Abstract
Purpose
To clarify differences patients with pathological GS (pGS) 3 + 4 according to biopsy Gleason score (bGS) after radical prostatectomy (RP) among candidates for active surveillance.
Methods
Between January 2006 and June 2014, 619 patients who met Royal Marsden criteria and had a pGS 3 + 4 after RP were identified. Patients were stratified into two groups according to bGS: Group A (n = 430) with bGS (3 + 3) and Group B (n = 189) with bGS 7 (3 + 4). Pathological outcomes were compared between the two groups, and the impact of bGS on adverse pathological outcomes was analyzed by logistic regression and biochemical recurrence (BCR)-free survival compared by log-rank test and the Cox proportional hazards model.
Results
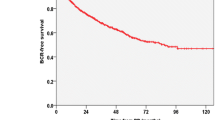
The patients in Group B had a higher rate of extracapsular extension (ECE), seminal vesicle invasion and positive surgical margins than those in Group A (p < 0.001, p = 0.005, p = 0.046, respectively). In univariate and multivariate, bGS was significantly associated with ECE [odds ratio (OR) 2.615, p < 0.001; OR 1.769, p < 0.001]. In Kaplan–Meier analysis, BCR-free survival rate was higher in Group A than in Group B (log rank, p = 0.037). In multivariable Cox regression, maximum percentage of core involvement were strongly associated with BCR [hazard ratio (HR) 1.773 (1.248–2.519), p = 0.001].
Conclusions
pGS 3 + 4 was associated with heterogeneous pathologic and biochemical outcomes according to bGS. Patients with pGS 3 + 4 upgraded from bGS 3 + 3 had more favorable pathological outcomes and biochemical survival outcomes than those with bGS 3 + 4.

Similar content being viewed by others
References
Ploussard G, Epstein JI, Montironi R et al (2011) The contemporary concept of significant versus insignificant prostate cancer. Eur Urol 60(2):291–303
Klotz L, Zhang L, Lam A, Nam R, Mamedov A, Loblaw A (2010) Clinical results of long-term follow-up of a large, active surveillance cohort with localized prostate cancer. J Clin Oncol 28(1):126–131
Cooperberg MR, Carroll PR, Klotz L (2011) Active surveillance for prostate cancer: progress and promise. J Clin Oncol 29(27):3669–3676
Klotz L (2008) Active surveillance for prostate cancer: trials and tribulations. World J Urol 26(5):437–442
Eggener SE, Mueller A, Berglund RK et al (2013) A multi-institutional evaluation of active surveillance for low risk prostate cancer. J Urol 189(1 Suppl):S19–S25 discussion S25
Tosoian JJ, Trock BJ, Landis P et al (2011) Active surveillance program for prostate cancer: an update of the Johns Hopkins experience. J Clin Oncol 29(16):2185–2190
Bul M, van den Bergh RC, Zhu X et al (2012) Outcomes of initially expectantly managed patients with low or intermediate risk screen-detected localized prostate cancer. BJU Int 110(11):1672–1677
Godtman RA, Holmberg E, Khatami A, Stranne J, Hugosson J (2013) Outcome following active surveillance of men with screen-detected prostate cancer. Results from the Göteborg randomised population-based prostate cancer screening trial. Eur Urol 63(1):101–107
Reese AC, Landis P, Han M, Epstein JI, Carter HB (2013) Expanded criteria to identify men eligible for active surveillance of low risk prostate cancer at Johns Hopkins: a preliminary analysis. J Urol 190(6):2033–2038
Vellekoop A, Loeb S, Folkvaljon Y, Stattin P (2014) Population based study of predictors of adverse pathology among candidates for active surveillance with Gleason 6 prostate cancer. J Urol 191(2):350–357
Cooperberg MR, Cowan JE, Hilton JF et al (2011) Outcomes of active surveillance for men with intermediate-risk prostate cancer. J Clin Oncol 29(2):228–234
Epstein JI, Allsbrook WC Jr, Amin MB, Egevad LL; ISUP Grading Committee. The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol. 29(9):1228–1242
Klotz L (2013) Prostate cancer overdiagnosis and overtreatment. Curr Opin Endocrinol Diabetes Obes 20(3):204–209
Klotz L (2013) Active surveillance: patient selection. Curr Opin Urol 23(3):239–244
van As NJ, Norman AR, Thomas K et al (2008) Predicting the probability of deferred radical treatment for localised prostate cancer managed by active surveillance. Eur Urol 54(6):1297–1305
Cookson MS, Aus G, Burnett AL et al (2007) Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American urological association prostate guidelines for localized prostate cancer update panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol 177(2):540–545
Isbarn H, Karakiewicz PI, Ahyai SA et al (2010) Differences in histopathological and biochemical outcomes in patients with low Gleason score prostate cancer. BJU Int 105(6):818–823
Hardie C, Parker C, Norman A et al (2005) Early outcomes of active surveillance for localized prostate cancer. BJU Int 95(7):956–960
Selvadurai ED, Singhera M, Thomas K et al (2013) Medium-term outcomes of active surveillance for localised prostate cancer. Eur Urol 64(6):981–987
Epstein JI, Feng Z, Trock BJ, Pierorazio PM (2012) Upgrading and downgrading of prostate cancer from biopsy to radical prostatectomy: incidence and predictive factors using the modified Gleason grading system and factoring in tertiary grades. Eur Urol 61(5):1019–1024
Kwon O, Kim TJ, Lee IJ, Byun SS, Lee SE, Hong SK (2014) Can contemporary patients with biopsy Gleason score 3 + 4 be eligible for active surveillance? PLoS One 9(9):e109031
Donin NM, Laze J, Zhou M, Ren Q, Lepor H (2013) Gleason 6 prostate tumors diagnosed in the PSA era do not demonstrate the capacity for metastatic spread at the time of radical prostatectomy. Urology 82(1):148–152
Eggener SE, Scardino PT, Walsh PC et al (2011) Predicting 15-year prostate cancer specific mortality after radical prostatectomy. J Urol 185(3):869–875
Kweldam CF, Wildhagen MF, Steyerberg EW, Bangma CH, van der Kwast TH, van Leenders GJ (2014) Cribriform growth is highly predictive for postoperative metastasis and disease-specific death in Gleason score 7 prostate cancer. Mod Pathol
Sowalsky AG, Ye H, Bubley GJ, Balk SP (2013) Clonal progression of prostate cancers from Gleason grade 3 to grade 4. Cancer Res 73(3):1050–1055
Mullins JK, Bonekamp D, Landis P et al (2013) Multiparametric magnetic resonance imaging findings in men with low-risk prostate cancer followed using active surveillance. BJU Int 111(7):1037–1045
Park BH, Jeon HG, Choo SH et al (2014) Role of multiparametric 3.0-Tesla magnetic resonance imaging in patients with prostate cancer eligible for active surveillance. BJU Int 113(6):864–870
Lee DH, Koo KC, Lee SH et al (2013) Low-risk prostate cancer patients without visible tumor (T1c) on multiparametric MRI could qualify for active surveillance candidate even if they did not meet inclusion criteria of active surveillance protocol. Jpn J Clin Oncol 43(5):553–558
Conflict of interest
None of the authors have any conflicts of interest with any institution or product.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jo, J.K., Hong, S.K., Byun, SS. et al. Comparison of clinical outcomes between upgraded pathologic Gleason score 3 + 4 and non-upgraded 3 + 4 prostate cancer among patients who are candidates for active surveillance. World J Urol 33, 1729–1734 (2015). https://doi.org/10.1007/s00345-015-1527-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-015-1527-2