su W, Zhang Q (2010) Polydiacetylene-based colorimetric sensor microarray for volatile organic compounds. Sensors Actuators B Chem 143:789–794
Pumtang S, Siripornnoppakhun W, Sukwattanasinitt M, Ajavakom A (2011) Solvent colorimetric paper-based polydiacetylene sensors from diacetylene lipids. J Colloid Interface Sci 364:366–372
Hammond PT, Rubner MF (1997) Thermochromism in liquid crystalline polydiacetylenes. Macromolecules 30:5773–5782
Huang X, Jiang S, Liu M (2004) Metal ion modulated organization and function of the Langmuir-Blodgett films of amphiphilic diacetylene: photopolymerization, thermochromism, and supramolecular chirality. J Phys Chem B 109:114–119
Peng H, Tang J, Pang J, Chen D, Yang L, Ashbaugh HS et al (2005) Polydiacetylene/silica nanocomposites with tunable mesostructure and thermochromatism from diacetylenic assembling molecules. J Am Chem Soc 127:12782–12783
Kim JM, Lee YB, Chae SK, Ahn DJ (2006) Patterned color and fluorescent images with polydiacetylene supramolecules embedded in poly(vinyl alcohol) films. Adv Funct Mater 16:2103–2109
Lee S, Kim JM (2007) Alpha-cyclodextrin: a molecule for testing colorimetric reversibility of polydiacetylene supramolecules. Macromolecules 40:9201–9204
Park H, Lee JS, Choi H, Ahn DJ, Kim JM (2007) Rational design of supramolecular conjugated polymers displaying unusual colorimetric stability upon thermal stress. Adv Funct Mater 17:3447–3455
Batchelder DN, Evans SD, Freeman TL, Haeussling L, Ringsdorf H, Wolf H (1994) Self-assembled monolayers containing polydiacetylenes. J Am Chem Soc 116:1050–1053
Baughman RH (1972) Solid-state polymerization of diacetylenes. J Appl Phys 43:4362–4370
Robert WC, Darryl YS, Matthew SM, Eriksson MA, Alan RB (2004) Polydiacetylene films: a review of recent investigations into chromogenic transitions and nanomechanical properties. J Phys Condens Matter 16:R679–R697
Chanakul A, Traiphol N, Traiphol R (2013) Controlling the reversible thermochromism of polydiacetylene/zinc oxide nanocomposites by varying alkyl chain length. J Colloid Interface Sci 389:106–114
Charoenthai N, Pattanatornchai T, Wacharasindhu S, Sukwattanasinitt M, Traiphol R (2011) Roles of head group architecture and side chain length on colorimetric response of polydiacetylene vesicles to temperature, ethanol and pH. J Colloid Interface Sci 360:565–573
Gou M, Guo G, Zhang J, Men K, Song J, Luo F et al (2010) Time–temperature chromatic sensor based on polydiacetylene (PDA) vesicle and amphiphilic copolymer. Sensors Actuators B Chem 150:406–411
Ryu S, Yoo I, Song S, Yoon B, Kim J-M (2009) A thermoresponsive fluorogenic conjugated polymer for a temperature sensor in microfluidic devices. J Am Chem Soc 131:3800–3801
Traiphol N, Rungruangviriya N, Potai R, Traiphol R (2011) Stable polydiacetylene/ZnO nanocomposites with two-steps reversible and irreversible thermochromism: the influence of strong surface anchoring. J Colloid Interface Sci 356:481–489
Peng H, Sun X, Cai F, Chen X, Zhu Y, Liao G et al (2009) Electrochromatic carbon nanotube/polydiacetylene nanocomposite fibres. Nat Nanotechnol 4:738–741
Yoon B, Ham D-Y, Yarimaga O, An H, Lee CW, Kim J-M (2011) Inkjet printing of conjugated polymer precursors on paper substrates for colorimetric sensing and flexible electrothermochromic display. Adv Mater 23:5492–5497
Gatebe E, Herron H, Zakeri R, Ramiah Rajasekaran P, Aouadi S, Kohli P (2008) Synthesis and characterization of polydiacetylene films and nanotubes. Langmuir 24:11947–11954
Patlolla A, Zunino J, Frenkel AI, Iqbal Z (2012) Thermochromism in polydiacetylene-metal oxide nanocomposites. J Mater Chem 22:7028–7035
Wu A, Beck C, Ying Y, Federici J, Iqbal Z (2013) Thermochromism in polydiacetylene–ZnO nanocomposites. J Phys Chem C 117:19593–19600
Acknowledgments
The authors acknowledge support by ARDEC, Picatinny Arsenal, and CarboMet LLC.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
(DOCX 156 kb)
Rights and permissions
About this article
Cite this article
Wu, A., Gu, Y., Tian, H. et al. Effect of alkyl chain length on chemical sensing of polydiacetylene and polydiacetylene/ZnO nanocomposites. Colloid Polym Sci 292, 3137–3146 (2014). https://doi.org/10.1007/s00396-014-3365-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-014-3365-y