Abstract
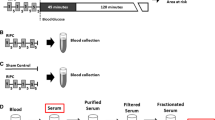
Ischaemic preconditioning (IPC) provides myocardial resistance to ischaemia/reperfusion (I/R) injuries. The protection afforded by IPC is not limited to the target tissue but extends to remote tissues, suggesting a mechanism mediated by humoral factors. The aim of the present study was to identify the humoral factors that are responsible for the cardioprotection induced by the coronary effluent transferred from IPC to naïve hearts. Isolated rat hearts were submitted to IPC (three cycles of 5 min I/R) before 30-min global ischaemia and 60-min reperfusion. The coronary effluent (Efl_IPC) collected during IPC was fractionated by ultrafiltration in different molecular weight ranges (<3, 3–5, 5–10, 10–30, 30–50, and >50 kDa) and evaluated for cardioprotective effects by perfusion before I/R in naïve hearts. Only the <3, 5–10 and <10 kDa fractions of hydrophobic eluate reduced I/R injuries. The cardioprotective effect of the 5–10 fraction was blocked by KATP channel blockers and a PKC inhibitor. An Efl_IPC proteomic analysis revealed 14 cytoprotection-related proteins in 4–12 kDa peptides. HSP10 perfusion protected the heart against I/R injuries. These data provide insights into the mechanisms of cardioprotection in humoral factors released by IPC. Cardioprotection is afforded by hydrophobic peptides in the 4–12 kDa size range, which activate pathways that are dependent on PKC and KATP. Fourteen 4–12 kDa peptides were identified, suggesting a potential therapeutic role for these molecules in ischaemic diseases. One of these, HSP10, identified by mass spectrometry, reduced I/R injuries and may be a potential candidate as a therapeutic target.








Similar content being viewed by others
References
Birnbaum Y, Hale SL, Kloner RA (1997) Ischemic preconditioning at a distance: reduction of myocardial infarct size by partial reduction of blood supply combined with rapid stimulation of the gastrocnemius muscle in the rabbit. Circulation 96:1641–1646. doi:10.1161/01.CIR.96.5.1641
Breivik L, Helgeland E, Aarnes EK, Mrdalj J, Jonassen AK (2011) Remote postconditioning by humoral factors in effluent from ischemic preconditioned rat hearts is mediated via PI3K/Akt-dependent cell-survival signaling at reperfusion. Basic Res Cardiol 106:135–145. doi:10.1007/s00395-010-0133-0
Brzozowskia T, Konturekb PC, Kontureka SJ, Pajdoa R, Kwieciena S, Pawlika M, Drozdowicza D, Sliwowskia Z, Pawlik WW (2004) Ischemic preconditioning of remote organs attenuates gastric ischemia–reperfusion injury through involvement of prostaglandins and sensory nerves. Eur J Pharmacol 499:201–213. doi:10.1016/j.ejphar.2004.07.072
Buerke M, Murohara T, Skurk C, Nuss C, Tomaselli K, Lefer AM (1995) Cardioprotective effect of insulin-like growth factor I in myocardial ischemia followed by reperfusion. Proc Natl Acad Sci USA 92:8031–8035
Cabrera-Fuentes HA, Aragones J, Bernhagen J, Boening A, Boisvert WA, Bøtker HE, Bulluck H, Cook S, Di Lisa F, Engel FB, Engelmann B, Ferrazzi F, Ferdinandy P, Fong A, Fleming I, Gnaiger E, Hernández-Reséndiz S, Kalkhoran SB, Kim MH, Lecour S, Liehn EA, Marber MS, Mayr M, Miura T, Ong SB, Peter K, Sedding D, Singh MK, Suleiman MS, Schnittler HJ, Schulz R, Shim W, Tello D, Vogel CW, Walker M, Li QO, Yellon DM, Hausenloy DJ, Preissner KT (2016) From basic mechanisms to clinical applications in heart protection, new players in cardiovascular diseases and cardiac theranostics: meeting report from the third international symposium on “New frontiers in cardiovascular research”. Basic Res Cardiol 111:69. doi:10.1007/s00395-016-0586-x
Cai ZP, Parajuli N, Zheng X, Becker L (2012) Remote ischemic preconditioning confers late protection against myocardial ischemia–reperfusion injury in mice by upregulating interleukin-10. Basic Res Cardiol 107:277–288. doi:10.1007/s00395-012-0277-1
Chen Y, Cao F, Wan B, Dou Y, Lei M (2012) Structure of the SPRY domain of human Ash2L and its interactions with RbBP5 and DPY30. Cell Res 22:598–602. doi:10.1038/cr.2012.9
Davani EY, Brumme Z, Singhera GK, Côté HC, Harrigan PR, Dorscheid D (2003) Insulin-like growth factor-1 protects ischemic murine myocardium from ischemia/reperfusion associated injury. Crit Care 7:176–183. doi:10.1186/cc2375
Davidson SM, Selvaraj P, He D, Boi-Doku C, Yellon RL, Vicencio JM, Yellon DM (2013) Remote ischaemic preconditioning involves signalling through the SDF-1α/CXCR4 signalling axis. Basic Res Cardiol 108:377–386. doi:10.1007/s00395-013-0377-6
del Peso L, González-Garcia M, Page C, Herrera R, Nuñez G (1997) Interleukin-3-induced phosphorylation of BAD through the protein kinase Akt. Science 278:687–689
Dickson EW, Lorbar M, Porcaro WA, Fenton RA, Heard SO, Reinhardt CP, Gysembergh A, Przyklenk K (1999) Rabbit heart can be “preconditioned” via transfer of coronary effluent. Am J Physiol 277:2451–2457
Donato R (1999) Functional roles of S100 proteins, calcium-binding proteins of the EF-hand type. Biochim Biophys Acta 1450:191–231
Dorn GW, Souroujon MC, Liron T, Chen CH, Gray MO, Zhou HZ, Csukai M, Wu G, Lorenz JN, Mochly-Rosen D (1999) Sustained in vivo cardiac protection by a rationally designed peptide that causes ε protein kinase C translocation. Proc Natl Acad Sci USA 96:12798–12803. doi:10.1073/pnas.96.22.12798
Doroudgar S, Quijada P, Konstandin M, Ilves K, Broughton K, Khalafalla FG, Casillas A, Nguyen K, Gude N, Toko H, Ornelas L, Thuerauf DJ, Glembotski CC, Sussman MA, Völkers M (2016) S100A4 protects the myocardium against ischemic stress. J Mol Cell Cardiol 100:54–63. doi:10.1016/j.yjmcc.2016.10.001
Ernst P, Vakoc CR (2012) WRAD: enabler of the SET1-family of H3K4 methyltransferases. Brief Funct Genomics 11:217–226. doi:10.1093/bfgp/els017
Fan G-C, Ren X, Qian J, Yuan Q, Nicolaou P, Wang Y, Jones K, Chu G, Kranias EG (2005) Novel cardioprotective role of a small heat-shock protein, HSP20, against ischemia/reperfusion injury. Circulation 111:1792–1799. doi:10.1161/01.CIR.0000160851.41872.C6
Ferrera R, Benhabbouche S, Bopassa JC, Li B, Ovize M (2009) One hour reperfusion is enough to assess function and infarct size with TTC staining in Langendorff rat model. Cardiovasc Drugs Ther 23:327–331. doi:10.1007/s10557-009-6176-5
Garg V, Hu K (2007) Protein kinase C isoform-dependent modulation of ATP-sensitive K+ channels in mitochondrial inner membrane. Am J Physiol Heart Circ Physiol 293:322–332. doi:10.1152/ajpheart.01035.2006
Gedik N, Maciel L, Schulte C, Skyschally A, Heusch G, Kleinbongard P (2017) Cardiomyocyte mitochondria as targets of humoral factors released by remote ischemic preconditioning. Arch Med Sci 13:448–458. doi:10.5114/aoms.2016.61789
Gho BC, Schoemaker RG, van den Doel MA, Duncker DJ, Verdouw PD (1996) Myocardial protection by brief ischemia in noncardiac tissue. Circulation 94:2193–2200. doi:10.1161/01.CIR.94.9.2193
Grose JH, Langston K, Wang X, Squires S, Mustafi SB, Hayes W, Neubert J, Fischer SK, Fasano M, Saunders GM, Dai Q, Christians E, Lewandowski ED, Ping P, Benjamin IJ (2015) Characterization of the cardiac overexpression of HSPB2 reveals mitochondrial and myogenic roles supported by a cardiac HspB2 interactome. PLoS One. doi:10.1371/journal.pone.0133994
Harkin DW, Barros D’Sa AAB, McCallion K, Hoper M, Campbell FC (2002) Ischemic preconditioning before lower limb ischemia–reperfusion protects against acute lung injury. J Vasc Surg 35:1264–1273. doi:10.1067/mva.2002.121981
Hausenloy DJ, Barrabes JA, Bøtker HE, Davidson SM, Di Lisa F, Downey J, Engstrom T, Ferdinandy P, Carbrera-Fuentes HA, Heusch G, Ibanez B, Iliodromitis EK, Inserte J, Jennings R, Kalia N, Kharbanda R, Lecour S, Marber M, Miura T, Ovize M, Perez-Pinzon MA, Piper HM, Przyklenk K, Schmidt MR, Redington A, Ruiz-Meana M, Vilahur G, Vinten-Johansen J, Yellon DM, Garcia-Dorado D (2016) Ischaemic conditioning and targeting reperfusion injury: a 30 year voyage of discovery. Basic Res Cardiol 111:70. doi:10.1007/s00395-016-0588-8
Hausenloy DJ, Candilio L, Evans R, Ariti C, Jenkins DP, Kolvekar S, Knight R, Kunst G, Laing C, Nicholas J, Pepper J, Robertson S, Xenou M, Clayton T, Yellon DM, Trial Investigators ERICCA (2015) Remote ischemic preconditioning and outcomes of cardiac surgery. N Engl J Med 373:1408–1417. doi:10.1056/NEJMoa1413534
Helgeland E, Breivik LE, Vaudel M, Svendsen ØS, Garberg H, Nordrehaug JE, Berven FS (2014) Exploring the human plasma proteome for humoral mediators of remote ischemic preconditioning—a word of caution. PLoS One 9:e109279. doi:10.1371/journal.pone.0109279
Hepponstall M, Ignjatovic V, Binos S, Monagle P, Jones B, Cheung MH, d´Udekem Y, Konstantinov IE (2012) Remote ischemic preconditioning (RIPC) modifies plasma proteome in humans. PLoS One 7:e48284. doi:10.1371/journal.pone.0048284
Héron-Milhavet L, LeRoith D (2002) Insulin-like growth factor I induces MDM2-dependent degradation of p53 via the p38 MAPK pathway in response to DNA damage. J Biol Chem 277:15600–15606. doi:10.1074/jbc.M111142200
Heusch G (2015) Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116:674–699. doi:10.1161/CIRCRESAHA.116.305348
Heusch G (2017) Critical issues for the translation of cardioprotection. Circ Res 120:1477–1486. doi:10.1161/CIRCRESAHA.117.310820
Heusch G, Bøtker HE, Przyklenk K, Redington A, Yellon D (2015) Remote ischemic conditioning. J Am Coll Cardiol 65:177–195. doi:10.1016/j.jacc.2014.10.031
Heusch G, Gersh BJ (2016) ERICCA and RIPHeart: two nails in the coffin for cardioprotection by remote ischemic conditioning? Probably not! Eur Heart J 37:200–202. doi:10.1093/eurheartj/ehv606
Heusch G, Rassaf T (2016) Time to give up on cardioprotection? A critical appraisal of clinical studies on ischemic pre-, post-, and remote conditioning. Circ Res 119:676–695. doi:10.1161/CIRCRESAHA.116.308736
Hibert P, Prunier-Mirebeau D, Beseme O, Chwastyniak M, Tamareille S, Lamon D, Furber A, Pinet F, Prunier F (2013) Apolipoprotein A-I is a potential mediator of remote ischemic preconditioning. PLoS One 8:e77211. doi:10.1371/journal.pone.0077211
Hildebrandt HA, Kreienkamp V, Gent S, Kahlert P, Heusch G, Kleinbongard P (2016) Kinetics and signal activation properties of circulating factor(s) from healthy volunteers undergoing remote ischemic pre-conditioning. JACC Basic Transl Sci 1:3–13. doi:10.1016/j.jacbts.2016.01.007
Jensen RV, Støttrup NB, Kristiansen SB, Bøtker HE (2012) Release of a humoral circulating cardioprotective factor by remote ischemic preconditioning is dependent on preserved neural pathways in diabetic patients. Basic Res Cardiol 107:285–293. doi:10.1007/s00395-012-0285-1
Jensen RV, Zachara NE, Nielsen PH, Kimose HH, Kristiansen SB, Bøtker HE (2013) Impact of O-GlcNAc on cardioprotection by remote ischaemic preconditioning in non-diabetic and diabetic patients. Cardiovasc Res 97:369–378. doi:10.1093/cvr/cvs337jiang2011
Jiang H, Shukla A, Wang X, Chen W, Bernstein BE, Roeder RG (2011) Role for Dpy-30 in ES cell-fate specification by regulation of H3K4 methylation within bivalent domains. Cell 144:513–525. doi:10.1016/j.cell.2011.01.020
Johnsen J, Pryds K, Salman R, Løfgren B, Kristiansen SB, Bøtker HE (2016) The remote ischemic preconditioning algorithm: effect of number of cycles, cycle duration and effector organ mass on efficacy of protection. Basic Res Cardiol 111:10. doi:10.1007/s00395-016-0529-6
Kalakech H, Hibert P, Prunier-Mirebeau D, Tamareille S, Letournel F, Macchi L, Pinet F, Furber A, Prunier F (2014) RISK and SAFE signaling pathway involvement in apolipoprotein A-I-induced cardioprotection. PLoS One 9:e107950. doi:10.1371/journal.pone.0107950
Kappé G, Franck E, Verschuure P, Boelens WC, Leunissen JA, De Jong WW (2003) The human genome encodes 10 alpha-crystallin-related small heat shock proteins: hspB1-10. Cell Stress Chaperones 8:53–61. doi:10.1379/1466-1268(2003)8<53:THGECS>2.0.CO;2
Kharbanda RK, Mortensen UM, White PA, Kristiansen SB, Schmidt MR, Hoschtitzky JA, Vogel M, Sorensen K, Redington AN, MacAllister R (2002) Transient limb ischemia induces remote ischemic preconditioning in vivo. Circulation 106:2881–2883. doi:10.1161/01.CIR.0000043806.51912.9B
Kleinbongard P, Neuhauser M, Thielmann M, Kottenberg E, Peters J, Jakob H, Heusch G (2016) Confounders of cardioprotection by remote ischemic preconditioning in patients undergoing coronary artery bypass grafting. Cardiology 133:128–133. doi:10.1159/000441216
Kleinbongard P, Skyschally A, Heusch G (2017) Cardioprotection by remote ischemic conditioning and its signal transduction. Pflugers Arch 469:159–181. doi:10.1007/s00424-016-1922-6
Lang SC, Elsässer A, Scheler C, Vetter S, Tiefenbacher CP, Kübler W, Katus HA, Vogt AM (2006) Myocardial preconditioning and remote renal preconditioning—identifying a protective factor using proteomic methods? Basic Res Cardiol 101:149–158. doi:10.1007/s00395-005-0565-0
Li Q, Li B, Wang X, Leri A, Jana KP, Liu Y, Kajstura J, Baserga R, Anversa P (1997) Overexpression of insulin-like growth factor-1 in mice protects from myocyte death after infarction, attenuating ventricular dilation, wall stress, and cardiac hypertrophy. J Clin Invest 100:1991–1999. doi:10.1172/JCI119730
Lin KM, Lin B, Lian IY, Mestril R, Scheffler IE, Dillmann WH (2001) Combined and individual mitochondrial HSP60 and HSP10 expression in cardiac myocytes protects mitochondrial function and prevents apoptotic cell deaths induced by simulated ischemia–reoxygenation. Circulation 103:1787–1792. doi:10.1161/01.CIR.103.13.1787
Liu GS, Cohen MV, Mochly-Rosen D, Downey JM (1999) Protein kinase C-ε is responsible for the protection of preconditioning in rabbit cardiomyocytes. J Mol Cell Cardiol 31:1937–1948. doi:10.1006/jmcc.1999.1026
Liu GS, Thornton J, Van Winkle DM, Stanley AWH, Olsson RA, Downey JM (1991) Protection against infarction afforded by preconditioning is mediated by A1 adenosine receptors in rabbit heart. Circulation 84:350–356. doi:10.1161/01.CIR.84.1.350
Mastitskaya S, Basalay M, Hosford PS, Ramage AG, Gourine A, Gourine AV (2016) Identifying the source of a humoral factor of remote (pre)conditioning cardioprotection. PLoS One 11:e0150108. doi:10.1371/journal.pone.0150108
Mastitskaya S, Marina N, Gourine A, Gilbey MP, Spyer KM, Teschemacher AG, Kasparov S, Trapp S, Ackland GL, Gourine AV (2012) Cardioprotection evoked by remote ischaemic preconditioning is critically dependent on the activity of vagal pre-ganglionic neurones. Cardiovasc Res 95:487–494. doi:10.1093/cvr/cvs212
Maulik N, Engelman RM, Rousou JA, Flack JE III, Deaton D, Das DK (1999) Ischemic preconditioning reduces apoptosis by upregulating anti-death gene Bcl-2. Circulation 100:II-369–II-375. doi:10.1161/01.CIR.100.suppl_2.II-369
Meldrum KK, Meldrum DR, Sezen SF, Crone JK, Burnett AL (2001) Heat shock prevents simulated ischemia-induced apoptosis in renal tubular cells via a PKC-dependent mechanism. Am J Physiol Regul Integr Comp Physiol 281:R359–R364
Meybohm P, Bein B, Brosteanu O, Cremer J, Gruenewald M, Stoppe C, Coburn M, Schaelte G, Böning A, Niemann B, Roesner J, Kletzin F, Strouhal U, Reyher C, Laufenberg-Feldmann R, Ferner M, Brandes IF, Bauer M, Stehr SN, Kortgen A, Wittmann M, Baumgarten G, Meyer-Treschan T, Kienbaum P, Heringlake M, Schön J, Sander M, Treskatsch S, Smul T, Wolwender E, Schilling T, Fuernau G, Hasenclever D, Zacharowski K (2015) A multicenter trial of remote ischemic preconditioning for heart surgery. New Engl J Med 373:1397–1407. doi:10.1056/NEJMoa1413579
Michelsen MM, Støttrup NB, Schmidt MR, Løfgren B, Jensen RV, Tropak M, St-Michel EJ, Redington AN, Bøtker HE (2012) Exercise-induced cardioprotection is mediated by a bloodborne, transferable factor. Basic Res Cardiol 107:260–268. doi:10.1007/s00395-012-0260-x
Mocanu MM, Yellon DM (2003) p53 down-regulation: a new molecular mechanism involved in ischaemic preconditioning. FEBS Lett 555:302–306. doi:10.1016/S0014-5793(03)01260-2
Monastyrskaya K, Tschumi F, Babiychuk EB, Stroka D, Draeger A (2008) Annexins sense changes in intracellular pH during hypoxia. Biochem J 409:65–75. doi:10.1042/BJ20071116
Muimo R (2009) Regulation of CFTR function by annexin A2-S100A10 complex in health and disease. Gen Physiol Biophys 28:F14–F19
Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 74:1124–1136. doi:10.1161/01.CIR.74.5.1124
Olenchock BA, Moslehi J, Baik AH, Davidson SM, Williams J, Gibson WJ, Chakraborty AA, Pierce KA, Miller CM, Hanse EA, Kelekar A, Sullivan LB, Wagers AJ, Clish CB, Heiden MGV, Kaelin WG Jr (2016) EGLN1 inhibition and rerouting of α-ketoglutarate suffice for remote ischemic protection. Cell 164:884–895. doi:10.1016/j.cell.2016.02.006
Orre LM, Panizza E, Kaminskyy VO, Vernet E, Gräslund T, Zhivotovsky B, Lehtiö J (2013) S100A4 interacts with p53 in the nucleus and promotes p53 degradation. Oncogene 32:5531–5540. doi:10.1038/onc.2013.213
Ottani A, Galantucci M, Ardimento E, Neria L, Canalini F, Calevro A, Zaffe D, Novellino E, Grieco P, Giuliani D, Guarini S (2013) Modulation of the JAK/ERK/STAT signaling in melanocortin-induced inhibition of local and systemic responses to myocardial ischemia/reperfusion. Pharmacol Res 72:1–8. doi:10.1016/j.phrs.2013.03.005
Oxman T, Arad M, Klein R, Avazov N, Rabinowitz B (1997) Limb ischemia preconditions the heart against reperfusion tachyarrhythmia. Am J Physiol Heart Circ Physiol 273:H1707–H1712
Pell TJ, Baxter GF, Yellon DM, Drew GM (1998) Renal ischemia preconditions myocardium: role of adenosine receptors and ATP-sensitive potassium channels. Am J Physiol Heart Circ Physiol 275:H1542–H1547
Povlsen JA, Løfgren B, Dalgas C, Jespersen NR, Johnsen J, Bøtker HE (2014) Frequent biomarker analysis in the isolated perfused heart reveals two distinct phases of reperfusion injury. Int J Cardiol 171:9–14. doi:10.1016/j.ijcard.2013.11.035
Przyklenk K, Bauer B, Ovize M, Kloner RA, Whittaker P (1993) Regional ischemic “preconditioning” protects remote virgin myocardium from subsequent sustained coronary occlusion. Circulation 87:893–899. doi:10.1161/01.CIR.87.3.893
Redington KL, Disenhouse T, Strantzas SC, Gladstone R, Wei C, Tropak MB, Dai X, Manlhiot C, Li J, Redington AN (2012) Remote cardioprotection by direct peripheral nerve stimulation and topical capsaicin is mediated by circulating humoral factors. Basic Res Cardiol 107:241–250. doi:10.1007/s00395-011-0241-5
Rossello X, Hall AR, Bell RM, Yellon DM (2016) Characterization of the Langendorff perfused isolated mouse heart model of global ischemia–reperfusion injury: impact of ischemia and reperfusion length on infarct size and LDH release. J Cardiovasc Pharmacol Ther 21:286–295. doi:10.1177/1074248415604462
Saurin AT, Pennington DJ, Raat NJ, Latchman DS, Owen MJ, Marber MS (2002) Targeted disruption of the protein kinase C epsilon gene abolishes the infarct size reduction that follows ischaemic preconditioning of isolated buffer-perfused mouse hearts. Cardiovasc Res 55:672–680. doi:10.1016/S0008-6363(02)00325-5
Schulman D, Latchman DS, Yellon DM (2002) Urocortin protects the heart from reperfusion injury via upregulation of p42/p44 MAPK signaling pathway. Am J Physiol Heart Circ Physiol 283:H1481–H1488. doi:10.1152/ajpheart.01089.2001
Schultz JE, Rose E, Yao Z, Gross GJ (1995) Evidence for involvement of opioid receptors in ischemic preconditioning in rat hearts. Am J Physiol Heart Circ Physiol 268:H2157–H2161
Serejo FC, Rodrigues-Junior LF, Tavares KCS, Campos de Carvalho AC, Nascimento JHM (2007) Cardioprotective properties of humoral factors released from rat hearts subject to ischemic preconditioning. J Cardiovasc Pharmacol 49:214–220. doi:10.1097/FJC.0b013e3180325ad9
Shan YX, Liu TJ, Su HF, Samsamshariat A, Mestril R, Wang PH (2003) Hsp10 and Hsp60 modulate bcl-2 family and mitochondria apoptosis signaling induced by doxorubicin in cardiac muscle cells. J Mol Cell Cardiol 35:1135–1143. doi:10.1016/S0022-2828(03)00229-3
Shimizu M, Tropak M, Diaz RJ, Suto F, Surendra H, Kuzmin E, Li J, Gross G, Wilson GJ, Callahan J, Redington AN (2009) Transient limb ischaemia remotely preconditions through a humoral mechanism acting directly on the myocardium: evidence suggesting cross-species protection. Clin Sci 117:191–200. doi:10.1042/CS20080523
Skrzypiec-Spring M, Grotthus B, Szeląg A, Schulz R (2007) Isolated heart perfusion according to Langendorff—still viable in the new millennium. J Pharmacol Toxicol Methods 55:113–126. doi:10.1016/j.vascn.2006.05.006
Skyschally A, Gent S, Amanakis G, Schulte C, Kleinbongard P, Heusch G (2015) Across-species transfer of protection by remote ischemic preconditioning with species-specific myocardial signal transduction by reperfusion injury salvage kinase and survival activating factor enhancement pathways. Circ Res 117:279–288. doi:10.1161/CIRCRESAHA.117.306878
Sloth AD, Schmidt MR, Munk K, Schmidt M, Pedersen L, Sørensen HT, Bøtker HE (2015) Impact of cardiovascular risk factors and medication use on the efficacy of remote ischaemic conditioning: post hoc subgroup analysis of a randomised controlled trial. BMJ Open 5:e006923. doi:10.1136/bmjopen-2014-006923
Smith CCT, Dixon RA, Wynne AM, Theodorou L, Ong S-G, Subrayan S, Davidson SM, Hausenloy DJ, Yellon DM (2010) Leptin-induced cardioprotection involves JAK/STAT signaling that may be linked to the mitochondrial permeability transition pore. Am J Physiol Heart Circ Physiol 299:H1265–H1270. doi:10.1152/ajpheart.00092.2010
Steensrud T, Li J, Dai X, Manlhiot C, Kharbanda RK, Tropak M, Redington A (2010) Pretreatment with the nitric oxide donor SNAP or nerve transection blocks humoral preconditioning by remote limb ischemia or intra-arterial adenosine. Am J Physiol Heart Circ Physiol 299:1598–1603. doi:10.1152/ajpheart.00396.2010
Sutherland FJ, Hearse DJ (2000) The isolated blood and perfusion fluid perfused heart. Pharmacol Res 41:613–627. doi:10.1006/phrs.1999.0653
Tong H, Chen W, Steenbergen C, Murphy E (2000) Ischemic preconditioning activates phosphatidylinositol-3-kinase upstream of protein kinase C. Circ Res 87:309–315. doi:10.1161/01.RES.87.4.309
Vaseva AV, Moll UM (2009) The mitochondrial p53 pathway. Biochim Biophys Acta 1787:414–420. doi:10.1016/j.bbabio.2008.10.005
Vogt AM, Htun P, Kluge A, Zimmermann R, Schaper W (1997) Insulin-like growth factor-II delays myocardial infarction in experimental coronary artery occlusion. Cardiovasc Res 33:469–477
Wall TM, Sheehy R, Hartman JC (1994) Role of bradykinin in myocardial preconditioning. J Pharmacol Exp Ther 270:681–689
Wang L, Oka N, Tropak M, Callahan J, Lee J, Wilson G, Redington A, Caldarone CA (2008) Remote ischemic preconditioning elaborates a transferable blood-borne effector that protects mitochondrial structure and function and preserves myocardial performance after neonatal cardioplegic arrest. J Thorac Cardiovasc Surg 136:335–432. doi:10.1016/j.jtcvs.2007.12.055
Ytrehus K, Liu Y, Downey JM (1994) Preconditioning protects ischemic rabbit heart by protein kinase C activation. Am J Physiol 266:1145–1152
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Sources of funding
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [Grant 483639/2013-3] and the Fundação Carlos Chagas Filho de Amparo a Pesquisa do Estado do Rio de Janeiro (FAPERJ) [Grant E-26/112.085/2012]. JHMN is a research fellow from CNPq. LM received fellowships from CNPq and FAPERJ. DFO received a fellowship from CNPq.
Conflict of interest
The authors declare no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
395_2017_641_MOESM1_ESM.tif
Supplementary material 1 (TIFF 11799 kb) Figure S1 Protein–protein interactions from the STRING interaction database. Proteins smaller than 12 kDa identified by mass-spectrometry analysis of the preconditioned coronary effluent. Rat pre-pro-insulin-like growth factor (Igf1), Heat shock protein beta-7 (Hspb7), Spermatid nuclear transition protein 1 (Tnp1), Protein dpy-30 homolog (Dpy30), DCM5 protein (Dcm5), Pneumo secretory protein 2 (Scgb3a1), CAMPATH-1 antigen (Cd52), Guanine nucleotide-binding protein, gamma subunit (Gnb3), 10 kDa heat shock protein, mitochondrial (Hspe1), Protein S100-A10, Protein S100-A4, Retinal cone rhodopsin-sensitive cGMP 3’,5’-cyclic phosphodiesterase, gamma subunit (Pde6 h), Protamine-3 (Prm3), and Apolipoprotein C-II (Apoc2). proteins are represented as nodes
395_2017_641_MOESM2_ESM.tif
Supplementary material 2 (TIFF 47941 kb) Figure S2 Effect of the perfusion of different concentrations of exogenous HSP10 on post-ischaemic cardiac performance and infarct size. a Left ventricular developed pressure after 60-min reperfusion in hearts preconditioned with 0.1, 0.3, 0.5, and 1 µM HSP10. Effects of HSP10 concentrations on the end-diastolic pressure (b) and infarct size (c), after 60-min reperfusion. Representative images of TTC-stained heart sections are shown beneath each bar in the graph c. Developed pressure was expressed as percentual of the basal value. Data are mean ± SEM. Number in each colunm is n of hearts. *P < 0.05 vs. CTL
Rights and permissions
About this article
Cite this article
Maciel, L., de Oliveira, D.F., Verissimo da Costa, G.C. et al. Cardioprotection by the transfer of coronary effluent from ischaemic preconditioned rat hearts: identification of cardioprotective humoral factors. Basic Res Cardiol 112, 52 (2017). https://doi.org/10.1007/s00395-017-0641-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-017-0641-2