Abstract
Purpose
Probiotic Lactobacillus gasseri SBT2055 (LG2055) has an anti-obesity effect although it is unknown whether the effect influences inflammatory responses in adipose tissue and lipid metabolism in the liver, which are considered substantially relevant to adiposity.
Methods
C57BL/6 mice were fed a 10 %-fat diet containing LG2055 cells for 24 weeks. We then studied body weight, fat tissue mass, liver fat content and inflammatory genes in the adipose tissue, and lipogenic and lipolytic genes in the liver.
Results
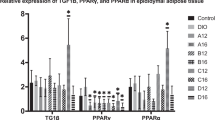
Consumption of LG2055 resulted in a significant reduction in body weight and fat tissue mass (epididymal and perirenal/retroperitoneal), with a lowered level of triglyceride content in the liver. DNA microarray analysis showed that LG2055 generally inhibited the up-regulation of pro-inflammatory genes, including CCL2 and CCR2, in the epididymal adipose tissue. In the liver, LG2055 tended to inhibit lipogenic gene up-regulation, including ACC1, FAS and SREBP1, but LG2055 did not markedly alter lipolytic genes. Real-time PCR analysis confirmed the DNA microarray results in part, showing a significant reduction in the mRNA expression of CCL2 in the epididymal adipose tissue, and a downward tendency in FAS mRNA expression in the liver, in the LG2055-fed group.
Conclusions
LG2055 significantly prevented body weight gain, fat accumulation and pro-inflammatory gene expression in the adipose tissue. Relatively lower triglyceride levels and reduced expression of lipogenic genes were also observed in the liver. It is suggested that improvement in the inflammatory state of the adipose tissue might be a possible mechanism underlying the anti-obesity effect of LG2055.


Similar content being viewed by others
References
WHO (2012) WHO | obesity and overweight. http://www.who.int/mediacentre/factsheets/fs311/en/index.html. Accessed 21 Feb 2013
Fox CS, Massaro JM, Hoffmann U et al (2007) Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham heart study. Circulation 116:39–48
Ford ES, Williamson DF, Liu S (1997) Weight change and diabetes incidence: findings from a national cohort of US adults. Am J Epidemiol 146:214–222
Liu B, Balkwill A, Reeves G, Beral V (2010) Body mass index and risk of liver cirrhosis in middle aged UK women: prospective study. BMJ 340:c912
Hubert HB, Feinleib M, McNamara PM, Castelli WP (1983) Obesity as an independent risk factor for cardiovascular disease: a 26-year follow-up of participants in the Framingham heart study. Circulation 67:968–977
Huffman D, Barzilai N (2009) Role of visceral adipose tissue in aging. Biochim Biophys Acta 1790:1117–1123
Gupta A, Gupta V (2010) Metabolic syndrome: what are the risks for humans? Biosci Trends 4:204–212
Horton ES (2009) Effects of lifestyle changes to reduce risks of diabetes and associated cardiovascular risks: results from large scale efficacy trials. Obesity (Silver Spring) 17(Suppl 3):S43–S48
Thomas CM, Versalovic J (2010) Probiotics-host communication: modulation of signaling pathways in the intestine. Gut microbes 1:148–163
Lye HS, Kuan CY, Ewe JA, Fung WY, Liong MT (2009) The improvement of hypertension by probiotics: effects on cholesterol, diabetes, renin, and phytoestrogens. Int J Mol Sci 10:3755–3775
Mallappa RH, Rokana N, Duary RK, Panwar H, Batish VK, Grover S (2012) Management of metabolic syndrome through probiotic and prebiotic interventions. Indian J Endocrinol Metab 16:20–27
Lee HY, Park JH, Seok SH, Baek MW, Kim DJ, Lee KE, Paek KS, Lee Y, Park JH (2006) Human originated bacteria, Lactobacillus rhamnosus PL60, produce conjugated linoleic acid and show anti-obesity effects in diet-induced obese mice. Biochim Biophys Acta 1761:736–744
Lee K, Paek K, Lee HY, Park JH, Lee Y (2007) Antiobesity effect of trans-10, cis-12-conjugated linoleic acid-producing Lactobacillus plantarum PL62 on diet-induced obese mice. J Appl Microbiol 103:1140–1146
Kondo S, Xiao J, Satoh T, Odamaki T, Takahashi S, Sugahara H, Yaeshima T, Iwatsuki K, Kamei A, Abe K (2010) Antiobesity effects of Bifidobacterium breve strain b-3 supplementation in a mouse model with high-fat diet-induced obesity. Biosci Biotechnol Biochem 74:1656–1661
Aronsson L, Huang Y, Parini P, Korach-André M, Håkansson J, Gustafsson J-Å, Pettersson S, Arulampalam V, Rafter J (2010) Decreased fat storage by Lactobacillus paracasei is associated with increased levels of angiopoietin-like 4 protein (ANGPTL4). PLoS One 5:e13087
Sato M, Uzu K, Yoshida T, Hamad EM, Kawakami H, Matsuyama H, Abd El-Gawad IA, Imaizumi K (2008) Effects of milk fermented by Lactobacillus gasseri SBT2055 on adipocyte size in rats. Br J Nutr 99:1013–1017
Hamad EM, Sato M, Uzu K, Yoshida T, Higashi S, Kawakami H, Kadooka Y, Matsuyama H, Abd El-Gawad IA, Imaizumi K (2009) Milk fermented by Lactobacillus gasseri SBT2055 influences adipocyte size via inhibition of dietary fat absorption in Zucker rats. Br J Nutr 101:716–724
Kadooka Y, Ogawa A, Ikuyama K, Sato M (2011) The probiotic Lactobacillus gasseri SBT2055 inhibits enlargement of visceral adipocytes and upregulation of serum soluble adhesion molecule (sICAM-1) in rats. Int Dairy J 21:623–627
Kadooka Y, Sato M, Imaizumi K, Ogawa A, Ikuyama K, Akai Y, Okano M, Kagoshima M, Tsuchida T (2010) Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur J Clin Nutr 64:636–643
Fujiwara S, Seto Y, Kimura A, Hashiba H (2001) Establishment of orally-administered Lactobacillus gasseri SBT2055SR in the gastrointestinal tract of humans and its influence on intestinal microflora and. J Appl Microbiol 90:343–352
Imai E, Fukui K, Ohta N, Tomitsuka T (2002) Effects of Lactobacillus gasseri SBT2055 on dextran sulfate sodium-induced ulcerative colitis model in rats. Biosci Microflora 21:179–183
Bastard J, Maachi M, Lagathu C, Kim MJ, Caron M, Vidal H, Capeau J, Feve B (2006) Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 17:4–12
Hotamisligil GS (2006) Inflammation and metabolic disorders. Nature 444:860–867
Ding S, Chi MM, Scull BP, Rigby R, Schwerbrock NMJ, Magness S, Jobin C, Lund PK (2010) High-fat diet: bacteria interactions promote intestinal inflammation which precedes and correlates with obesity and insulin resistance in mouse. PLoS One 5:e12191
Cani PD, Amar J, Iglesias MA et al (2007) Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56:1761–1772
Yang ZH, Miyahara H, Takeo J, Katayama M (2012) Diet high in fat and sucrose induces rapid onset of obesity-related metabolic syndrome partly through rapid response of genes involved in lipogenesis, insulin signalling and inflammation in mice. Diabetol Metab Syndr 4:32
American Institute of Nutrition (1977) Report of the American Institute of Nutrition ad hoc committee on standards for nutritional studies. J Nutr 107:1340–1348
Folch J, Lees M, Sloane-Stanley G (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Lumeng CN, Bodzin JL, Saltiel AR (2007) Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin Invest 117:175–184
Kanda H, Tateya S, Tamori Y et al (2006) MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J Clin Invest 116:1494–1505
Weisberg S, Hunter D, Huber R, Lemieux J, Slaymaker S, Vaddi K, Charo I, Leibel RL, Ferrante AWJ (2006) CCR2 modulates inflammatory and metabolic effects of high-fat feeding. J Clin Invest 116:115–124
Wellen K, Hotamisligil G (2003) Obesity-induced inflammatory changes in adipose tissue. J Clin Invest 112:1785–1788
Suganami T, Nishida J, Ogawa Y (2005) A paracrine loop between adipocytes and macrophages aggravates inflammatory changes: role of free fatty acids and tumor necrosis factor alpha. Arterioscler Thromb Vasc Biol 25:2062–2068
Shoelson SSE, Lee J, Goldfine AB (2006) Inflammation and insulin resistance. J Clin Invest 116:1793–1801
Tamura Y, Sugimoto M, Murayama T, Minami M, Nishikaze Y, Ariyasu H, Akamizu T, Kita T, Yokode M, Arai H (2010) C–C chemokine receptor 2 inhibitor improves diet-induced development of insulin resistance and hepatic steatosis in mice. J Atheroscler Thromb 17:219–228
Li H, Lelliott C, Håkansson P, Ploj K, Tuneld A, Verolin-Johansson M, Benthem L, Carlsson B, Storlien L, Michaëlsson E (2008) Intestinal, adipose, and liver inflammation in diet-induced obese mice. Metabolism 57:1704–1710
DeLany JP, Windhauser MM, Champagne CM, Bray GA (2000) Differential oxidation of individual dietary fatty acids in humans. Am J Clin Nutr 72:905–911
Tjonneland A, Overvad K, Bergmann MM et al (2009) Linoleic acid, a dietary n-6 polyunsaturated fatty acid, and the aetiology of ulcerative colitis: a nested case-control study within a European prospective cohort study. Gut 58:1606–1611
Postic C, Girard J (2008) Contribution of de novo fatty acid synthesis to hepatic steatosis and insulin resistance: lessons from genetically engineered mice. J Clin Invest 118:829–838
Mao J, DeMayo FJ, Li H, Abu-Elheiga L, Gu Z, Shaikenov TE, Kordari P, Chirala SS, Heird WC, Wakil SJ (2006) Liver-specific deletion of acetyl-CoA carboxylase 1 reduces hepatic triglyceride accumulation without affecting glucose homeostasis. Proc Natl Acad Sci USA 103:8552–8557
Ntambi JM, Miyazaki M, Stoehr JP, Lan H, Kendziorski CM, Yandell BS, Song Y, Cohen P, Friedman JM, Attie AD (2002) Loss of stearoyl-CoA desaturase-1 function protects mice against adiposity. Proc Natl Acad Sci USA 99:11482–11486
Jiang G, Li Z, Liu F et al (2005) Prevention of obesity in mice by antisense oligonucleotide inhibitors of stearoyl-CoA desaturase-1. J Clin Invest 115:1030–1038
Guerre-Millo M, Gervois P (2000) Peroxisome proliferator-activated receptor α activators improve insulin sensitivity and reduce adiposity. J Biol Chem 275:16638–16642
Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ (2005) Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest 115:1343–1351
Sanal MG (2008) The blind men “see” the elephant-the many faces of fatty liver disease. World J Gastroenterol 14:831–844
Gibbons GF (1990) Assembly and secretion of hepatic very-low-density lipoprotein. Biochem J 268:1–13
Wolfe RR, Durkot MJ (1985) Role of very low density lipoproteins in the energy metabolism of the rat. J Lipid Res 26:210–217
Raabe M, Véniant MM, Sullivan MA, Zlot CH, Björkegren J, Nielsen LB, Wong JS, Hamilton RL, Young SG (1999) Analysis of the role of microsomal triglyceride transfer protein in the liver of tissue-specific knockout mice. J Clin Invest 103:1287–1298
Hoffmann M, Rath E, Hölzlwimmer G, Quintanilla-Martinez L, Loach D, Tannock G, Haller D (2008) Lactobacillus reuteri 100–23 transiently activates intestinal epithelial cells of mice that have a complex microbiota during early stages of colonization. J Nutr 138:1684–1691
Li N, Russell WM, Douglas-escobar M, Hauser N, Lopez M, Neu J (2009) Live and heat-killed Lactobacillus rhamnosus GG: effects on proinflammatory and anti-inflammatory cytokines/chemokines in gastrostomy-fed infant rats. Pediatr Res 66:203–207
Acknowledgments
We are grateful to Ken Ukibe, Maya Yamashita, Michio Kawano, Tomohiro Hosoya, Hiroshi Uenishi and Fumihiko Sakai for their skillful collaboration and useful suggestions. We also thank Emiko Yamaguchi for preparing the experimental diets and technical assistance, and YBS Co. Ltd. for animal care. Contribution: all authors contributed to the experimental design. MM and AO mainly performed the experiments. YK supervized the study design. SH assisted with the animal experiment. MM and YK wrote the manuscript.
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miyoshi, M., Ogawa, A., Higurashi, S. et al. Anti-obesity effect of Lactobacillus gasseri SBT2055 accompanied by inhibition of pro-inflammatory gene expression in the visceral adipose tissue in diet-induced obese mice. Eur J Nutr 53, 599–606 (2014). https://doi.org/10.1007/s00394-013-0568-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-013-0568-9