Abstract
Key message
Metabolomic profiling of a maize line engineered with an endosperm-specific carotenogenic pathway revealed unexpected metabolic readjustments of primary metabolism in leaves and roots.
Abstract
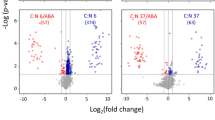
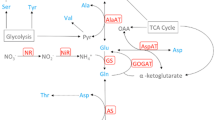
High-carotenoid (HC) maize was engineered to accumulate high levels of carotenoids in the endosperm. The metabolic interventions influenced the flux through non-target pathways in tissues that were not affected by the targeted intervention. HC maize at the vegetative stage also showed a reduced susceptibility to insect feeding. It is unknown, however, whether the metabolic history of the embryo has any impact on the metabolite composition in vegetative tissues. We, therefore, compared HC maize and its isogenic counterpart (M37W) to test the hypothesis that boosting the carotenoid content in the endosperm triggers compensatory effects in core metabolism in vegetative tissues. Specifically, we investigated whether the metabolite composition of leaves and roots at the V6 stage differs between HC and M37W, and whether N inputs further alter the core metabolism of HC compared to M37W. We found an increase in the abundance of organic acids from the tricarboxylic acid (TCA) cycle in HC even under restricted N conditions. In contrast, low levels of carotenoids and chlorophyll were measured regardless of N levels. Sugars were also significantly depleted in HC under low N. We propose a model explaining the observed genotype-dependent and input-dependent effects, in which organic acids derived from the TCA cycle accumulate during vegetative growth and contribute to the increased demand for pyruvate and/or acetyl-CoA in the endosperm and embryo. This response may in part reflect the transgenerational priming of vegetative tissues in the embryo induced by the increased demand for metabolic precursors during seed development in the previous generation.





Similar content being viewed by others
Availability of data and materials
All relevant data are within the paper and its Supplementary material file.
Code availability
Not applicable.
References
Amiour N, Imbaud S, Clement G, Agier N, Zivy M, Valot B, Balliau T, Armengaud P, Quillere I, Canas R, Tercet-Laforgue TT, Hirel B (2012) The use of metabolomics integrated with transcriptomic and proteomic studies for identifying key steps involved in the control of nitrogen metabolism in crops such as maize. J Exp Bot 63:5017–5033
Araújo WL, Tohge T, Ishizaki K, Leaver CJ, Fernie AR (2011) Protein degradation—an alternative respiratory substrate for stressed plants. Trends in Plant Sci 16:489–498
Babu R, Rojas NP, Gao SB, Yan JB, Pixley K (2013) Validation of the effects of molecular marker polymorphisms in LcyE and CrtRB1 on provitamin A concentrations for 26 tropical maize populations. Theor Appl Genet 126:389–399
Bai C, Twyman RM, Farré G, Sanahuja G, Christou P, Capell T, Zhu CF (2011) A golden era-pro-vitamin A enhancement in diverse crops. Vitro Cell Dev Biol Plant 47:205–221
Barros E, Lezer S, Anttonen MJ, van Dijk JP, Röhlig RM, Kok EJ, Engel KH (2010) Comparison of two GM maize varieties with near-isogenic non-GM variety using transcriptomics, proteomics, and metabolomics. Plant Biotechnol J. 8:436–451
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Bruce TJA, Matthes MC, Napier JA, Pickett JA (2007) Stressful “memories” of plants: evidence and possible mechanisms. Plant Sci 173:603–608
Chen K, Arora R (2013) Priming memory invokes seed stress-tolerance. Environ Exp Bot 94:33–45
Cocuron JC, Koubaa M, Kimmelfield R, Ross Z, Alonso AP (2019) A combined metabolomics and fluxomics analysis identifies steps limiting oil synthesis in maize embryos. Plant Physiol 181:961–975
Decourcelle M, Perez-Fons L, Baulande S, Steiger S, Couvelard L, Hem S, Zhu CF, Capell T, Christou P, Fraser P, Sandmann G (2015) Combined transcript, proteome, and metabolite analysis of transgenic maize seeds engineered for enhanced carotenoid synthesis reveals pleotropic effects in core metabolism. J Exp Bot 66:3141–3150
Detarsio E, Maurino VG, Alvarez CE, Müller GL, Andreo CS, Drincovich MF (2008) Maize cytosolic NADP-malic enzyme (ZmCytNADP-ME): a phylogenetically distant isoform specifically expressed in embryo and emerging roots. Plant Mol Biol 68:355–367
Farré G, Ramessar K, Twyman RM, Capell T, Christou P (2010a) The humanitarian impact of plant biotechnology: recent breakthroughs vs bottlenecks for adoption. Curr Opin Plant Biol 13:219–225
Farré G, Sanahuja G, Naqvi S, Bai C, Capell T, Zhu C, Christou P (2010b) Travel advice on the road to carotenoids in plants. Plant Sci 179:28–48
Fraser PD, Pinto MES, Holloway DE, Bramley PM (2000) Application of high-performance liquid chromatography with photodiode array detection to the metabolic profiling of plant isoprenoids. Plant J 24:551–558
Fritz C, Mueller C, Matt P, Feil R, Stitt M (2006) Impact of the C-N status on the amino acid profile in tobacco source leaves. Plant Cell Environ 29:2055–2076
Galloway LF, Etterson JR (2007) Transgenerational plasticity is adaptive in the wild. Science 318:1134–1136
Gamir J, Sánchez-Bel P, Flors V (2014) Molecular and physiological stages of priming: how plants prepare for environmental challenges. Plant Cell Rep 33:1935–1949
Harjes CE, Rocheford TR, Bai L, Brutnell TP, Kandianis CB, Sowinski SG, Stapleton AE, Vallabhaneni R, Williams M, Wurtzel ET et al (2008) Natural genetic variation in lycopene epsilon cyclase tapped for maize biofortification. Science 319:330–333
Heyneke E, Watanabe M, Erban A, Duan G, Buchner D, Kopka J, Hawkesford MJ, Hoefgen R (2017) Characterization of the wheat leaf metabolome during grain filling and under varied n-supply. Front Plant Sci 8:2048
Hildebrandt TM, Nunes-Nesi A, Araújo WL, Braun HP (2015) Amino acid catabolism in plants. Mol Plant 8:1563–1579
Hilker M, Schwachtje J, Baier M, Balazadeh S, Bäurle I, Geiselhardt S, Hincha DK, Kunze R, Mueller-Roeber B, Rillig MC et al (2016) Priming and memory of stress responses in organisms lacking a nervous system. Biol Rev 91:1118–1133
Hirel B, Andrieu B, Valadiera MH, Sylvain Renarda S, Quilleré I, Chelleb M, Pommel B, Fournier C, Drouet JL (2005a) Physiology of maize II: identification of physiological markers representative of the nitrogen status of maize (Zea mays) leaves during grain filling. Physiol Plant 124:178–188
Hirel B, Martina A, Thérèse Tercé-Laforgue T, Gonzalez-Moro MB, Estavillo JM (2005b) Physiology of maize I: a comprehensive and integrated view of nitrogen metabolism in a C4 plant. Physiol Plant 124:166–177
Holeski LM, Jander G, Agrawal AA (2012) Transgenerational defense induction and epigenetic inheritance in plants. Trends Ecol Evol 27:618–626
Jablonka E (2013) Epigenetic inheritance and plasticity: the responsive germline. Prog Biophys Mol Biol 111:99–107
Jiang Y, Ling L, Zhnag L, Wang K, Li X, Cai M, Zhan M, Li C, Wang J, Cao C (2018) Comparison of transgenic Bt rice and their non-Bt counterpart in yield and physiological response to drought stress. Field Crops Res 217:45–52
Kinoshita T, Seki M (2014) Epigenetic memory for stress response and adaptation in plants. Plant Cell Physiol 55:1859–1863
Krapp A, Berthomé R, Orsel M, Mercey-Boutet S, Yu A, Castaings L, Elftieh S, Major H, Renou J-P, Daniel-Vedele F (2011) Arabidopsis roots and shoots show distinct temporal adaptation patterns toward nitrogen starvation. Plant Physiol 157:1255–1282
Nogueira M, Mora L, Enfissi EMA, Bramley PM, Fraser PD (2013) Subchromoplast sequestration of carotenoids affects regulatory mechanisms in tomato lines expressing different carotenoid gene combinations. Plant Cell 25:4560–4579
Paine JA, Shipton CA, Chaggar S, Howells RM, Kennedy MJ, Vernon G, Wright SY, Hinchliffe E, Adams JL (2005) Improving the nutritional value of Golden Rice through increased pro-vitamin A content. Nature Biotechnol 23:482–548
Palmer L, Dias D, Boughton B, Roessner U, Graham R, Stangoulis J (2014) Metabolite profiling of wheat (Triticum aestivum L.) phloem exudate. Plant Methods 10:1–9
Pastor V, Balmer A, Gamir J, Flors V, Mauch-Mani B (2014) Preparing to fight back: generation and storage of priming compounds. Front Plant Sci 5:1–12
Paszkowski J, Grossniklaus U (2011) Selected aspects of transgenerational epigenetic inheritance and resetting in plants. Curr Opin Plant Biol 14:195–203
Perez-Fons L, Bramley PM, Fraser PD (2014) The optimization and application of a metabolite profiling procedure for the metabolic phenotyping of Bacillus species. Metabolomics 10:77–90
Rao AV, Rao LG (2007) Carotenoids and human health. Pharmacol Res 55(3):207–216
Sahu PP, Pandey G, Sharma N, Puranik S, Muthamilarasan M, Prasad M (2013) Epigenetic mechanisms of plant stress responses and adaptation. Plant Cell Rep 32:1151–1159
Sandmann G (2001) Genetic manipulation of carotenoid biosynthesis: strategies, problems and achievements. Trends in Plant Sci 6:14–17
Schlüter U, Colmsee C, Scholz U, Bräutigam A, Weber APM, Zellerhoff N, Bucher M, Fahnenstich H, Sonnewald U (2013) Adaptation of maize source leaf metabolism to stress related disturbances in carbon, nitrogen, and phosphorus balance. BMC Genomics 14:442
Schlüter U, Mascher M, Colmsee C, Scholz U, Bräutigam A, Fahnenstich H, Sonnewald U (2012) Maize source leaf adaptation to nitrogen deficiency affects not only nitrogen and carbon metabolism but also control of phosphate homeostasis. Plant Physiol 160:1384–1406
Schwachtje J, Whitcomb SJ, Firmino AAP, Zuther E, Hincha DK, Kopka J (2019) Induced, imprinted, and primed responses to changing environments: does metabolism store and process information? Front Plant Sci 10:106
Slaughter A, Daniel X, Flors V, Luna E, Hohn B, Mauch-Mani B (2012) Descendants of primed Arabidopsis plants exhibit resistance to biotic stress. Plant Physiol 158:835–843
Smith RG, Gauthier DA, Dennis DT, Turpin DH (1992) Malate- and pyruvate-dependent fatty acid synthesis in leucoplasts from developing castor endosperm. Plant Physiol 98:1233–1238
Suwarno WB, Pixley KV, Palacios-Rojas N, Kaeppler SM, Babu R (2015) Genome-wide association analysis reveals new targets for carotenoid biofortification in maize. Theor Appl Genet 128:851–864
Turgut-Kara N, Arikan B, Celik H (2020) Epigenetic memory and priming in plants. Genetica 148:47–54. https://doi.org/10.1007/s10709-020-00093-4
Weinhold A (2018) Transgenerational stress-adaption: an opportunity for ecological epigenetics. Plant Cell Rep 37:3–9
Yan JB, Kandianis CB, Harjes CE, Bai L, Kim EH, Yang XH, Skinner DJ, Fu ZY, Mitchell S, Li Q et al (2010) Rare genetic variation at Zea mays crtRB1 increases beta-carotene in maize grain. Nat Genet 42:322-U374
Ye X, Al-Babili S, Klöti A, Zhang J, Lucca P, Beyer P, Potrykus I (2000) Engineering the provitamin A (β-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Science 287:303–305
Yesbergenova-Cuny Z, Dinant S, Martin-Magniette ML, Quilleré I, Armengaud P, Monfalet P, Lea PJ, Hirel B (2016) Genetic variability of the phloem sap metabolite content of maize (Zea mays L.) during the kernel-filling period. Plant Sci 252:347–357
Zakhartsev M, Medvedeva I, Orlov Y, Akberdin I, Rebs O, Schulze XW (2016) Metabolic model of central carbon and energy metabolisms of growing Arabidopsis thaliana in relation to sucrose translocation. BMC Plant Biol 16:262
Zalucki MP, Clarke AR, Malcolm SB (2002) Ecology and behavior of first instar larval Lepidoptera. Annu Rev Entomol 47:361–393. https://doi.org/10.1146/annurev.ento.47.091201.145220
Zanga D, Sanahuja G, Eizaguirre M et al (2018) Carotenoids moderate the effectiveness of a Bt gene against the European corn borer, Ostrinia nubilalis. PLoS ONE 13:1–9. https://doi.org/10.1371/journal.pone.0199317
Zanga D, Capell T, Slafer GA, Christou P, Savin R (2016) A carotenogenic mini-pathway introduced into white corn does not affect development or agronomic performance. Sci Rep 6:38288
Zhang Y, Lin X, Zhang Y, Zheng SJ, Du S (2005) Effects of nitrogen levels and nitrate/ammonium ratios on oxalate concentrations of different forms in edible parts of spinach. J Plant Nutr 28:2011–2025
Zheng X, Chen L, Xia H, Wei H, Lou Q, Li M, Li T, Luo L (2017) Transgenerational epimutations induced by multi-generation drought imposition mediate rice plant’s adaptation to drought condition. Sci Rep 7:1–13
Zhu C, Naqvi S, Breitenbach J, Sandmann G, Christou P, Capell T (2008) Combinatorial genetic transformation generates a library of metabolic phenotypes for the carotenoid pathway in maize. Proc Natl Acad Sci USA 105:18232–21823
Zhu CF, Sanahuja G, Yuan DW, Farré G, Arjo G, Berman J, Zorrilla-Lopez U, Banakar R, Bai C, Perez-Massot E et al (2013) Biofortification of plants with altered antioxidant content and composition: genetic engineering strategies. Plant Biotechnol J 11:129–141
Acknowledgements
We thank Dr. Roxana Savin for helping with the design of nitrogen treatments, as well as Jose Perez, Derry Alvarez, Pedro Cerdá and Dr. Teresa Capell for helping with the preparation of soil substrates. Special thanks to Jaume Capell for all the assistance provided in the greenhouse. P.C. acknowledges support from the ERA-NET SUSCROP PROSTRIG project funded by the Spanish Ministry of Science, Innovation and Universities (project number PCI2019-103382). P.D.F. and L.P.-F. are grateful to DEFRA for funding the PROSTRIG project through the ERA-NET SUSCROP scheme (project number CH0217). P.S.G.-C. was supported through an Agrotecnio postdoctoral fellowship.
Funding
P.C. acknowledges support from the ERA-NET SUSCROP PROSTRIG project funded by the Spanish Ministry of Science, Innovation and Universities (Project Number PCI2019-103382). P.D.F. and L.P.-F. are grateful to DEFRA for funding the PROSTRIG project through the ERA-NET SUSCROP scheme (project number CH0217). P.S.G.-C. was supported through an Agrotecnio postdoctoral fellowship.
Author information
Authors and Affiliations
Contributions
PC conceived the original idea; PSG-C designed and performed the experiments in the greenhouse; PSG-C prepared samples for GCMS and UPLC analysis; LP-F conducted the GCMS and UPLC analysis. PSG-C analyzed the data and wrote the article; LP-F supervised the data analysis; PDF and GS assisted in data interpretation; PC supervised the writing and edited the article. PDF and PC agree to serve as the authors responsible for contact and ensure communication.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Ethics approval
Not applicable.
Additional information
Communicated by Stefan Schillberg.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
299_2021_2689_MOESM1_ESM.docx
Supplementary file 1 Online Resource 1. Metabolite profile and changes in metabolite concentrations in M37W and HC leaves under low-nitrogen (N−) and normal (N +) input regimens. Online Resource 2. Metabolite profile and changes in metabolite concentrations in M37W and HC roots under low-nitrogen (N−) and normal (N +) input regimens. Online Resource 3. Plant material and N treatments used in this study. (DOCX 959 KB).
299_2021_2689_MOESM2_ESM.xlsx
Supplementary file 2 Online Resource 4. Data sets generated during and/or analyzed during the current study. (XLSX 36 KB).
Rights and permissions
About this article
Cite this article
Girón-Calva, P.S., Pérez-Fons, L., Sandmann, G. et al. Nitrogen inputs influence vegetative metabolism in maize engineered with a seed-specific carotenoid pathway. Plant Cell Rep 40, 899–911 (2021). https://doi.org/10.1007/s00299-021-02689-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-021-02689-2