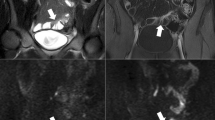
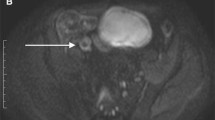
Abstract
Objectives
To quantify intravoxel incoherent motion (IVIM)-DWI and dynamic contrast-enhanced (DCE)-MRI parameters in normal and abnormal ileal segments in Crohn’s disease (CD) patients and to assess the association of these parameters with clinical and MRI-based measurements of CD activity.
Methods
In this prospective study, 27 CD patients (M/F 18/9, mean age 42 years) underwent MR enterography, including IVIM-DWI and DCE-MRI. IVIM-DWI and DCE-MRI parameters were quantified in normal and abnormal small bowel segments, the latter identified by the presence of inflammatory changes. MRI parameter differences between normal and abnormal bowel were tested using Wilcoxon signed-rank tests. IVIM-DWI and DCE-MRI parameters were correlated with clinical data (C-reactive protein, Harvey-Bradshaw Index), conventional MRI parameters (wall thickness, length of involvement) and MRI activity scores (MaRIA, Clermont). Diagnostic performance of (combined) parameters for differentiation between normal and abnormal bowel was determined using ROC analysis.
Results
The DCE-MRI parameters peak concentration Cpeak, upslope, area-under-the-curve at 60s (AUC60), Ktrans and ve were significantly increased (p<0.023), while IVIM-DWI parameters perfusion fraction (PF) and ADC were significantly decreased (p<0.001) in abnormal bowel segments. None of the DCE-MRI and IVIM-DWI parameters correlated with clinical parameters (p>0.105). DCE-MRI parameters exhibited multiple significant correlations with wall thickness (Cpeak, upslope, AUC60, Ktrans; r range 0.431–0.664, p<0.025) and MaRIA/Clermont scores (Cpeak, AUC60, Ktrans; r range 0.441–0.617, p<0.021). Combined Ktrans+ve+PF+ADC showed highest AUC (0.963) for differentiation between normal and abnormal bowel, while ADC performed best for individual parameters (AUC=0.800).
Conclusions
DCE-MRI and IVIM-DWI, particularly when used in combination, are promising for non-invasive evaluation of small bowel CD.
Key Points
• IVIM-DWI and DCE-MRI parameters were significantly different between normal and abnormal bowel segments in CD patients.
• DCE-MRI parameters showed a significant association with wall thickness and MRI activity scores.
• Combination of IVIM-DWI and DCE-MRI parameters led to the highest diagnostic performance for differentiation between normal and abnormal bowel segments, while ADC showed the highest diagnostic performance of individual parameters.



Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AIF:
-
Arterial input function
- AUC60:
-
Area under the curve at 60 s
- [CA]:
-
Contrast agent concentration
- CD:
-
Crohn’s disease
- CDEIS:
-
Crohn’s Disease Endoscopic Index of Severity
- Cpeak :
-
Peak concentration
- CRP:
-
C-reactive protein
- D:
-
Diffusion coefficient
- D*:
-
Pseudodiffusion coefficient
- DCE-MRI:
-
Dynamic contrast-enhanced MRI
- DWI:
-
Diffusion-weighted imaging
- HBI:
-
Harvey-Bradshaw index
- IVIM-DWI:
-
Intravoxel incoherent motion diffusion-weighted imaging
- kep :
-
Wash-out constant
- Ktrans :
-
Transfer constant
- MaRIA:
-
Magnetic Resonance Index of Activity
- OR:
-
Odds ratio
- PF:
-
Perfusion fraction
- RCE:
-
Relative contrast enhancement
- ROI:
-
Region of interest
- SD:
-
Standard deviation
- SI:
-
Signal intensity
- T1WI:
-
T1-weighted imaging
- T2WI:
-
T2-weighted imaging
- TTP:
-
Time-to-peak
- VFA:
-
Variable flip angle
- vp :
-
Plasma volume fraction
References
Peyrin-Biroulet L, Loftus EV Jr, Colombel JF, Sandborn WJ (2010) The natural history of adult Crohn's disease in population-based cohorts. Am J Gastroenterol 105:289–297
Van Assche G, Dignass A, Reinisch W et al (2010) The second European evidence-based Consensus on the diagnosis and management of Crohn's disease: Special situations. J Crohns Colitis 4:63–101
Bhatnagar G, Von Stempel C, Halligan S, Taylor SA (2017) Utility of MR enterography and ultrasound for the investigation of small bowel Crohn's disease. J Magn Reson Imaging 45:1573–1588
Coimbra AJ, Rimola J, O'Byrne S et al (2016) Magnetic resonance enterography is feasible and reliable in multicenter clinical trials in patients with Crohn's disease, and may help select subjects with active inflammation. Aliment Pharmacol Ther 43:61–72
Rimola J, Ordas I, Rodriguez S et al (2011) Magnetic resonance imaging for evaluation of Crohn's disease: validation of parameters of severity and quantitative index of activity. Inflamm Bowel Dis 17:1759–1768
Hordonneau C, Buisson A, Scanzi J et al (2014) Diffusion-weighted magnetic resonance imaging in ileocolonic Crohn's disease: validation of quantitative index of activity. Am J Gastroenterol 109:89–98
Florie J, Wasser MN, Arts-Cieslik K, Akkerman EM, Siersema PD, Stoker J (2006) Dynamic contrast-enhanced MRI of the bowel wall for assessment of disease activity in Crohn's disease. AJR. Am J Roentgenol 186:1384–1392
Oto A, Kayhan A, Williams JT et al (2011) Active Crohn's disease in the small bowel: evaluation by diffusion weighted imaging and quantitative dynamic contrast enhanced MR imaging. J Magn Reson Imaging 33:615–624
Tielbeek JA, Ziech ML, Li Z et al (2014) Evaluation of conventional, dynamic contrast enhanced and diffusion weighted MRI for quantitative Crohn's disease assessment with histopathology of surgical specimens. Eur Radiol 24:619–629
Zhu J, Zhang F, Luan Y et al (2016) Can Dynamic Contrast-Enhanced MRI (DCE-MRI) and Diffusion-Weighted MRI (DW-MRI) Evaluate Inflammation Disease: A Preliminary Study of Crohn's Disease. Medicine (Baltimore) 95:e3239
Ziech ML, Lavini C, Bipat S et al (2013) Dynamic contrast-enhanced MRI in determining disease activity in perianal fistulizing Crohn disease: a pilot study. AJR Am J Roentgenol 200:W170–W177
Bhatnagar G, Dikaios N, Prezzi D, Vega R, Halligan S, Taylor SA (2015) Changes in dynamic contrast-enhanced pharmacokinetic and diffusion-weighted imaging parameters reflect response to anti-TNF therapy in Crohn's disease. Br J Radiol 88:20150547
Zhu J, Zhang F, Zhou J, Li H (2017) Assessment of therapeutic response in Crohn's disease using quantitative dynamic contrast enhanced MRI (DCE-MRI) parameters: A preliminary study. Medicine (Baltimore) 96:e7759
Le Bihan D, Breton E, Lallemand D, Aubin ML, Vignaud J, Laval-Jeantet M (1988) Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology 168:497–505
Koh DM, Collins DJ, Orton MR (2011) Intravoxel incoherent motion in body diffusion-weighted MRI: reality and challenges. AJR Am J Roentgenol 196:1351–1361
Freiman M, Perez-Rossello JM, Callahan MJ et al (2013) Characterization of fast and slow diffusion from diffusion-weighted MRI of pediatric Crohn's disease. J Magn Reson Imaging 37:156–163
Harvey RF, Bradshaw JM (1980) A simple index of Crohn's-disease activity. Lancet 1:514
Yoon K, Chang KT, Lee HJ (2015) MRI for Crohn's Disease: Present and Future. Biomed Res Int 2015:786802
Rimola J, Alvarez-Cofino A, Perez-Jeldres T et al (2017) Comparison of three magnetic resonance enterography indices for grading activity in Crohn's disease. J Gastroenterol 52:585–593
Hectors SJ, Besa C, Wagner M et al (2017) DCE-MRI of the prostate using shutter-speed vs. Tofts model for tumor characterization and assessment of aggressiveness. J Magn Reson Imaging 46:837–849
Li X, Priest RA, Woodward WJ et al (2013) Feasibility of shutter-speed DCE-MRI for improved prostate cancer detection. Magn Reson Med 69:171–178
Rohrer M, Bauer H, Mintorovitch J, Requardt M, Weinmann HJ (2005) Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Investig Radiol 40:715–724
Zhang X, Petersen ET, Ghariq E et al (2013) In vivo blood T(1) measurements at 1.5 T, 3 T, and 7 T. Magn Reson Med 70:1082–1086
Sourbron SP, Buckley DL (2011) On the scope and interpretation of the Tofts models for DCE-MRI. Magn Reson Med 66:735–745
Dyvorne HA, Galea N, Nevers T et al (2013) Diffusion-weighted imaging of the liver with multiple b values: effect of diffusion gradient polarity and breathing acquisition on image quality and intravoxel incoherent motion parameters--a pilot study. Radiology 266:920–929
Orton MR, Collins DJ, Koh DM, Leach MO (2014) Improved intravoxel incoherent motion analysis of diffusion weighted imaging by data driven Bayesian modeling. Magn Reson Med 71:411–420
Danese S, Sans M, de la Motte C et al (2006) Angiogenesis as a novel component of inflammatory bowel disease pathogenesis. Gastroenterology 130:2060–2073
Carr ND, Pullan BR, Schofield PF (1986) Microvascular studies in non-specific inflammatory bowel disease. Gut 27:542–549
Oto A, Zhu F, Kulkarni K, Karczmar GS, Turner JR, Rubin D (2009) Evaluation of diffusion-weighted MR imaging for detection of bowel inflammation in patients with Crohn's disease. Acad Radiol 16:597–603
Dohan A, Taylor S, Hoeffel C et al (2016) Diffusion-weighted MRI in Crohn's disease: Current status and recommendations. J Magn Reson Imaging 44:1381–1396
Wagner M, Ko HM, Chatterji M et al (2018) Magnetic resonance imaging predicts histopathologic composition of ileal Crohn's disease. J Crohns Colitis. https://doi.org/10.1093/ecco-jcc/jjx186
Li XH, Mao R, Huang SY et al (2018) Characterization of Degree of Intestinal Fibrosis in Patients with Crohn Disease by Using Magnetization Transfer MR Imaging. Radiology 287:494–503
Funding
This study has received funding by research grants from Guerbet LLC and the Sanford J Grossman Charitable Trust for Integrative Studies in IBD at Mount Sinai.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Bachir Taouli, MD.
Conflict of interest
The authors of this manuscript declare relationships with the following companies: Jean-Frederic Colombel is a consultant for AbbVie, Amgen, Boehringer-Ingelheim, Celgene Corporation, Celltrion, Enterome, Ferring, Genentech, Janssen and Janssen, Medimmune, Merck & Co., Pfizer, Protagonist, Second Genome, Seres, Shire, Takeda and Theradiag, a speaker for AbbVie, Ferring and Speaker’s bureau for Amgen and received grant support from AbbVie, Takeda and Janssen and Janssen. Bachir Taouli received grant support from Guerbet and Bayer.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• prospective
• observational
• performed at one institution
Rights and permissions
About this article
Cite this article
Hectors, S.J., Gordic, S., Semaan, S. et al. Diffusion and perfusion MRI quantification in ileal Crohn’s disease. Eur Radiol 29, 993–1002 (2019). https://doi.org/10.1007/s00330-018-5627-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5627-4