Abstract
Objectives
To determine the degree of relationship between iodine concentrations derived from dual-energy CT (DECT) and perfusion CT parameters in patients with advanced HCC under treatment.
Methods
In this single-centre IRB approved study, 16 patients with advanced HCC treated with sorafenib or radioembolization who underwent concurrent dynamic perfusion CT and multiphase DECT using a single source, fast kV switching DECT scanner were included. Written informed consent was obtained for all patients. HCC late-arterial and portal iodine concentrations, blood flow (BF)-related and blood volume (BV)-related perfusion parameters maps were calculated. Mixed-effects models of the relationship between iodine concentrations and perfusion parameters were computed. An adjusted p value (Bonferroni method) < 0.05 was considered significant.
Results
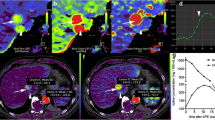
Mean HCC late-arterial and portal iodine concentrations were 22.7±12.7 mg/mL and 18.7±8.3 mg/mL, respectively. Late-arterial iodine concentration was significantly related to BV (mixed-effects model F statistic (F)=28.52, p<0.0001), arterial BF (aBF, F=17.62, p<0.0001), hepatic perfusion index (F=28.24, p<0.0001), positive enhancement integral (PEI, F=66.75, p<0.0001) and mean slope of increase (F=32.96, p<0.0001), while portal-venous iodine concentration was mainly related to BV (F=29.68, p<0.0001) and PEI (F=66.75, p<0.0001).
Conclusions
In advanced HCC lesions, DECT-derived late-arterial iodine concentration is strongly related to both aBF and BV, while portal iodine concentration mainly reflects BV, offering DECT the ability to evaluate both morphological and perfusion changes.
Key points
• Late-arterial iodine concentration is highly related to arterial BF and BV.
• Portal iodine concentration mainly reflects tumour blood volume.
• Dual-energy CT offers significantly decreased radiation dose compared with perfusion CT.



Similar content being viewed by others
Abbreviations
- DECT:
-
Dual-energy CT
- BF:
-
Blood flow
- BV:
-
Blood volume
- aBF:
-
Arterial blood flow
- pBF:
-
Portal blood flow
- HPI:
-
Hepatic perfusion index
- MTT:
-
Mean transit time
- MSI:
-
Mean slope of increase
- PS:
-
Capillary permeability surface product
- TTP:
-
Time to peak
- PEI:
-
Positive enhancement integral
- AASLD:
-
American Association for the Study of Liver Diseases
References
Miles KA (2003) Functional CT imaging in oncology. Eur Radiol 13:M134–M138
Sahani DV, Holalkere NS, Mueller PR, Zhu AX (2007) Advanced hepatocellular carcinoma: CT perfusion of liver and tumor tissue--initial experience. Radiology 243:736–743
Ippolito D, Capraro C, Casiraghi A, Cestari C, Sironi S (2012) Quantitative assessment of tumour associated neovascularisation in patients with liver cirrhosis and hepatocellular carcinoma: role of dynamic-CT perfusion imaging. Eur Radiol 22:803–811
Jiang T, Kambadakone A, Kulkarni NM, Zhu AX, Sahani DV (2012) Monitoring response to antiangiogenic treatment and predicting outcomes in advanced hepatocellular carcinoma using image biomarkers, CT perfusion, tumor density, and tumor size (RECIST). Invest Radiol 47:11–17
Ippolito D, Fior D, Bonaffini PA et al (2014) Quantitative evaluation of CT-perfusion map as indicator of tumor response to transarterial chemoembolization and radiofrequency ablation in HCC patients. Eur J Radiol 83:1665–1671
Dai X, Schlemmer HP, Schmidt B et al (2013) Quantitative therapy response assessment by volumetric iodine-uptake measurement: initial experience in patients with advanced hepatocellular carcinoma treated with sorafenib. Eur J Radiol 82:327–334
Kim SH, Kamaya A, Willmann JK (2014) CT perfusion of the liver: principles and applications in oncology. Radiology 272:322–344
Marin D, Boll DT, Mileto A, Nelson RC (2014) State of the art: dual-energy CT of the abdomen. Radiology 271:327–342
Matsumoto K, Jinzaki M, Tanami Y, Ueno A, Yamada M, Kuribayashi S (2011) Virtual monochromatic spectral imaging with fast kilovoltage switching: improved image quality as compared with that obtained with conventional 120-kVp CT. Radiology 259:257–262
Stiller W, Skornitzke S, Fritz F et al (2015) Correlation of quantitative dual-energy computed tomography iodine maps and abdominal computed tomography perfusion measurements: are single-acquisition dual-energy computed tomography iodine maps more than a reduced-dose surrogate of conventional computed tomography perfusion? Invest Radiol 50:703–708
Lv P, Liu J, Yan X et al (2017) CT spectral imaging for monitoring the therapeutic efficacy of VEGF receptor kinase inhibitor AG-013736 in rabbit VX2 liver tumours. Eur Radiol 27:918–926
Gordic S, Puippe GD, Krauss B et al (2016) Correlation between Dual-Energy and Perfusion CT in Patients with Hepatocellular Carcinoma. Radiology 280:78–87
Thaiss WM, Haberland U, Kaufmann S et al (2016) Iodine concentration as a perfusion surrogate marker in oncology: Further elucidation of the underlying mechanisms using Volume Perfusion CT with 80 kVp. Eur Radiol 26:2929–2936
Kagawa Y, Okada M, Yagyu Y et al (2013) Optimal scan timing of hepatic arterial-phase imaging of hypervascular hepatocellular carcinoma determined by multiphasic fast CT imaging technique. Acta Radiol 54:843–850
Hammerstingl RM, Vogl TJ (2005) Abdominal MDCT: protocols and contrast considerations. Eur Radiol 15:E78–E90
Vilgrain V, Abdel-Rehim M, Sibert A et al (2014) Radioembolisation with yttrium90 microspheres versus sorafenib for treatment of advanced hepatocellular carcinoma (SARAH): study protocol for a randomised controlled trial. Trials 15:474
Bruix J, Sherman M (2011) Management of hepatocellular carcinoma: an update. Hepatology 53:1020–1022
American College of Radiology. Liver Imaging Reporting and Data System version 2014. Available via http://www.acr.org/Quality-Safety/Resources/LIRADS. Accessed April 2016
St Lawrence KS, Lee TY (1998) An adiabatic approximation to the tissue homogeneity model for water exchange in the brain: I. Theoretical derivation. J Cereb Blood Flow Metab 18:1365–1377
Kaufmann S, Horger T, Oelker A et al (2015) Characterization of hepatocellular carcinoma (HCC) lesions using a novel CT-based volume perfusion (VPCT) technique. Eur J Radiol 84:1029–1035
Miles KA (2003) Perfusion CT for the assessment of tumour vascularity: which protocol? Br J Radiol 76:S36–S42
Ng CS, Hobbs BP, Wei W et al (2015) Effect on perfusion values of sampling interval of computed tomographic perfusion acquisitions in neuroendocrine liver metastases and normal liver. J Comput Assist Tomogr 39:373–382
Miles KA, Lee TY, Goh V et al (2012) Current status and guidelines for the assessment of tumour vascular support with dynamic contrast-enhanced computed tomography. Eur Radiol 22:1430–1441
Djuric-Stefanovic A, Saranovic D, Masulovic D, Ivanovic A, Pesko P (2013) Comparison between the deconvolution and maximum slope 64-MDCT perfusion analysis of the esophageal cancer: is conversion possible? Eur J Radiol 82:1716–1723
Ng CS, Hobbs BP, Chandler AG et al (2013) Metastases to the liver from neuroendocrine tumors: effect of duration of scan acquisition on CT perfusion values. Radiology 269:758–767
NCD Risk Factor Collaboration (2016) Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 387:1377–1396
Ng CS, Chandler AG, Wei W et al (2012) Effect of dual vascular input functions on CT perfusion parameter values and reproducibility in liver tumors and normal liver. J Comput Assist Tomogr 36:388–393
Chen BB, Hsu CY, Yu CW et al (2017) Early perfusion changes within 1 week of systemic treatment measured by dynamic contrast-enhanced MRI may predict survival in patients with advanced hepatocellular carcinoma. Eur Radiol 27:3069–3079
Acknowledgements
This work was an ancillary study to the SARAH multicentric trial (ClinicalTrials.gov identifier NCT01482442).
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Pr. Alain Luciani.
Conflict of interest
Two authors of this manuscript (R.Q. and P.R.) are employees of GE Healthcare. All other authors retained full control of all data and were responsible for all analyses performed in the study.
Statistics and biometry
Revised statistical analysis was performed by A.T., a PhD statistician with 11 years of experience.
Informed consent
Written informed consent was obtained from all patients in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• prospective
• cross sectional study
• performed at one institution
Rights and permissions
About this article
Cite this article
Mulé, S., Pigneur, F., Quelever, R. et al. Can dual-energy CT replace perfusion CT for the functional evaluation of advanced hepatocellular carcinoma?. Eur Radiol 28, 1977–1985 (2018). https://doi.org/10.1007/s00330-017-5151-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5151-y