Abstract
Objectives
To develop criteria to improve discrimination between vertebral metastases from neuroendocrine tumours (NETs) and benign bone lesions on PET combined with CT using DOTA-D-Phe1-Tyr3-octreotide labelled with gallium-68 (68Ga-DOTA-TOC).
Methods
In 535 NET patients, 68Ga-DOTA-TOC PET/CT examinations were reviewed retrospectively for vertebral CT lesions and/or PET foci. For each vertebral PET abnormality, appearance on CT, biological volume (BV), standardized uptake value (SUVmax) and ratios to those of reference organs were determined. All vertebral abnormalities were characterized as a metastasis, a typical vertebral haemangioma (VH) or other benign lesion.
Results
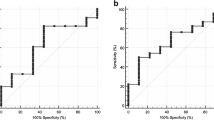
In 79 patients (14.8 %), we found 107 metastases, 34 VHs and 31 other benign lesions in the spine. The optimal cut-off values to differentiate metastases from benign lesions were BV ≥0.72 cm3, SUVmax ≥2, SUVmax ratio to a reference vertebra ≥2.1, to liver ≥0.28 and to spleen ≥0.14. They corresponded to lesion-based 68Ga-DOTA-TOC PET/CT sensitivity of 87 %, 98 %, 97 %, 99 % and 94 %, and specificity of 55 %, 100 %, 90 %, 97 %, 100 %, respectively.
Conclusions
The high sensitivity of 68Ga-DOTA-TOC-PET/CT in detecting NET vertebral metastases was confirmed; this study showed that specificity could be improved by combining CT features and quantifying 68Ga-DOTA-TOC uptake.
Key Points
• Bone metastases in neuroendocrine tumours correlate with prognosis.
• Benign bone lesions may mimic metastases on 68 Ga-DOTA-TOC PET/CT imaging.
• The specific polka-dot CT pattern may be missing in some vertebral haemangiomas.
• Lesion atypical for haemangiomas can be better characterized by quantifying 68 Ga-DOTA-TOC uptake.




Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the curve
- BV:
-
Biological volume
- CT:
-
Computed tomography
- DOTA:
-
1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid
- DOTA-TOC:
-
Edotreotide
- FDG:
-
18F-fluorodeoxyglucose
- 68Ga:
-
Gallium-68
- MRI:
-
Magnetic resonance imaging
- NET:
-
Neuroendocrine tumour
- NPV:
-
Negative predictive value
- OSEM:
-
Ordered Subsets Expectation-Maximization
- PET:
-
Positron emission tomography
- PPV:
-
Positive predictive value
- ROC:
-
Receiver operating characteristic
- RV:
-
Reference vertebra
- Se:
-
Sensitivity
- SOT:
-
Standard of truth
- Sp:
-
Specificity
- SR:
-
Somatostatin receptor
- SR-2:
-
Somatostatin receptors subtype 2
- SUV:
-
Standardized uptake value
- TOC:
-
Tyr3-octreotide
- VH:
-
Vertebral haemangioma
- VOI:
-
Volume of interest
References
Ross EM, Roberts WC (1985) The carcinoid syndrome: comparison of 21 necropsy subjects with carcinoid heart disease to 15 necropsy subjects without carcinoid heart disease. Am J Med 79:339–354
Panzuto F, Nasoni S, Falconi M et al (2005) Prognostic factors and survival in endocrine tumor patients: comparison between gastrointestinal and pancreatic localization. Endocr Relat Cancer 12:1083–1092
Skoura E, Michopoulou S, Mohmaduvesh M et al (2016) The impact of 68Ga-DOTATATE PET/CT imaging on management of patients with neuroendocrine tumors: experience from a national referral center in the United Kingdom. J Nucl Med 57:34–40
Albanus DR, Apitzsch J, Erdem Z et al (2015) Clinical value of 68Ga-DOTATATE-PET/CT compared to stand-alone contrast enhanced CT for the detection of extra-hepatic metastases in patients with neuroendocrine tumours (NET). Eur J Radiol 84:1866–1872
Putzer D, Gabriel M, Henninger B et al (2009) Bone metastases in patients with neuroendocrine tumor: 68Ga-DOTA-Tyr3-Octreotide PET in comparison to CT and bone scintigraphy. J Nucl Med 50:1214–1221
Kroiss A, Putzer D, Decristoforo C et al (2013) 68Ga-DOTA-TOC uptake in neuroendocrine tumour and healthy tissue: differentiation of physiological uptake and pathological processes in PET/CT. Eur J Nucl Med Mol Imaging 40:514–523
Prasad V, Baum RP (2010) Biodistribution of the Ga-68 labeled somatostatin analogue DOTA-NOC in patients with neuroendocrine tumors: characterization of uptake in normal organs and tumor lesions. Q J Nucl Med Mol Imaging 54:61–67
Ambrosini V, Nanni C, Zompatori M et al (2010) (68)Ga-DOTA-NOC PET/CT in comparison with CT for the detection of bone metastasis in patients with neuroendocrine tumours. Eur J Nucl Med Mol Imaging 37:722–727
Gabriel M, Decristoforo C, Kendler D et al (2007) 68Ga-DOTA-Tyr3-Octreotide PET in neuroendocrine tumors: comparison with somatostatin receptor scintigraphy and CT. J Nucl Med 48:508–518
Van Binnebeek S, Vanbilloen B, Baete K et al (2016) Comparison of diagnostic accuracy of 111In-pentetreotide SPECT and 68Ga-DOTATOC PET/CT: A lesion-by-lesion analysis in patients with metastatic neuroendocrine tumours. Eur Radiol 26:900–909
Slon V, Stein D, Cohen H et al (2015) Vertebral hemangiomas: their demographical characteristics, location along the spine and position within the vertebral body. Eur Spine J 24:2189–2195
Gaudino S, Martucci M, Colantonio R et al (2015) A systematic approach to vertebral hemangioma. Skeletal Radiol 44:25–36
Gilardi L, Vadrucci M, Grana CM (2017) Multiple vertebral hemangiomas: a potential pitfall in 68Ga-DOTATOC PET/CT interpretation. Endocrine 55:992–993
Skoura E, Alshammari A, Syed R et al (2015) Adolescent with 68Ga DOTATATE–avid vertebral hemangioma mimicking metastasis in PET imaging. Clin Nucl Med 40:e378–e379
Brogsitter C, Hofmockel T, Kotzerke J (2014) 68Ga DOTATATE uptake in vertebral hemangioma. Clin Nucl Med 39:462–463
Klinaki I, Al-Nahhas A, Soneji N, Win Z (2013) 68Ga DOTATATE PET/CT uptake in spinal lesions and MRI correlation on a patient with neuroendocrine tumor: potential pitfalls. Clin Nucl Med 38:e449–e453
Persaud T (2008) The Polka-dot sign. Radiology 246:980–981
Erlemann R (2006) Imaging and differential diagnosis of primary bone tumors and tumor-like lesions of the spine. Eur J Radiol 58:48–67
Balogova S, Talbot J-N, Nataf V et al (2013) 18F-fluorodihydroxyphenylalanine vs other radiopharmaceuticals for imaging neuroendocrine tumours according to their type. Eur J Nucl Med Mol Imaging 40:943–966
Thapa P, Ranade R, Ostwal V et al (2016) Performance of 177Lu-DOTATATE-based peptide receptor radionuclide therapy in metastatic gastroenteropancreatic neuroendocrine tumor: a multiparametric response evaluation correlating with primary tumor site, tumor proliferation index, and dual tracer imaging characteristics. Nucl Med Commun 37:1030–1037
Grillo F, Albertelli M, Brisigotti MP et al (2016) Grade increases in gastroenteropancreatic neuroendocrine tumor metastases compared to the primary tumor. Neuroendocrinology 103:452–459
Bucy PC (1929) The pathology of hemangioma of bone. Am J Pathol 5:381–388
Adams RL, Adams IP, Lindow SW et al (2005) Somatostatin receptors 2 and 5 are preferentially expressed in proliferating endothelium. Br J Cancer 92:1493–1498
Makhija M, Bofill ER (1988) Hemangioma, a rare cause of photopenic lesion on skeletal imaging. Clin Nucl Med 13:661–662
Jaimini A, D’Souza MM, Seniaray N et al (2016) Characterization of ‘cold’ vertebrae on 18F-FDG PET/CT. Nucl Med Commun 37:30–42
Domínguez M, Rayo J, Serrano J et al (2011) Vertebral hemangioma: ‘Cold’ vertebrae on bone scintigraphy and fluordeoxy-glucose positron emission tomography-computed tomography. Indian J Nucl Med 26:49–51
Savelli G, Perotti V, Rosso E et al (2016) 18F-fluorocholine PET/CT finding of a vertebral hemangioma. Clin Nucl Med 41:e390–e391
Nakayama M, Okizaki A, Ishitoya S, Aburano T (2012) ‘Hot’ vertebra on 18F-FDG PET scan: a case of vertebral hemangioma. Clin Nucl Med 37:1190–1193
Kazmierczak PM, Rominger A, Wenter V et al (2017) The added value of 68Ga-DOTA-TATE-PET to contrast-enhanced CT for primary site detection in CUP of neuroendocrine origin. Eur Radiol 27:1676–1684
Cives M, Rizzo F, Simone V et al (2016) Reviewing the osteotropism in neuroendocrine tumors: The role of epithelial-mesenchymal transition. Neuroendocrinology 103:321–334
Van Loon K, Zhang L, Keiser J et al (2015) Bone metastases and skeletal-related events from neuroendocrine tumors. Endocr Connect 4:9–17
Gauthé M, Richard-Molard M, Fayard J et al (2017) Prognostic impact of tumour burden assessed by metabolic tumour volume on FDG PET/CT in anal canal cancer. Eur J Nucl Med Mol Imaging 44:63–70
Dibble EH, Alvarez ACL, Truong M-T et al (2012) 18F-FDG metabolic tumor volume and total glycolytic activity of oral cavity and oropharyngeal squamous cell cancer: adding value to clinical staging. J Nucl Med 53:709–715
Riihimäki M, Hemminki A, Sundquist K et al (2016) The epidemiology of metastases in neuroendocrine tumors. Int J Cancer 139:2679–2686
Acknowledgements
This study includes the data of a ‘Projet de recherche clinique’ PHRC P040303 sponsored by Assistance Publique-Hôpitaux de Paris monitored by Mrs Zakia Idir. We acknowledge the help of the Cancer Est Clinical Research Assistants during the clinical trial. We thank the referring physicians for their confidence and the medical technologists of our department for their commitment to PET/CT imaging. We thank Professor Ralph McCready for his assistance with editing the manuscript.
Funding
The authors state that this retrospective analysis of data of a prospective series has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. J.N. Talbot.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic study
• performed at one institution
Rights and permissions
About this article
Cite this article
Gauthé, M., Testart Dardel, N., Ruiz Santiago, F. et al. Vertebral metastases from neuroendocrine tumours: How to avoid false positives on 68Ga-DOTA-TOC PET using CT pattern analysis?. Eur Radiol 28, 3943–3952 (2018). https://doi.org/10.1007/s00330-017-5294-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5294-x