Abstract
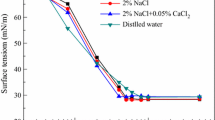
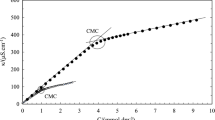
The interfacial effects of two bile salts (sodium deoxycholate (NaDC) and sodium dehydrocholate (NaDHC)) in a catanionic mixed adsorbed monolayer have been investigated at 25 °C. The surfactant interfacial composition, the interfacial orientation of the molecules and the energy changes are analysed to show a thermodynamic evidence of the hydrophobic BSs effect during its intercalation into interfacial adsorbed didodecyldimethyl ammonium bromide (DDAB) molecules. Both mixed systems (NaDC–DDAB and NaDHC–DDAB) have analogous adsorption efficiencies, which are similar from a pure DDAB monolayer and superior to that obtained for both bile salts molecules. Nevertheless, their adsorption effectiveness is different: NaDC causes an increment of Γ while NaDHC produces the opposite effect. The adsorption efficiency in surface tension reduction is due to the existence of interfacial synergistic interactions (confirmed by the analysis of β γ and ΔG ad 0 values). Maximum synergistic interaction is seen for α BSs = 0.4. The hydrophobic steroid backbone of NaDHC molecule presents a deep interfacial penetration than NaDC. This fact causes a great disturbance of DDAB hydrocarbon tails and conduces to a large separation of molecules (high A m values) which explains the reduction of adsorption effectiveness (low Γ m values).










Similar content being viewed by others
References
Radulovic J, Sefiane K, Shanahan MER (2009) Investigation of spreading of surfactant mixtures. Chem Eng Sci 64(14):3227–3235, and references therein
Zhang L, Luo L, Zhao S, Yu J (2002) Studies of synergism/antagonism for lowering dynamic interfacial tensions in surfactant/alkali/acidic oil systems, part 2: synergism/antagonism in binary surfactant mixtures. J Colloid Interface Sci 251(1):166–171
Tondre C, Caillet C (2001) Properties of the amphiphilic films in mixed cationic/anionic vesicles: a comprehensive view from a literature analysis. Adv Colloid Interface Sci 93:115–134
Knauf K, Meister A, Kerth A, Blume A (2010) Interaction of alkyltrimethylammonium bromides with DMPC-d54 and DMPG-d54 monolayers studied by infrared reflection absorption spectroscopy (IRRAS). J Colloid Interface Sci 342:243–252, and references therein
Dias R, Antunes F, Miguel M, Lindman S, Lindman B (2002) DNA-lipid systems. A physical chemistry study. Braz J Med Biol Res 35:509–522
Heerklotz H, Szadkowska H, Anderson T, Seelig J (2003) The sensitivity of lipid domains to small perturbations demonstrated by the effect of Triton. J Mol Biol 329:793–799
Blume A, Garidel P (1999) From macromolecules to man. In: Kemp RB (ed) The handbook of thermal analysis and calorimetry, vol. 4, 1st edn. Elsevier, Amsterdam, p 109, chapter 4
Janoff AS (1999) Liposomes. In: Janoff AS (ed) Rational design. Marcel Dekker, New York
Madenci D, Egelhaaf SU (2010) Self-assembly in aqueous bile salt solutions. Curr Opin Colloid Interface Sci 15(1–2):109–115
Trauner M, Boyer JL (2003) Bile salts transporters: molecular characterization, function and regulation. Physiol Rev 83:633–671
Mukhopadhyay S, Maitra U (2007) Chemistry and biology of bile acids. Curr Sci 87(12):1666–1682
Baskin R, Frost LD (2008) Bile salt–phospholipid aggregation at submicellar concentrations. Colloids Surf, B 62(2):238–242
Tung S-H, Huang Y-E, Raghavan SR (2007) A new wormlike micellar system: mixture of bile salt and lecithin in organic liquids. J Am Chem Soc 128:5751–5756
Nonomura Y, Nakayama K, Aoki Y, Fujimori A (2009) Phase behavior of bile acid/lipid/water systems containing model dietary lipids. J Colloid Interface Sci 339(1):222–229
Youssry M, Coppola L, Marques EF, Nicotera I (2008) Unravelling micellar structures and dynamic in an unusually extensive DDAB/bile salt catanionic solution by rheology and NMR-diffusometry. J Colloid Interface Sci 324:192–198
Guimarães RS, Moutinho C, Pereira E, de Castro B, Gameiro P, Lima JLFC (2007) β-Blockers and benzodiazepines location in SDS and bile salt micellar systems: an ESR study. J Pharm Biomed Anal 45(1):62–69
Marques EF, Regev O, Edlund H, Khan A (2000) Micelles, dispersions, and liquid crystals in the catanionic mixture bile salt-double-chained surfactant. The bile salt-rich area. Langmuir 16:8255–8262
Swanson-Vethamuthu M, Almgren M, Mukhtar E, Bahadur P (1992) Fluorescence quenching studies of the aggregation behavior of the mixed micelles of bile salts and cetyltrimethylammonium halides. Langmuir 8:2396–2404
Swanson-Vethamuthu M, Almgren M, Brown W, Mukhtar E (1995) Aggregate Structure, gelling, and coacervation within the l1 phase of the quasi-ternary system alkyltrimethylammonium bromide-sodium desoxycholate-water. J Colloid Interface Sci 174(2):461–479
Swanson-Vethamuthu M, Almgren M, Bergenståhl B, Mukhtar E (1996) The hexagonal phase and cylindrical micelles in the system alkyltrimethylammonium bromide–sodium desoxycholate–water as studied by X-ray diffraction and fluorescence. Colloid Interface Sci 178(2):538–548
Swanson-Vethamuthu M, Almgren M, Karlsson G, Bahadur P (1996) Effect of sodium chloride and varied alkyl chain length on aqueous cationic surfactant–bile salt systems. Cryo-TEM and fluorescence quenching studies. Langmuir 12(9):2173–2185
Swanson-Vethamuthu M, Almgren M, Hansson P, Zhao J (1996) Surface tension studies of cetyltrimethylammonium bromide-bile salt association. Langmuir 12:2186–2189
Heuman DM, Mills AS, McCall J, Hylemon PB, Pandak WM, Vlahcevic ZR (1991) Conjugates of ursodeoxycholate protect against cholestasis and hepatocellular necrosis caused by more hydrophobic bile salts. In vivo studies in the rat. Gastroenterology 100(1):203–211
Messina P, Fernández-Leyes M, Prieto G, Ruso JM, Sarmiento F, Schulz PC (2008) Spread mixed monolayers of deoxycholic and dehydrocholic acids at the air–water interface, effect of subphase pH. Characterization by axisymmetricdrop shape analysis. Biophys Chem 132:39–46
Fernández-Leyes M, Messina P, Schulz PC (2008) pH and surface tension dependence of mixed sodium deoxycholate–sodium dehydrocholate pre-micellar aggregation in aqueous solution. Colloids Surf, A Physicochem Eng Asp 329:24–30
Fernández-Leyes M, Messina P, Schulz PC (2007) Aqueous sodium dehydrocholate–sodium deoxycholate mixtures at low concentration, J. Colloid Interface Sci 314:659–664
Taylor JR (1982) An introduction to error analysis. The study of uncertainties in physical measurements. University Science Books, Mill Valley, CA
Rosen JM (2004) Surfactants and interfacial phenomena. Wiley, New York
Adamson AW (1990) Physical chemistry of surfaces, 5th edn. Wiley, New York, pp 53–101
Clint JH (1992) Surfactant aggregation. Chapman and Hall, New York, Chapter 2
Hua XY, Rosen MJ (1982) Calculation of the coefficient in the gibbs equation for the adsorption of ionic surfactants from aqueous binary mixtures with nonionic surfactants. J Colloid Interface Sci 87:469–477
Rubingh DN (1979) In: Mital K (ed) Solution chemistry of surfactants. Plenum, New York, p 337
Rosen JM, Aronson S (1981) Standard free energies of adsorption of surfactants at the aqueous solution/air interface from surface tension data in the vicinity of the critical micelle concentration. Colloid Surf 3:201–208
Szymczyk K, Jaczuk B (2007) The properties of a binary mixture of nonionic surfactants in water at the water/air interface. Langmuir 23(9):4972–4981
Messina PV, Prieto G, Ruso JM, Fernández-Leyes MD, Schulz PC, Sarmiento F (2010) Thermodynamic and elastic fluctuation analysis of langmuir mixed monolayers composed by dehydrocholic acid (HDHC) and didodecyldimethylammonium bromide (DDAB). Colloid Surf B: Biointerfaces 75(1):34–41
Messina PV, Ruso JM, Prieto G, Fernadez-Leyes M, Schulz P, Sarmiento F (2010) Ca2+ and Mg2+ induced molecular interactions in a dehydrocholic acid: didodecyldimethylammonium bromide mixed monolayer. Colloid Polym Sci 288:449–459
Hato M, Minamikawa H, Okamoto K (1993) Monolayers of ω-Hydroxyalkyldimethyloctadecylammonium bromide at water-air interface. J Colloid Interface Sci 161(1):155–162
Lu JR, Simister EA, Thomas RK, Penfold J (1993) Adsorption of alkyltrimethyl ammonium bromide at the air-water interface. Prog Colloid Polym Sci 93:92–97
Messina P, Morini MA, Schulz PC (2003) The hydration of sodium dehydrocholate micelles. Colloid Polym Sci 281(7):695–698
Garidel P, Hildebrand A, Neubert R, Blume A (2000) Thermodynamic characterization of bile salt aggregation as a function of temperature and ionic strength using isothermal titration calorimetry. Langmuir 16:5267–5275
Schulz PC, Messina P, Morini MA, Vuano B (2002) Potentiometric studies on sodium dehydrocholate micelles. Colloid Polym Sci 280(12):1104–1109
Gaines GL (1966) Insoluble monolayers at liquid gas interfaces. Wiley, New York
Gonzáles-Caballero F, Kerkeb ML (1994) Synergism at a liquid-solid interface. Adsorption of binary mixtures of bile salts onto a cholesterol surface. Langmuir 10:1268–1273
Motomura K, Yamanaka M, Aratono M (1984) Thermodynamic consideration of the mixed micelle of surfactants. Colloid Polym Sci 262(12):948–955
Small DM (1971) In: Nair PP, Kritchevsky D (eds) Bile acids, vol. 1. Plenum, New York, pp 249–356
Jańczuk B, Méndez-Sierra JA, González-Martín ML, Bruque JB, Wójcik W (1997) Properties of decylammonium chloride and cesium perfluorooctanoate at interfaces and standard free energy of their adsorption. J Colloid Interface Sci 192:408–414
Jańczuk B, Méndez-Sierra JA, González-Martín ML, Bruque JB, Wójcik W (1996) Decylammonium chloride and cesium perfluorooctanoate surface free energy and their critical micelle concentration. J Colloid Interface Sci 184:607–613
Acknowledgements
The authors acknowledge the financial support from the Universidad Nacional del Sur, Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) and Concejo Nacional de Investigaciones Científicas y Técnicas de la República Argentina (CONICET). PM is an adjunct researcher of (CONICET). MFL has a fellowship of the CONICET.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fernández-Leyes, M.D., Messina, P.V. & Schulz, P.C. Bile salt structural effect on the thermodynamic properties of a catanionic mixed adsorbed monolayer. Colloid Polym Sci 289, 179–191 (2011). https://doi.org/10.1007/s00396-010-2336-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-010-2336-1