Abstract
Whereas Arctic benthic decapods are not a species-rich group, they can dominate the local epifauna and play important roles in the ecosystem. We studied the decapod fauna from Isfjorden (Svalbard, Norway, 78°N) by sampling from 22 stations and comparing with 50 and 100-year-old data from the same localities. Our data provide no evidence of changes in the species composition of decapods during the last 50 years. Hence, we do not observe the poleward shift of species found in several pelagic communities in the North Atlantic. However, there is statistical evidence for changes in the community structure between 1908 and both 1958 and 2007. The main change is a shift towards communities more dominated by the spider crab Hyas araneus and the hermit crab Pagurus pubescens in 2007, and with a corresponding decrease in the two shrimp species Lebbeus polaris and Spirontocaris spinus. These shrimps are specialist predators compared to the more opportunistic, scavenging crabs. We argue that increased disturbance levels may be a causal factor behind the observed community change, with likely sources of disturbance including trawling and climatic fluctuations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Decadal and multi-decadal variability of the Arctic climate is well documented (Polyakov and Johnson, 2000) and revealed in time series of atmospheric, oceanic and sea ice parameters (Divine and Dick 2006; Polyakov et al. 2004; Johanessen et al. 2004). Decadal variability is linked to changes in the large-scale atmospheric pressure fields, whilst the lower frequency, multi-decadal variability is thought to be linked to fluctuations in ocean circulation (e.g. Polyakov et al. 2004). Long-term data series are perhaps the best way of identifying drivers of community change, but such time series are rare for Arctic marine systems. As the economic and scientific interests intensify in the Arctic, it is important to place contemporary observations of the marine system in a long-term perspective. One option is to consult historical records, replicate these studies and conduct comparative analyses of data sets spanning time scales long enough to encompass annual and decadal climate swings (see e.g. Falk-Petersen et al. (2008) for changes in the local climate at decadal and longer time scales).
Community structure and species distributions are dynamic and vary in response to a range of factors. Recent studies show that community changes and northward shifts of some marine species are linked to increasing water temperature (Beaugrand et al. 2002; Perry et al. 2005), but also disturbances such as intensive trawling can lead to changes in community structure (e.g. Worm and Myers 2003). Several benthic time series of up to 100 years have been analysed in the North Atlantic (Pearson et al. 1985; Bradshaw et al. 2002; Southward et al. 2005), but despite evidence that Arctic biological systems respond to climatic variability (Ambrose et al. 2006; Beuchel et al. 2006), no studies spanning more than two to three decades have to our knowledge been reported from the Arctic.
Decapod crustaceans are important trophic links to higher predators and some have commercial value. For example, the northern shrimp, Pandalus borealis, has been heavily fished in Svalbard waters since the 1970s. Most decapod species are sensitive to low temperatures, and only about 20% (ca 21 spp. versus ca 107 spp.) of the species found along the Norwegian coast are recorded along the coast of Svalbard (Brattegard and Holthe 1997; Gulliksen et al. 1999; T. Brattegard new data). As of 2008, 77 of the decapods species along the Norwegian coast are southern species with a northern border somewhere on the coast, while only 9 are northern species with a southern border somewhere on the Norwegian coast (Brattegard and Holthe 1997; Gulliksen et al. 1999). A northward expansion of at least 20 decapod species has also been observed over the last two decades, indicating an ecological response to a warming sea along the Norwegian coast (Brattegard and Holthe 1997; T. Brattegard new data), as such faunal changes are associated with altered hydrographic conditions, both locally (Beuchel et al. 2006; Willis et al. 2006, 2008) and regionally (reviewed in Drinkwater 2006; Renaud et al. 2008). Decapod species composition and distribution are thus well suited for studies of the effects caused by physical changes in the environment. In this study, we utilize current data in addition to 50 and 100-year-old data from the Isfjorden system to examine possible changes in the species composition and diversity of decapods during the last century.
In the Isfjorden complex, Svalbard, 110 stations were sampled both in 1908 and 1958 (von Hofsten 1916; Christiansen and Christiansen 1962). The investigations in 1958 were initiated to study the potential effects of a warming period in the North Atlantic and Arctic Oceans. No obvious trends were detected and reported in 1958 that could indicate changes at the species level, but community structure was not well addressed (Christiansen and Christiansen 1962). Changes in the functional role of a community, however, may be better understood by multivariate analysis of the species assemblage, especially if the target groups studied are likely to play important ecosystem roles.
Here, we present the results from a study based on a recent re-visit of a subset of the locations studied in 1908 and 1958. We studied whether the decapod community structure has changed since 1908 or 1958. We discuss our findings in the context of long-term records of change in the physical environment, specifically in terms of ocean temperatures.
Materials and methods
Study area
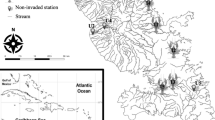
Isfjorden is the largest fjord on the west coast of Spitsbergen and forms a fjordic system comprising 4 side-fjords (Fig. 1). Despite the high-latitude location of Isfjorden, it is periodically influenced by significant influx of warmer Atlantic water from the West Spitsbergen Current (Berge et al. 2005; Nilsen et al. 2008), resulting in a hydrographic switch from typically Arctic to Atlantic conditions during both summer and winter (Cottier et al. 2007; Cottier et al. 2005). The degree to which the Atlantic water crosses the West Spitsbergen Shelf and enters the fjord is dependent on the strength of the density front between the water in the fjord and the Atlantic water. The formation of dense brine during winters of intense sea ice growth can block the inflow of Atlantic water, thus controlling the exchange between the fjord and the shelf (Nilsen et al. 2008). In effect, the fjord is dominated by winter-cooled water masses during the winter, with potential for large annual variations in the influx of warmer Atlantic water during summer.
Map of Svalbard and the study area Isfjorden. Station numbers correspond to those indicated in Table 1 for 2007
Sampling
The sampling in 2007 was carried out between 17 and 19 August during a cruise on RV “Jan Mayen”, at about the same time of the year as both the 1908 and 1958 campaigns. A total of 22 stations that were sampled in both 1908 and 1958 were selected and re-sampled in 2007 according to the descriptions given in Christiansen and Christiansen (1962). The 22 stations were selected so as to provide a reasonable cover of the full geographical and hydrographical range of the fjord complex (Fig. 1), and is thus a compromise between sampling effort and information yield. The sampling gear for all three sampling years consisted mainly of an Agassiz trawl and a triangular dredge, although in 2007 we used an epibenthic sledge and a Sneli sledge on stations 1 and 2. As the latter two gears were not used in 1908 and 1958, data analyses was carried out both with and without these two stations in order to control for biases in results due to the use of different sampling gears. These analyses did not yield conspicuously different results, and all further discussions are based on the full set of stations from 2007.
Samples were sorted and identified onboard the ship and all decapods were identified to the species level. Sampling locations, sampling gear and depth are presented in Table 1.
Analysis
Faunal communities were analysed by analysis of similarities (ANOSIM) of Bray–Curtis similarity matrices using the PRIMER software package (Clarke and Gorley 2006). As the sampling gear used in this study does not yield truly quantitative samples, the similarity matrices were constructed based on presence/absence, fourth-root transformed as well as raw count data in order to investigate the effect of both rare and dominant species. For the number of individuals per species and station for all 3 years, see Table 2 as well as von Hofsten (1916) and Christiansen and Christiansen (1962). ANOSIM was conducted to determine whether the community composition of decapods was different among the three sampling points spanning one century. To determine which taxa contributed to community differences among the stations over time, we also conducted correspondence analysis (CA) using the CANOCO software package (Ter Brak and Smilauer 2002). This analysis was performed on fourth-root transformed faunal data, with depth and year added as supplementary environmental data. Unfortunately, no other environmental data were available for all three sampling periods.
Results
Analyses of the species composition of decapods in Isfjorden in 2007 did not reveal any significant changes compared to the samples collected in 1958, but there was a significant difference between 1908 and 2007 (see Table 3 for statistics). As the statistics in Table 3 indicate, there are small but increasing differences from 1908 to 1958 and from 1908 to 2007, but the overall picture suggests that communities did not change very much (generally low R values despite significant P values). Furthermore, the results show that with one exception, no taxa have been lost or gained in the community, with nine species present throughout the century. The exception is Sabinea sarsi, which was recorded as a single individual in 1958 and eight individuals in 2007, but not observed in 1908 (see Table 2). Hence, differences between the three time periods are due to relative changes in abundances of the same set of species.
Changes in the community structure were apparent using a CA plot (Fig. 2). The first axis explained 32.3% of the variability among stations and was based on the presence of Pandalus borealis (positive direction) and Sclerocrangon boreas (negative direction). Stations were separated along the second axis (explaining nearly 19% of the variability) based on the relative abundances of Hyas araneus, Pagurus pubescens and Sabinea septemcarinata (positive) and Lebbeus polaris and the two Spirontocaris spp. (negative). While all years had stations scattered along the horizontal axis, there was a shift in community structure in the positive direction on the y-axis between 1908 and 2007. Stations from 1908 and 1958 scattered between both positive and negative positions along the y-axis, with a wider range in 1908 stations, while most 2007 stations plot above zero on the y-axis (Fig. 2). The supplementary variable depth was highly positively correlated with Axis 1 of the CA, while the year was highly correlated with Axis 2.
Ordination plot from the correspondence analysis (CA) using fourth-root transformed faunal data. Axis 1 explains 32.3% of the variance in faunal data and Axis 2 explains 18.3%. Filled triangles represent the species weighting in defining the plot corresponding to the following abbreviations: Hyas ar: Hyas arenarius (Linnaeus, 1758), Pag pub: Pagurus pubescens (Krøyer, 1838), Scl bor: Sclerocrangon borealis (Phipps, 1774), Sab sep: Sabinea septemcarinata (Sabine, 1824), Sab sar: Sabinea sarsi (Smith, 1879), Pan bor: Pandalus borealis (Krøyer, 1838), Eu gam: Eualus gaimardii (Milne Edwards, 1837), Spr ph: Spirontocaris phippsi (Krøyer, 1841), Spir sp: Spirontocaris spinus (Sowerby, 1805), Leb pol: Lebbeus polaris (Sabine, 1824). Open symbols indicate sampling year. Black circles: 1908, red squares: 1958, green stars: 2007. Grey lines indicate the passive (supplementary) environmental variables “depth” and “year” added to help interpret patterns generated by species relative abundances
Discussion
The similarity of the samples from 1908, 1958 and 2007 suggests that these Arctic decapods show a high degree of persistence in their distribution, as the species composition has, with one possible exception (see below), remained similar during the last century (Fig. 2; Table 3). This might reflect that decapods are well suited for studies of community resistance towards environmental changes, as the individual species seem very resistant to the physical (hydrographical) and anthropogenic changes that have taken place (see below). On the other hand, it might also reflect that Arctic ecosystems are more robust than what is generally assumed (but see e.g. Grebmeier et al. 2006 and Beuchel et al. 2006 for recent examples of Arctic communities responding more markedly to environmental or anthropogenic impacts). The community structure of infauna in van Mijenfjord, just south of Isfjord, showed little change over samplings separated by more than 20 years (Renaud et al. 2007); Kongsfjorden, north of Isfjorden, showed only very modest change in infaunal communities over a 10-year-period (M. Kedra, IOPAS, Sopot, Poland, personal communication). Additionally, a recently completed analysis of over 3,200 samples from the soft-sediment benthos along the European continental shelf has revealed that faunal diversity does not vary with latitude between 34 and 81°N (Renaud et al. 2009). The ability of benthic fauna to accommodate environmental variability may therefore be responsible for the stability in species composition observed over the past 100 years. These findings contrast studies of pelagic systems where clear latitudinal shifts in species distributions are observed, such as the northward shift of zooplankton species during the four last decades of the twentieth century (Beaugrand et al. 2002). Plankton species and communities will to a large degree follow changes in oceanographic conditions (but see Kaartvedt 2008). The pelagic larval stages of benthic organisms, however, are often short, movement in the benthic stage is usually limited, and benthic species are generally long-lived, compared with pelagic taxa, and less influenced by advection. Hence, there are reasons to expect that shifts in species distributions are slower in benthic compared to pelagic systems.
Recently, the temperature in the West Spitsbergen Current has increased (Schauer et al. 2004; Walczowski and Piechura 2006). This has contributed to the greater influence of Atlantic water in the fjords of West Spitsbergen (Cottier et al. 2007; Berge et al. 2005), and advective effects have been shown to have a direct impact on the pelagic communities in Svalbard fjords (Willis et al. 2008; Willis et al. 2006). Historically, the temperature of the Atlantic water in the Arctic has been shown to undergo a multi-decadal oscillation with negative anomalies during 1900–1920 and positive anomalies during 1920–1950 and from mid-1980s onwards (Polyakov et al. 2005). The transport of the Atlantic water through the Nordic Seas is sufficiently coherent along its path (Holliday et al. 2008) that we can infer an enhanced warming of the West Spitsbergen Current in the late 1950s from hydrographic time series in the North Atlantic. Oceanographic conditions in the 1908 period are more difficult to determine, but from the negative temperature anomaly (Polyakov et al. 2005) and records of sea ice extending further south than in the late 1950s or post-2000 (Falk-Petersen et al. 2008), one might infer that there was less influence of Atlantic Water and Isfjorden had a more Arctic character.
Our results from Isfjorden do, however, indicate a shift in the community structure between 1908 and the two later sampling periods in 1958 and 2007. The transition detected in community structure and supported by the positive correlation of year with Axis 2 (Fig. 2) seems to reflect the 1908 community being characterized by selective predators, such as Lebbeus polaris and Spirontocaris spinus (Birkely and Gulliksen 2003), while in 1958 and 2007 generalists play a larger role in the communities (Fig. 2). The generalist decapods include Hyas araneus, Pagurus pubescens and Sabinea septemcarinata species found in almost all types of benthic habitats (Christiansen 1972; J. Berge personal observation). Both H. araneus and P. pubescens have been shown to be scavengers (Guijarro Garcia et al. 2006; Aschan 1991) and are recorded in very high abundances in areas heavily disturbed by trawling and other fishing activities (Guijarro Garcia et al. 2006). In the Isfjorden area, however, commercial fishing activity did not commence until the late 1960s or early 1970s and therefore cannot explain the shift in community structure seen between 1908 and 1958. The knowledge about feeding ecology of the above-mentioned species from Isfjorden (Birkely and Gulliksen 2003) were based on examination of stomach contents of relatively few specimens, and the definition of these species as “specialists” might thus not be as clear as presented by Birkely and Gulliksen (2003). However, the morphology of these species (especially their very small claws on pereiopods 1 and 2) separates them from both the crangonid shrimps and the other “generalists” species, thus supporting the main distinction between the two functional groups. Based on the above, it seems logical to conclude that anthropogenic disturbance such as trawling has not been the primary cause for the observed shift in species composition; however, bottom-fishing disturbance may act to reduce the primary dietary items for specialists and hence reinforce an on-going change within the community. Bottom trawling is known to reduce abundances of erect habitat-forming species, such as hydroids (e.g. Watling and Norse 1999; Thrush and Dayton 2002). Both L. polaris and S. spinus in Isfjorden consume large amounts of hydroids, with L. polaris also consuming significant numbers of amphipods (Birkely and Gulliksen 2003).
The potential significance of a shift towards more scavengers and generalists remains unclear, but may relate to oscillations between “warm” and “cool” periods in the fjord. The North Atlantic experiences “warm” and “cool” periods lasting several decades each, and affecting both pelagic and benthic communities (reviewed in Drinkwater 2006; Renaud et al. 2008). In this study, sampling in 1908 was during a cooler period, sampling in 1958 was near the end of a two-decade long warm period, and sampling in 2007 occurred within the current warm period. This corresponds to the results showing a community structure in 1908 distinct from that in 1958 or 2007. Warm periods are characterized by a greater influx of warm Atlantic water masses into west Svalbard fjords (Cottier et al. 2007; Cottier et al. 2005) leading to the potential for higher annual and interannual variability in the hydrography. If a relatively stable environment becomes more disturbed, we may expect generalists to become more common and specialists less likely to persist (Begon et al. 1996, p897). Oceanographic variability as a source of disturbance might, therefore, lead to the observed shift from specialists towards generalists. The mechanism may be direct, through physical water mass properties such as temperature and salinity, or indirect, such as through associated changes in the pelagic community or effects on prey distributions. However, we have little direct information on tolerances of these taxa to such environmental variability.
CA results indicate that a large amount of the variability among stations was due to the relative abundance of Pandalus borealis compared to that of Sclerocrangon boreas (Fig. 2 first axis). This coincides with the general depth distribution of these taxa, with deeper stations characterized by Pandalus along the central axis of Isfjorden generally located to the right in the CA plot, regardless of the year sampled. This is supported by the position of the vector for depth in Fig. 2. It is not unexpected that the decapod communities from 22 stations spanning such a wide depth range (12–408 m) would in part separate according to this factor. Decapods, like most other invertebrates, are likely to have different depth preferences and our study identifies this. Further, these results suggest that we sampled (in 2007) a range of stations comparable to those sampled previously in terms of the factors (CA axis 1) responsible for a large part of the variability among sampling locations.
The recorded decapod fauna of Isfjorden include 13 species, 10 of which have been reported and identified in the studies discussed here. The one species that, based on the data treated herein, seems to have established itself in Isfjorden since 1908 is Sabinea sarsi, a species considered as an indicator species for warm Atlantic water (Blacker 1957; Birkely and Gulliksen 2003). A single individual of this species was recorded in 1958, but as it was found at a station not resampled in 2007, it appears in the analyses as “new” in the preset study. During the 2007 campaign, a total of eight individuals were collected at three of the stations. The three species not identified from any of the stations treated herein are Hyas coarctatus, Sclerocrangon ferox and Lithodes maja. The latter two species have only been recorded once in the area, whereas the former have been recorded several times (Gulliksen et al. 1999). Lithodes maja has since been reported at least once from areas close to Isfjorden (J. Sundet, Inst. Marine Research, Tromsø, Norway, personal communication), but we are unaware of any reports of this species from Isfjorden since 1962. Concerning Hyas coarctatus, it is usually separated from Hyas araneus by the size of the notch behind the eye (small/lacking versus distinct; see commonly used identification key from these waters: Christiansen 1972). Having observed a large variety in the size of this notch, we believe that this character is quite ambiguous in species identification and the records of this species may not be reliable.
All ten decapod species recorded in Isfjorden in this study have also been recorded along the Norwegian mainland coast, which supports the view that the benthic fauna of Svalbard is an impoverished fauna of the Norwegian coast (Gulliksen and Svendsen 2004). Four of the ten species have a northern distribution (southern border along the Norwegian coast), while the remaining six (Table 3) are found along the entire coast of Norway (Brattegard and Holthe 1997). Two of the three taxa that have increased in importance in Isfjorden (H. araneus and P. pubescens) are from this latter widespread group. The third species, S. septemcarinata, is a member of the northern group, but is not found in many of the colder areas of Svalbard (Gulliksen et al. 1999) and has a continuous distribution at least as far south as 61°N (Brattegard and Holthe 1997).
The distributional pattern of decapods on Svalbard does appear to be consistent with our observations of faunal changes being related to regional warming in 1958 and 2007 compared to 1908. Despite limited temporal resolution, our analyses provide at least two valuable insights: (1) that the decapod species composition of Arctic fjords may be relatively persistent and resistant to changes; and (2) that the subtle, but potentially ecologically significant, changes in community structure that have taken place during this 100-year-period suggests a potential relationship to increased disturbance, caused by higher oceanographic variability or by fisheries. If warm periods, such as that we are currently experiencing, are characterized by a greater role of generalist consumers, then ecosystem function may be considerably different from that under conditions favoring specialist predators. More information on the generality of these findings, and the potential mechanisms behind them, would be valuable in predicting how the ecosystem will react to a warmer Arctic.
References
Ambrose WG, Carroll ML, Greenacre M, Thorrold SR, McMahon KW (2006) Variation in Serripes groenlandicus (Bivalvia) growth in a Norwegian high-Arctic fjord: evidence for local- and large-scale climatic forcing. Glob Change Biol 12(9):1595–1607. doi:10.1111/j.1365-2486.2006.01181.x
Aschan MM (1991) Effects of Icelandic scallop dredging on benthic communities in the Northeast Atlantic. ICES Benthos Ecology Working Group, special international workshop on the effects of physical disturbance of the seafloor on benthic and epibenthic ecosystems. Bedford Institute of Oceanography, p 10
Beaugrand G, Reid PC, Ibanez F, Liondley JA, Edwards M (2002) Reorganization of North Atlantic marine copepod biodiversity and climate. Science 296:1692–1694. doi:10.1126/science.1071329
Begon M, Harper JL, Townsend CR (1996) Ecology, 3rd edn. Blackwell Science, Oxford
Berge J, Johnsen G, Nilsen F, Gulliksen B, Slagstad D (2005) Ocean temperature oscillations enable reappearance of blue mussels Mytilus edulis in Svalbard after a 1,000-year absence. Mar Ecol Prog Ser 303:167–175. doi:10.3354/meps303167
Beuchel F, Gulliksen B, Carroll ML (2006) Long-term patterns of rocky bottom macrobenthic community structure in an Arctic fjord (Kongsfjorden, Svalbard) in relation to climate variability (1980–2003). J Mar Syst 63:35–48. doi:10.1016/j.jmarsys.2006.05.002
Birkely S-R, Gulliksen B (2003) Feeding ecology in five shrimp species (Decapoda, Caridea) from an Arctic fjord (Isfjorden, Svalbard), with emphasis on Sclerocrangon boreas (Phipps, 1774). Crustaceana 76(6):699–715. doi:10.1163/156854003322381513
Blacker RW (1957) Benthic animals as indicators of hydrographic conditions and climatic change in Svalbard waters. Fish Invest 2:1–49
Bradshaw C, Veale LO, Brand AR (2002) The role of scallopdredge disturbance in long-term change in Irish Sea benthic communities: a re-analysis of an historical data set. J Sea Res 47:161–184. doi:10.1016/S1385-1101(02)00096-5
Brattegard T, Holthe T (1997) Distribution of marine, benthic macro-invertebrates in Norway. Research Report for DN 1997-1. Directorate for Nature Management, p 409
Christiansen ME (1972) Crustacea Decapoda, tifotkreps. Universitetsforlaget, Oslo, pp 1–71
Christiansen ME, Christiansen BO (1962) The Crustacea Decapoda of Isfjorden: a comparison with the Swedish Spitsbergen Expedition in 1908. Acta Scientia 19:1–52
Clarke KR, Gorley RN (2006) Primer v6: user manual/tutorial. Primer-E Ltd, Plymouth
Cottier FR, Tverberg V, Inall M, Svendsen H, Nilsen F, Griffiths C (2005) Water mass modification in an Arctic fjord through cross-shelf exchange: The seasonal hydrography of Kongsfjorden, Svalbard. J Geophysl Res-Oceans 110. doi:10.1029/2004JC002757
Cottier FR, Nilsen F, Inall ME, Gerland S, Tverberg V, Svendsen H (2007) Wintertime warming of an Arctic shelf in response to large-scale atmospheric circulation. Geophys Res Lett 34. doi:10.1029/2007GL029948
Divine DV, Dick C (2006) Historical variability of sea ice edge position in the Nordic Seas. J Geophys Res Oceans 111. doi:10.1029/2004JC002851
Drinkwater KF (2006) The regime shifts of the 1920s and 1930s in the North Atlantic. Prog Oceanogr 68:134–151
Falk-Petersen S, Leu E, Berge J, Kwasniewski S, Nygård H, Røstad A, Keskinen E, Thormar J, von Quillfeldt C, Wold A, Gulliksen B (2008) Calanus hyperboreus feeding on an Arctic autumn bloom at 82°N induced by the northernmost location of the ice edge since 1751. Deep Sea Res Part II Top Stud Oceanogr 55:2275–2284. doi:10.1016/j.dsr2.2008.05.010
Grebmeier JM, Overland JE, Moore SE, Farley EV, Carmack EC, Cooper LW, Frey KE, Helle JH, McLaughlin FA, McNutt SL (2006) A major ecosystem shift in the northern Bering Sea. Science 311:1461–1464. doi:10.1126/science.1121365
Guijarro Garcia E, Ragnarsson SA, Eiríksson H (2006) Effects of scallop dredging on macrobenthic communities in west Iceland. ICES J Mar Sci 63:434–443. doi:10.1016/j.icesjms.2005.08.013
Gulliksen B, Svendsen E (2004) Svalbard and life in polar oceans. Kom forlag, Kristiansund
Gulliksen B, Palerud R, Brattegard T, Sneli J (1999) Distribution of marine benthic macro-organisms at Svalbard (including Bear Island) and Jan Mayen. Research Report for DN 1999-4, Directorate for Nature Management, p 148
Holliday NP, Hughes S, Bacon S, Beszczynska-Moller A, Hansen B, Lavin A, Loeng H, Mork KA, Østerhus S, Sherwin TJ, Walczowski W (2008) Reversal of the 1960s to 1990s freshening trend in the northeast North Atlantic and Nordic Seas. Geophys Res Lett 35. doi:10.1029/2007GL032675
Johanessen O, Miles LB, Kuzmina M, Semenov S, Aledseev V, Nagumyi G, Zakharov A, Bobylev V, Pettersson L, Hasselmann L, Cattle K (2004) Arctic climate change: observed and modelled temperature and sea-ice variability. Tellus 56A:328–341
Kaartvedt S (2008) Photoperiod may constrain the effect of global warming in arctic marine systems. J Plankton Res 30:1203–1206. doi:10.1093/plankt/fbn075
Nilsen F, Cottier F, Skogseth R, Mattson S (2008) Fjord–shelf exchanges controlled by ice and brine production: the interannual variation of Atlantic Water in Isfjorden, Svalbard. Cont Shelf Res 28:1838–1853. doi:10.1016/j.csr.2008.04.015
Pearson TH, Josefson AB, Rosenberg R (1985) Peterson’s benthic stations revisited I Is the Kattegatt becoming eutrophic? J Exp Mar Biol Ecol 92:157–206. doi:10.1016/0022-0981(85)90094-2
Perry AL, Low PJ, Ellis JR, Reynold JD (2005) Climate change and distribution shifts in marine fishes. Science 308:1912–1915. doi:10.1126/science.1111322
Polyakov IV, Johnson MA (2000) Arctic decadal and interdecadal variability. Geophys Res Lett 27:4097–4100. doi:10.1029/2000GL011909
Polyakov IV, Alekseev G, Timokhov L, Bhatt U, Colony R, Simmons H, Walsh D, Walsh J, Zakharov V (2004) Variability of the intermediate Atlantic Water of the Arctic ocean over the last 100 years. J Clim 17:4485–4497. doi:10.1175/JCLI-3224.1
Polyakov IV, Beszczynska A, Carmack EC, Dmitrenko IA, Fahrbach E, Frolov I.E, Gerdes R, Hansen E, Holfort J, Ivanov VV, Johnson MA, Karcher M, Kauker F, Morison J, Orvik KA, Schauer U, Simmons HL, Skagseth Ø, Sokolov VT, Steele M, Timokhov LA, Walsh D, Walsh JE (2005) One more step toward a warmer Arctic. Geophys Res Lett 32. doi:10.1029/2005GL023740
Renaud PE, Włodarska-Kowalczuk M, Trannum H, Holte B, Wezsławski JM, Cochrane S, Dahle S, Gulliksen B (2007) Multidecadal stability of benthic community structure in a high-Arctic glacial fjord (van Mijenfjord, Spitsbergen). Polar Biol 30:295–305. doi:10.1007/s00300-006-0183-9
Renaud PE, Carroll ML, Ambrose WG Jr (2008) Effects of global warming on Arctic sea-floor communities and its consequences for higher trophic levels. In: Duarte C (ed) Impacts of global warming on polar ecosystems. FBBVA Press, Bilbao, pp 139–175
Renaud PE, Webb TJ, Bjørgesæter A, Karakassis I, Kędra M, Kendall MA, Labrune C, Lampadariou N, Somerfield PJ, Włodarska-Kowalczuk M, Berghe EV, Claus S, Aleffi F, Amouroux J-M, Bryne KH, Cochrane SJ, Dahle S, Degraer S, Denisenko SG, Deprez T, Dounas C, Fleischer D, Gil J, Grémare A, Janas U, Mackie ASY, Palerud R, Rumohr H, Sardá R, Speybroeck J, Taboada S, Van Hoey G, Węsławski JM, Whomersley P, Zettler ML (2009) Continental-scale patterns in benthic invertebrate diversity: insights from the MarBEF database. Mar Ecol Prog Ser
Schauer U, Fahrbach E, Østerhus S, Rohardt G (2004) Arctic warming through the Fram Strait: oceanic heat transport from 3 years of measurements. J Geophys Res-Oceans 109:C06026. doi:10.1029/2003JC001823
Ter Braak CJF Smilauer (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
Southward AJ, Langmead O, Hardman-Mountford NJ, Aiken J, Boalch GT, Dando PR, Genner MJ, Joint I, Kendall MA, Halliday NC, Harris RP, Leaper R, Mieszkowska N, Pingree RD, Richardson AJ, Sims DK, Smith T, Walne AW, Hawkins SJ (2005) Long-term oceanographic and ecological research in the western English Channel. Adv Mar Biol 47:1–105. doi:10.1016/S0065-2881(04)47001-1
Thrush SF, Dayton PK (2002) Disturbance to marine benthic habitats by trawling and dredging: implications for marine biodiversity. Annu Rev Ecol Syst 33:449–473. doi:10.1146/annurev.ecolsys.33.010802.150515
von Hofsten N (1916) Die Decapoden Crustaceen des Eisfjords. Kunglige Svenska Vetenskaps-Akademiens Handlingar 54(7):108
Walczowski W, Piechura J (2006) New evidence of warming propagating toward the Arctic Ocean. Geophys Res Lett 33. doi:10.1029/2006GL025872
Watling L, Norse EA (1999) Disturbance of the seabed by motile fishing gear: a comparison with forest clear-cutting. Conserv Biol 12:180–197
Willis KJ, Cottier FR, Kwasniewski S, Wold A, Falk-Petersen S (2006) The influence of advection on zooplankton community composition in an Arctic fjord (Kongsfjorden, Svalbard). J Mar Syst 61:39–54. doi:10.1016/j.jmarsys.2005.11.013
Willis KJ, Cottier FR, Kwasniewski S (2008) Impact of warm water advection on the winter zooplankton community in an Arctic fjord. Polar Biol 31:475–481. doi:10.1007/s00300-007-0373-0
Worm B, Myers RA (2003) Meta-analysis of cod-shrimp interactions reveals top–down control in oceanic food webs. Ecology 84:162–173. doi:10.1890/0012-9658(2003)084[0162:MAOCSI]2.0.CO;2
Acknowledgments
We are grateful to the captain and crew of F/F “Jan Mayen” and all students on AB321 for their help during this study, and to Colin Griffiths for his help producing the map. The study was co-financed by Conoco-Philips. The manuscript was improved by comments provided by F Beuchel, JM Weslawski and one anonymous reviewer.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Berge, J., Renaud, P.E., Eiane, K. et al. Changes in the decapod fauna of an Arctic fjord during the last 100 years (1908–2007). Polar Biol 32, 953–961 (2009). https://doi.org/10.1007/s00300-009-0594-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-009-0594-5