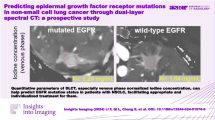
Abstract
Objectives
To explore the role of dual-energy spectral computed tomography (DESCT) quantitative characteristics for the identification of epidermal growth factor receptor (EGFR) mutation status in a cohort of East Asian patients with pulmonary adenocarcinoma.
Materials and methods
Patients with lung adenocarcinoma who underwent both DESCT chest examination and EGFR test were retrospectively selected from our institution’s database. The DESCT visual morphological features and quantitative parameters, including the CT number at 70 keV, normalized iodine concentration (NIC), normalized water concentration, and slopes of the spectral attenuation curves (slope λ HU [Hounsfield unit]), were evaluated or calculated. The patients were divided into two groups: the EGFR mutation group and EGFR wild-type group. Statistical analyses were performed to identify the DESCT quantitative parameters for diagnosis of EGFR mutation status.
Results
EGFR mutations were detected in 66 (55.0%) of the 120 enrolled patients. The univariate analysis revealed that sex, smoking history, CT texture, NIC, and slope λ HU were significantly associated with EGFR mutation status (p = 0.037, 0.001, 0.047, 0.010, and 0.018, respectively). The multivariate logistic analysis revealed that smoking history (odds ratio [OR] = 3.23, p = 0.005) and NIC (OR = 58.026, p = 0.049) were the two significant predictive factors associated with EGFR mutations. Based on this analysis, the smoking history and NIC were combined to determine the predictive value for EGFR mutations with the area under the curve of 0.702.
Conclusions
NIC may be a potential quantitative DESCT parameter for predicting EGFR mutations in patients with pulmonary adenocarcinoma.
Key Points
• DESCT can provide multiple quantitative image parameters compared to conventional CT.
• Identification of the radio-genomic relation between DESCT and EGFR status can help to define molecular subcategories of lung adenocarcinoma, which is valuable for personalized clinical targeted therapy.
• NIC may be a potential DESCT quantitative parameter for predicting EGFR mutations in pulmonary adenocarcinoma.




Similar content being viewed by others
Abbreviations
- ALK:
-
Anaplastic lymphoma kinase
- DESCT:
-
Dual-energy spectral computed tomography
- EGFR:
-
Epidermal growth factor receptor
- GGO:
-
Ground-glass opacity
- GSI:
-
Gemstone spectral imaging
- KRAS:
-
Kirsten rat sarcoma viral oncogene homolog
- NIC:
-
Normalized iodine concentration
- NWC:
-
Normalized water concentration
- PSN:
-
Part-solid nodule
- Slope λ HU:
-
The slope of the spectral Hounsfield unit curve
- SSN:
-
Sub-solid nodule
References
Zhang L, Li M, Wu N, Chen Y (2015) Time trends in epidemiologic characteristics and imaging features of lung adenocarcinoma: a population study of 21,113 cases in China. PLoS One 10:e0136727
Auerbach O, Garfinkel L (1991) The changing pattern of lung carcinoma. Cancer 68:1973–1977
Travis WD (2009) Reporting lung cancer pathology specimens. Impact of the anticipated 7th edition TNM classification based on recommendations of the IASLC staging committee. Histopathology 54:3–11
Zhou C, Wu YL, Chen G et al (2015) Final overall survival results from a randomised, phase III study of erlotinib versus chemotherapy as first-line treatment of EGFR mutation-positive advanced non-small-cell lung cancer (OPTIMAL, CTONG-0802). Ann Oncol 26:1877–1883
Maemondo M, Inoue A, Kobayashi K et al (2010) Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med 362:2380–2388
Rosell R, Moran T, Queralt C et al (2009) Screening for epidermal growth factor receptor mutations in lung cancer. N Engl J Med 361:958–967
Dearden S, Stevens J, Wu YL, Blowers D (2013) Mutation incidence and coincidence in non small-cell lung cancer: meta-analyses by ethnicity and histology (mutMap). Ann Oncol 24:2371–2376
Goo JM, Park CM, Lee HJ (2011) Ground-glass nodules on chest CT as imaging biomarkers in the management of lung adenocarcinoma. AJR Am J Roentgenol 196:533–543
Liu Y, Kim J, Qu F et al (2016) CT features associated with epidermal growth factor receptor mutation status in patients with lung adenocarcinoma. Radiology 280:271–280
Yang Y, Yang Y, Zhou X et al (2015) EGFR L858R mutation is associated with lung adenocarcinoma patients with dominant ground-glass opacity. Lung Cancer 87:272–277
Hong SJ, Kim TJ, Choi YW, Park JS, Chung JH, Lee KW (2016) Radiogenomic correlation in lung adenocarcinoma with epidermal growth factor receptor mutations: imaging features and histological subtypes. Eur Radiol 26:3660–3668
Kim TJ, Lee CT, Jheon SH, Park JS, Chung JH (2016) Radiologic characteristics of surgically resected non-small cell lung cancer with ALK rearrangement or EGFR mutations. Ann Thorac Surg 101:473–480
Cheng Z, Shan F, Yang Y, Shi Y, Zhang Z (2017) CT characteristics of non-small cell lung cancer with epidermal growth factor receptor mutation: a systematic review and meta-analysis. BMC Med Imaging 17:5
Goo HW, Goo JM (2017) Dual-energy CT: new horizon in medical imaging. Korean J Radiol 18:555–569
Johnson TR, Krauss B, Sedlmair M et al (2007) Material differentiation by dual energy CT: initial experience. Eur Radiol 17:1510–1517
Simons D, Kachelriess M, Schlemmer HP (2014) Recent developments of dual-energy CT in oncology. Eur Radiol 24:930–939
De Cecco CN, Darnell A, Rengo M et al (2012) Dual-energy CT: oncologic applications. AJR Am J Roentgenol 199:S98–S105
González-Pérez V, Arana E, Barrios M et al (2016) Differentiation of benign and malignant lung lesions: dual-energy computed tomography findings. Eur J Radiol 85:1765–1772
Wang G, Zhang C, Li M, Deng K, Li W (2014) Preliminary application of high-definition computed tomographic gemstone spectral imaging in lung cancer. J Comput Assist Tomogr 38:77–81
Sudarski S, Hagelstein C, Weis M, Schoenberg SO, Apfaltrer P (2015) Dual-energy snap-shot perfusion CT in suspect pulmonary nodules and masses and for lung cancer staging. Eur J Radiol 84:2393–2400
Hou WS, Wu HW, Yin Y, Cheng JJ, Zhang Q, Xu JR (2015) Differentiation of lung cancers from inflammatory masses with dual-energy spectral CT imaging. Acad Radiol 22:337–344
Otrakji A, Digumarthy SR, Lo Gullo R, Flores EJ, Shepard JA, Kalra MK (2016) Dual-energy CT: spectrum of thoracic abnormalities. Radiographics 36:38–52
Chae EJ, Song JW, Seo JB, Krauss B, Jang YM, Song KS (2008) Clinical utility of dual-energy CT in the evaluation of solitary pulmonary nodules: initial experience. Radiology 249:671–681
Remy-Jardin M, Faivre JB, Pontana F, Molinari F, Tacelli N, Remy J (2014) Thoracic applications of dual energy. Semin Respir Crit Care Med 35:64–73
Ohana M, Jeung MY, Labani A, El Ghannudi S, Roy C (2014) Thoracic dual energy CT: acquisition protocols, current applications and future developments. Diagn Interv Imaging 95:1017–1026
Hansell DM, Bankier AA, MacMahon H, McLoud TC, Müller NL, Remy J (2008) Fleischner society: glossary of terms for thoracic imaging. Radiology 246:697–722
Godoy MC, Naidich DP (2009) Subsolid pulmonary nodules and the spectrum of peripheral adenocarcinomas of the lung: recommended interim guidelines for assessment and management. Radiology 253:606–622
Raad RA, Suh J, Harari S, Naidich DP, Shiau M, Ko JP (2014) Nodule characterization: subsolid nodules. Radiol Clin North Am 52:47–67
Truong MT, Ko JP, Rossi SE et al (2014) Update in the evaluation of the solitary pulmonary nodule. Radiographics 34:1658–1679
Wang L, Liu B, Wu XW et al (2012) Correlation between CT attenuation value and iodine concentration in vitro: discrepancy between gemstone spectral imaging on single-source dual-energy CT and traditional polychromatic X-ray imaging. J Med Imaging Radiat Oncol 56:379–383
Knöss N, Hoffmann B, Krauss B, Heller M, Biederer J (2011) Dual energy computed tomography of lung nodules: differentiation of iodine and calcium in artificial pulmonary nodules in vitro. Eur J Radiol 80:e516–e519
Manning BD, Cantley LC (2007) AKT/PKB signaling: navigating downstream. Cell 129:1261–1274
Gordan JD, Simon MC (2007) Hypoxia-inducible factors: central regulators of the tumor phenotype. Curr Opin Genet Dev 17:71–77
Matsuda I, Akahane M, Sato J et al (2012) Precision of the measurement of CT numbers: comparison of dual-energy CT spectral imaging with fast kVp switching and conventional CT with phantoms. Jpn J Radiol 30:34–39
Patino M, Prochowski A, Agrawal MD et al (2016) Material separation using dual-energy CT: current and emerging applications. Radiographics 36:1087–1105
Travis WD, Brambilla E, Noguchi M et al (2011) International association for the study of lung cancer/American thoracic society/European respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 6:244–285
Lee HJ, Kim YT, Kang CH et al (2013) Epidermal growth factor receptor mutation in lung adenocarcinomas: relationship with CT characteristics and histologic subtypes. Radiology 268:254–264
Aoki M, Takai Y, Narita Y et al (2014) Correlation between tumor size and blood volume in lung tumors: a prospective study on dual-energy gemstone spectral CT imaging. J Radiat Res 55:917–923
Kawai T, Shibamoto Y, Hara M, Arakawa T, Nagai K, Ohashi K (2011) Can dual-energy CT evaluate contrast enhancement of ground-glass attenuation? Phantom and preliminary clinical studies. Acad Radiol 18:682–689
Ascenti G, Mileto A, Krauss B et al (2013) Distinguishing enhancing from nonenhancing renal masses with dual-source dual-energy CT: iodine quantification versus standard enhancement measurements. Eur Radiol 23:2288–2295
Funding
This study has received funding by the National Natural Science Foundation of China (Grant No. 81601494) and the PUMC Youth Fund/Fundamental Research Funds for the Central Universities (Grant No. 3332016030).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Ning Wu.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
Ni Li kindly provided statistical advice for this manuscript.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• observational
• performed at one institution
Rights and permissions
About this article
Cite this article
Li, M., Zhang, L., Tang, W. et al. Identification of epidermal growth factor receptor mutations in pulmonary adenocarcinoma using dual-energy spectral computed tomography. Eur Radiol 29, 2989–2997 (2019). https://doi.org/10.1007/s00330-018-5756-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5756-9