Abstract
The SARS-CoV-2 virus is known to mediate attack via ACE-2 Receptor, thus having adverse effects on cardiovascular, respiratory, digestive and reproductive systems, the latter being an area of emerging concern, due to the associated impact on fertility, with potential for an outsized effect on population distribution and socioeconomic road map in subsequent years. This narrative review aims to put forth the current evidence of effect of SARS-CoV-2 on human fertility from a multipronged immunologic, haematologic, and gynaecologic perspective; highlighting the areas of contradiction and potential future measures. A literature search was conducted through the MEDLINE and SCOPUS databases to identify articles on the subject in English. Relevant information was extracted from around 300 articles for this review. The existing data give non-conclusive evidence about the impact of SARS-CoV-2 infection on fertility; however, a greater impact on male fertility as compared to females merits further exploration. However, reproduction and fertility is a key concern and considering the pandemic is prolonged, natural conception or ART require extra precautions.
Similar content being viewed by others
Introduction
The COVID-19 pandemic has led to a remarkable upheaval in global health and economy in a span of 18 months. While steady research on the subject has thrown light on the pathogenic mechanisms of the disease, a better understanding has also paved the way to newer concerns. It is now well-accepted that human cells that express angiotensin converting enzyme-2 (ACE-2) and transmembrane serine protease 2 (TMPRSS2) receptors act as portal of entry for the virus into the human body and immune system. The viral attack spans various systems, ranging from cardiovascular, respiratory, and digestive, to urinary and gonadal organs. The latter has led to emerging concerns regarding potential effect of SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2 on human fertility [1, 2]). COVID-19 is also emerging as a disease that potentially triggers autoimmunity, and this raises the case for severe and lasting effects on various systems, including reproductive capacity, as seen with several autoimmune rheumatic diseases [3,4,5]. Moreover, several new drugs being used to treat these conditions may potentially have a bearing on future fertility, a risk that remains largely unexplored [6, 7].
ACE-2 receptor is present in higher concentrations in testes and in ovaries. It is hypothesized and proved by some small cohorts that SARS-CoV-2 can result in impaired spermatogenesis, by direct viral mediated destruction or via autoimmune orchitis or thrombosis in males; and adversely affects the oocyte quality in females [8, 9]. Therefore, COVID-19 is known to cause alteration in both male and female reproductive functions, raising the concern of infertility. A keen understanding of these effects lies at the intersection of viral microbiology, immunology, reproductive medicine and hematology.
Effects of this kind may plausibly impact mankind and the generations to come in ways never imagined, have an outsized impact on population distribution in subsequent years, and even turn the economic roadmap upside down. In addition, the impact on fertility and birth trends in response to the pandemic may potentially vary according to the socioeconomic conditions, accessibility to contraception and assisted reproductive techniques (ART) [10]. Understanding these effects may be the first step towards formulating further course of action to prevent long-standing population-level morbidity. Therefore, this narrative review explores the effects of SARS-CoV-2 on human fertility and possibility of its sexual transmission from a gynecological, immunological, endocrinological and hematological viewpoint.
Methods
A literature search was conducted through the MEDLINE and SCOPUS databases to identify articles on the subject in English. Several types of articles, ranging from case reports, case series, letter to editors, systematic reviews, narrative reviews, and observational studies were reviewed published anytime till 13th June 2021. The search terms included: “COVID-19 OR SARS-CoV2 OR SARS-COV-2 OR novel coronavirus OR nCOV”, “Fertility OR Reproduction OR Spermatogenesis OR Pregnancy OR Sexual Health OR Birth OR Testes OR Ovaries OR Placenta OR ACE-2 OR Breast”, “Male OR Men”, “Female OR Women”, “Autoimmune OR Immunological” and “Vertical transmission, Sexual transmission”. Different combinations of the above-mentioned terms were used to identify around 4900 articles on 13th June 2021 (Supplementary figure). After an initial screening of Title and abstracts, 600 relevant articles were retained. Full text screening was then carried out independently by two co-authors; JT and LG, to shortlist approximately 250 articles meeting the inclusion criteria and relevant information was extracted from 73 articles for this review. Since, there is ample data on the effects of COVID-19 on pregnancy, articles dealing exclusively with this topic were excluded from review.
COVID-19 and female fertility
The ACE-2 receptor is widely expressed in uterus, ovaries, fallopian tubes, vagina and placenta; hence female reproductive system is predisposed to effects as a result of SARS-CoV-2 infection, although the risk is thought to be lesser as compared to males [11, 12]. The SARS CoV-2 affects the renin angiotensin aldosterone system (RAAS) which is involved in follicular maturation, ovulation phases, luteal angiogenesis and degeneration, and embryo development. Thus, COVID-19 infection may potentially adversely impact the ovarian granulosa cells or interfere with follicular growth, consequently affecting the quality of oocytes [11]. The ACE-2 receptor is also found in endometrium and the virus might intervene with implantation of embryo or lead to miscarriage [13].
A study by Ding et al. [14] showed the effect of COVID-19 on the ovarian function in women whereby higher levels of testosterone and prolactin, and lower levels of AMH (Anti-Müllerian hormone) were observed in infected patients as opposed to their healthy counterparts. Therefore, ovarian reserve can be significantly reduced in such patients [14]. On the contrary, Li et al.’s study did not show any alteration in sex hormones and AMH levels; but did notice delayed menstrual cycle and changes in menstrual volume in some patients [15]. Another study did notice raised LH levels in SARS-CoV-2-infected women, indicative of hypogonadism [9].
It is further suggested that stress and panic associated with the COVID-19 pandemic contribute to menstrual irregularities and adversely affect the quality of oocytes [16]. These psychological effects can also translate into sexual dysfunction; indirectly impacting conception [16]. It has been noticed that after SARS-CoV-2 infection, women had a decrease in frequency of sexual intercourse and satisfaction [17].
COVID-19 and male fertility
Studies in the past have shown that viruses like HPV, HIV, HSV, HBV, and HCV can adversely affect male fertility by causing orchitis or affecting the quality, motility, count, liquefaction time and morphology of sperms [18]. Thus, it is reasonable to extrapolate that SARS-CoV-2 too may have some effect on male fertility. The male spermatozoa has a high concentration of ACE-2 receptors and they can be a major target of direct destruction by SARS-CoV-2 which raises the concern about resulting male infertility and the risk of sexual transmission of the virus [1]. A study by Wang et al. [19] showed the highest concentration of ACE 2 receptors, within the testes, are in the spermatogonia, Sertoli and Leydig’s cells. When compared to ACE-2 negative cells, the ACE-2 receptor positive spermatogonia showed higher frequency of viral reproduction, viral gene expression and transmission. ACE-2 positive spermatogonia also showed significantly reduced spermatid differentiation, sperm-egg recognition, acrosome reaction, sperm chromatin condensation and male meiosis [19].
A study by Ma et al. [20] showed deranged ratios of testosterone to LH and of FSH to LH, indicating potential adverse effect on spermatogenesis. Some other studies also support the finding of decreased testosterone levels in SARS-CoV-2-infected males [21, 22]. Sperm quality was also seen to be affected in those with active infection particularly, sperm count and motility [23]. SARS-CoV-2 infection has been associated with orchitis. The possible mechanisms of orchitis could be autoimmune inflammatory orchitis or direct destruction of the testicular cells by the cytopathic effect of the virus [24, 25]. Another hypothesis is vascular thrombosis and vasculitis like phenomenon which may result in ischemic orchitis [26]. Elevated core body temperature during fevers caused by COVID could also negatively impact the physiology of spermatogenesis [27]. Another interesting aspect that has been discussed in literature is the derangement of the hypothalamic pituitary adrenal axis due to psychological stress of the disease, thus having adverse effects on spermatogenesis [16]. However, presence of SARS-CoV-2 in semen was not noticed in the majority of studies [28,29,30,31], but Li et al.’s study did show the presence in few patients [32].The possibility of sexual transmission cannot be completely excluded, since most studies are based on small sample sizes and have contradictory results. Similar to the effect on female fertility, current evidence on SARS-CoV-2’s effect on male fertility is divided.
Table 1 highlights the major findings about male and female infertility during COVID-19.
COVID-19—sexual and vertical transmission
A study conducted by Pan et al. [33] showed no evidence of presence of SARS-CoV-2 in the semen of infected patients, ruling out sexual transmission; however, long term effects are still unclear. Several other studies listed in Table 1, demonstrate the absence of viral shedding in semen; but Li et al.’s study demonstrated positive semen samples both in acutely infected and recovering patients [32]. Thus, current literature is non-conclusive regarding this aspect of COVID pathophysiology.
Data on the risk of vertical transmission of SARS-CoV-2 from mother to fetus is disputed, with most studies against it [34, 35] but some do favor the finding [36]. The co-expression of ACE-2 and TMPRSS2 was found to be negligible in placental tissues, thus minimizing the potential of entry of virus into the products of conception [37]. It was also proposed that Caveolin, a membrane bound structure which allows endocytosis of viruses is not present in syncytiotrophoblast, thus preventing inflammation [38]. However, the virus could use alternate entry routes which are yet to be discovered.
There are no reports of vertical transmission for SARS (severe acute respiratory syndrome) and MERS (middle east respiratory syndrome), but the same cannot be assumed for SARS-CoV-2. A study by Schwartz et al. [39] investigating 38 pregnant women did not show any vertical transmission, with negative RT-PCR results for placenta and no maternal deaths were recorded. Another study by Chen et al. investigating nine pregnant patients with COVID-19 infection, did not find presence of the virus in amniotic fluid, cord blood and breast milk, however; all births were via Cesarean section, all nine mothers did develop some symptoms, fetal distress was noticed in two cases but no death was reported [34].
A recent case report published in JAMA, supports the possibility of vertical transmission. The baby was born via cesarean to a COVID-19 positive female and tested positive for both IgG and IgM against SARS-CoV-2. IgM antibody cannot be attributed to placental transfer, and is indicative of in utero infection [13]. Another case reported a miscarriage in second trimester which seemingly was attributed to the inflammation at placenta based on the histopathological findings [40].
A study by Menter et al. [41] evaluated the placentas of 5 SARS-CoV-2 positive pregnant women which showed signs of placental villitis and maternal–fetal malperfusion; thus indicative of placental infection. These findings can further support the hypothesis of a potential vertical transmission to fetus due to inflammation and subsequent infection at placenta [41]. Also, the maternal vascular malperfusion and inflammation are known to be causes of earlier pregnancy loss, and might also be the reason for losses seen with COVID-19 infection [42]. In a small cohort study, Bergen reported histopathological findings of placentas of 20 COVID positive patients, out of which eight showed fetal vascular malperfusion without any umbilical cord abnormalities. This is suggestive of thrombosis in fetal vasculature as a possible etiology [43].
A possibility of transmission of SARS-CoV-2 infection through breast milk also exists. Some case reports showed presence of the virus in breast milk; however, only one infant tested positive for COVID-19 amongst these; however, the route of transmission in this child was not confirmed [44]. Periera et al. [45] did not find any transmission through breast milk.
Effect of COVID-19 on sexual health from an immunologic perspective
It has been proposed that SARS-CoV-2 could trigger an autoimmune reaction in the genetically predisposed population [46]. Immunologically mediated conditions like Immune thrombocytopenic purpura (ITP), Guillain-Barré syndrome (GBS), Miller-Fisher syndrome (MFS) have been reported in COVID-19 patients [47]. A study by Sacchi et al. investigated the presence of autoimmune markers in 40 SARS-CoV-2 positive patients to support their hypothesis of association of COVID-19 with autoimmunity: 57.5% patients were positive for ANA (anti-nuclear antibodies), 12.5% had anti-cardiolipin antibodies (aCL), 25% tested positive for ANCA (anti-neutrophil cytoplasmic antibodies), and 5% had anti-β2 glycoprotein (anti-β2 GPI). Therefore, a significant prevalence of autoantibodies were found in COVID-19 patients as compared to healthy counterparts [48]. Another study by Wang et al. shows higher prevalence and reactivity of autoantibodies in SARS-CoV-2-infected patients which are suggestive of their role in the associated pathology [49].
The findings of deep vein thrombosis, stroke, disseminated intravascular coagulopathy and other vascular manifestations in COVID-19 infection have been associated with presence of aPL (Anti-phospholipid antibodies) [47]. However, studies have noticed that COVID-19-associated aPL do not seem to be anti-domain 1 which are the very pathogenic antibodies in APS [50]. Auto-immune antibody-associated thrombosis have been reported in three SARS-CoV-2-infected patients [50]. aPL are autoantibodies known to be directly involved in pathogenesis of thrombotic damage in several organs and of obstetric complications with reports of miscarriages. Although the rate of pregnancy loss has been no different during the COVID-19 pandemic, some aPL-associated conditions like HELLP syndrome (hemolysis; elevated liver enzymes, and low platelet count) have been reported [47]. A case of ovarian thrombosis in a pregnant women with COVID-19 infection was also reported [51]. Based on scarce data it can’t be concluded confidently whether or not aPL autoantibodies play a role in determining pregnancy outcomes in COVID positive patients; but studies do suggest that aPL-induced thrombosis is enhanced during an infection or hyper-inflammatory condition which is noticed in COVID-19 [52].
It has been reported that prednisone used in the management of Juvenile Systemic Lupus Erythematosus (JSLE) resulted in amenorrhea with marked decrease in FSH and LH levels [53]. The high doses of steroids used to treat severe COVID-19 may have similar effect on the menstrual cycle. Also, considering COVID-19 is likely an autoimmune disease, it was proposed to use immunosuppressants for its management [53], which may potentially impact fertility and/or pregnancy outcomes [54].
SARS-CoV-2 infection is well-known to cause a cytokine storm which leads to overproduction of cytokines like IL-6, TNF-alpha that could affect the sexual health adversely [55, 56]. The therapeutic effects of steroids and IL-6 inhibitors in COVID-19 infection are suggestive of the underlying inflammatory mechanism as pathogenic [57]. In males, the associated inflammation disrupts the blood-testis barrier [58] and cause an orchitis-like syndrome [59]. The cytokines could also suppress the hypothalamic-pituitary–testicular axis, leading to hypogonadism and thus decreased testosterone levels [60]. Since SARS-CoV-2 is known to cause hypercoagulation due to vasculitis, damage to testes could also be due to segmental vasculitis [61]. It is suggested that orchitis could also be a result of vasculitis; hence, SARS-CoV2-induced immune response might be the reason behind damage to testes and alteration in hormone levels [60]. Moreover, IgG detected on immunohistochemistry of seminiferous tubules which is indicative of an autoimmune response leading to auto-immune orchitis [62, 63]. Anti-phospholipid antibodies were detected in COVID-positive patients which are hypothesized to intervene with fertility by acting against sperm, although this remains largely unproven [64,65,66].
A recent study addressing the hypothesis of role of autoimmunity in COVID-19 identified 28 human proteins harbouring regions homologous to SARS-CoV-2 peptides that could possibly be acting as autoantigens in COVID-19 patients displaying autoimmune conditions. Interestingly, these conserved regions are amongst the experimentally validated B cell epitopes of SARS-CoV-2 proteins. The reported human proteins have demonstrated presence of autoantibodies against them in typical autoimmune conditions which may explain the frequent occurrence of autoimmune conditions following SARS-CoV-2 infection. Moreover, the proposed autoantigens' widespread tissue distribution is suggestive of their involvement in multi-organ manifestations via molecular mimicry [67]. Another study found the presence of anti TPO (thyroid peroxidase antibodies) in long COVID patients on follow-up [68].
Likewise, Lingel et al. [69] noticed raised titers of anti-CCP (Cyclic Citrullinated peptide antibodies) and anti-TG (Tissue Transglutaminase) in SARS-CoV-2-infected patients on follow-up indicating the formation of auto-antibodies and hence the role of autoimmunity in COVID-19. COVID-19-associated viral myositis is also thought to be a result of the autoimmunity [70]. Also many patients with severe infection were noticed to have autoantibodies against NET (Neutrophil Extracellular Traps) which play a role in decreased clearance of NET, therefore, further exacerbating the severity of infection [71].
Areas of contradiction
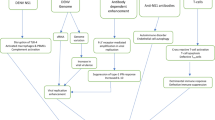
Several areas still require extensive research since most of the currently available data is contradictory, rendering it difficult to make a conclusive statement. It is still unclear whether or not sexual and vertical transmission occur, and, if it does, is it only restricted to certain stages of infection or depends or severity of infection. Although studies do highlight the role of autoimmunity in COVID-19, the crossroads of infection, autoimmunity and immunosuppression are yet to be solved. Also the extent of effect on fertility is not yet clear, but it is known that male fertility can be affected more than female fertility. Figure 1 summarizes the current findings.
Conclusion
The existing data give non-conclusive evidence about the impact of SARS-CoV-2 infection on fertility; however, a greater impact on male fertility as compared to female has been noticed. The presence of ACE2 in testes, with reports of autoimmune orchitis and impaired spermatogenesis, alteration in sex hormones and some findings of viral shedding in semen are definite areas of concern. However, most studies are observational with small sample size and therefore large, controlled trials or prospective studies are needed to make a definite conclusion. As for females, the SARS-CoV-2 infection has so far shown to be less dreadful to the pregnancy outcomes as compared to SARS and MERS. However, reproduction and fertility is a key concern and considering the pandemic is prolonged, natural conception or ART require extra precautions. The impact on fertility can change the socio-economic dynamics of all countries and that’s why it deserves prime attention, requiring an urgent need for global studies to address the plausible issue about male infertility.
Abbreviations
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus 2
- ACE-2:
-
Angiotensin converting enzyme-2
- TMPRSS2:
-
Transmembrane serine protease 2
- ART:
-
Assisted reproductive techniques
- RAAS:
-
Renin–angiotensin–aldosterone system
- HPA:
-
Hypothalamic–pituitary–adrenal axis
- AMH:
-
Anti-Müllerian hormone
- ITP:
-
Immune thrombocytopenic purpura
- GBS:
-
Guillain-Barré Syndrome
- MFS:
-
Miller-Fisher Syndrome
- ANA:
-
Anti-nuclear Antibodies
- aCL:
-
Anti-cardiolipin antibodies
- ANCA:
-
Anti-neutrophil cytoplasmic antibodies
- Anti-β2 GPI:
-
Anti-β2 glycoprotein
- aPL:
-
Anti-phospholipid antibodies
References
Aitken RJ (2021) COVID-19 and human spermatozoa—potential risks for infertility and sexual transmission? Andrology 9(1):48–52. https://doi.org/10.1111/andr.12859
Zou X, Chen K, Zou J, Han P, Hao J, Han Z (2020) Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med 14(2):185–192. https://doi.org/10.1007/s11684-020-0754-0
El Miedany Y, Palmer D (2021) Rheumatology-led pregnancy clinic: men perspective. Clin Rheumatol 40(8):3067–3077. https://doi.org/10.1007/s10067-020-05551-0
Almeida BP, Saad CG, Souza FH, Moraes JC, Nukumizu LA, Viana VS, Bonfá E, Silva CA (2013) Testicular sertoli cell function in ankylosing spondylitis. Clin Rheumatol 32(7):1075–1079. https://doi.org/10.1007/s10067-013-2215-5
Rajaei E, Shahbazian N, Rezaeeyan H, Mohammadi AK, Hesam S, Zayeri ZD (2019) The effect of lupus disease on the pregnant women and embryos: a retrospective study from 2010 to 2014. Clin Rheumatol 38(11):3211–3215. https://doi.org/10.1007/s10067-019-04682-3
Tomioka RB, Ferreira GRV, Aikawa NE, Maciel GAR, Serafini PC, Sallum AM, Campos LMA, Goldestein-Schainberg C, Bonfá E, Silva CA (2018) Non-steroidal anti-inflammatory drug induces luteinized unruptured follicle syndrome in young female juvenile idiopathic arthritis patients. Clin Rheumatol 37(10):2869–2873. https://doi.org/10.1007/s10067-018-4208-x
Jutiviboonsuk A, Salang L, Eamudomkarn N, Mahakkanukrauh A, Suwannaroj S, Foocharoen C (2021) Prevalence and clinical associations with premature ovarian insufficiency, early menopause, and low ovarian reserve in systemic sclerosis. Clin Rheumatol 40(6):2267–2275. https://doi.org/10.1007/s10067-020-05522-5
Fan C, Lu W, Li K, Ding Y, Wang J (2021) ACE2 expression in kidney and testis may cause kidney and testis infection in COVID-19 patients. Front Med 7:563893. https://doi.org/10.3389/fmed.2020.563893
Gomes PRC, da Rocha MDR, da Coelho FAR, Pinho S, de Lira JA, de Sousa Carmo RR, Nascimento HMS, de Oliveira SM, da Silva WR, Medeiros RG, Alves EHP, Dos Santos CA, Vasconcelos DFP (2021) Alterations of the male and female reproductive systems induced by COVID-19. Wien Klin Wochenschr 28:1–7. https://doi.org/10.1007/s00508-021-01875-2
Aassve A, Cavalli N, Mencarini L, Plach S, Bacci ML (2020) The COVID-19 pandemic and human fertility. Science 369(6502):370–371. https://doi.org/10.1126/science.abc9520
Jing Y, Run-Qian L, Hao-Ran W, Hao-Ran C, Ya-Bin L, Yang G, Fei C (2020) Potential influence of COVID-19/ACE2 on the female reproductive system. Mol Hum Reprod 26(6):367–373. https://doi.org/10.1093/molehr/gaaa030
Huang HH, Wang PH, Yang YP, Chou SJ, Chu PW, Wu GJ, Chang CC (2020) A review of severe acute respiratory syndrome coronavirus 2 infection in the reproductive system. J Chin Med Assoc 83(10):895–897. https://doi.org/10.1097/JCMA.0000000000000388
Dong L, Tian J, He S, Zhu C, Wang J, Liu C, Yang J (2020) Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn. JAMA 323(18):1846–1848. https://doi.org/10.1001/jama.2020.4621
Ding T, Wang T, Zhang J, Cui P, Chen Z, Zhou S, Yuan S, Ma W, Zhang M, Rong Y, Chang J, Miao X, Ma X, Wang S (2021) Analysis of ovarian injury associated with COVID-19 disease in reproductive-aged women in Wuhan, China: an observational study. Front Med 8:635255. https://doi.org/10.3389/fmed.2021.635255
Li K, Chen G, Hou H, Liao Q, Chen J, Bai H, Lee S, Wang C, Li H, Cheng L, Ai J (2021) Analysis of sex hormones and menstruation in COVID-19 women of child-bearing age. Reprod Biomed Online 42(1):260–267. https://doi.org/10.1016/j.rbmo.2020.09.020
Li R, Yin T, Fang F, Li Q, Chen J, Wang Y, Hao Y, Wu G, Duan P, Wang Y, Cheng D, Zhou Q, Zafar MI, Xiong C, Li H, Yang J, Qiao J (2020) Potential risks of SARS-CoV-2 infection on reproductive health. Reprod Biomed Online 41(1):89–95. https://doi.org/10.1016/j.rbmo.2020.04.018
Kaya Y, Kaya C, Tahta T, Kartal T, Tokgöz VY (2021) Examination of the effect of COVID-19 on sexual dysfunction in women. Int J Clin Pract 75(3):e13923. https://doi.org/10.1111/ijcp.13923
Batiha O, Al-Deeb T, Al-Zoubi E, Alsharu E (2020) Impact of COVID-19 and other viruses on reproductive health. Andrologia 52(9):e13791. https://doi.org/10.1111/and.13791
Wang Z, Xu X (2020) scRNA-seq profiling of human testes reveals the presence of the ACE2 receptor, a target for SARS-CoV-2 infection in spermatogonia, Leydig and Sertoli cells. Cells 9(4):920. https://doi.org/10.3390/cells9040920
Ma L, Xie W, Li D, Shi L, Mao Y, Xiong Y et al (2020) Effect of SARS-CoV-2 infection upon male gonadal function: a single center-based study. medRxiv. https://doi.org/10.1101/2020.03.21.20037267
Salonia A, Pontillo M, Capogrosso P, Gregori S, Tassara M, Boeri L, Carenzi C, Abbate C, Cignoli D, Ferrara AM, Cazzaniga W, Rowe I, Ramirez GA, Tresoldi C, Mushtaq J, Locatelli M, Santoleri L, Castagna A, Zangrillo A, De Cobelli F, Tresoldi M, Landoni G, Rovere-Querini P, Ciceri F, Montorsi F (2021) Severely low testosterone in males with COVID-19: a case-control study. Andrology 9(4):1043–1052. https://doi.org/10.1111/andr.12993
Schroeder M, Schaumburg B, Müller Z et al (2020) Sex hormone and metabolic dysregulations are associated with critical. medRxiv. https://doi.org/10.1101/2020.05.07.20073817
Holtmann N, Edimiris P, Andree M et al (2020) Assessment of SARS-CoV-2 in human semen-a cohort study. Fertil Steril 114(2):233–238. https://doi.org/10.1016/j.fertnstert.2020.05.028
Li H, Xiao X, Zhang J, Zafar MI, Wu C, Long Y, Lu W, Pan F, Meng T, Zhao K, Zhou L, Shen S, Liu L, Liu Q, Xiong C (2020) Impaired spermatogenesis in COVID-19 patients. EClinicalMedicine 28:100604. https://doi.org/10.1016/j.eclinm.2020.100604
Kharbach Y, Khallouk A (2021) Male genital damage in COVID-19 patients: are available data relevant? Asian J Urol 8(3):324–326. https://doi.org/10.1016/j.ajur.2020.06.005
José FG, González JGÁ, Molina JMC et al (2020) SARS-CoV-2 infection: implications for sexual and reproductive health. A position statement of the Asociación Española de Andrología, Medicina Sexual y Reproductiva (ASESA). Revista Internacional de Andrologia 18(3):117–123. https://doi.org/10.1016/j.androl.2020.06.001
Sergerie M, Mieusset R, Croute F, Daudin M, Bujan L (2007) High risk of temporary alteration of semen parameters after recent acute febrile illness. Fertil Steril 88(4):970. https://doi.org/10.1016/j.fertnstert.2006.12.045
Omolaoye TS, Adeniji AA, Maya WDC, du Plessis SS (2021) SARS-COV-2 (COVID-19) and male fertility: where are we? Reprod Toxicol (Elmsford, NY) 99:65–70. https://doi.org/10.1016/j.reprotox.2020.11.012
Song C, Wang Y, Li W, Hu B, Chen G, Xia P, Wang W, Li C, Diao F, Hu Z, Yang X, Yao B, Liu Y (2020) Absence of 2019 novel coronavirus in semen and testes of COVID-19 patients†. Biol Reprod 103(1):4–6. https://doi.org/10.1093/biolre/ioaa050
Paoli D, Pallotti F, Colangelo S, Basilico F, Mazzuti L, Turriziani O, Antonelli G, Lenzi A, Lombardo F (2020) Study of SARS-CoV-2 in semen and urine samples of a volunteer with positive naso-pharyngeal swab. J Endocrinol Invest 43(12):1819–1822. https://doi.org/10.1007/s40618-020-01261-1
Ning J, Li W, Ruan Y, Xia Y, Wu X, Hu K, Ding X, Wu X, Yu L, Zhou J, Mao Z, Xu W, Yu W, Cheng F (2020) Effects of 2019 novel coronavirus on male reproductive system: a retrospective study. Preprints. https://doi.org/10.20944/preprints202004.0280.v1
Li D, Jin M, Bao P, Zhao W, Zhang S (2020) Clinical characteristics and results of semen tests among men with coronavirus disease 2019. JAMA Netw Open 3(5):e208292. https://doi.org/10.1001/jamanetworkopen.2020.8292
Pan F, Xiao X, Guo J, Song Y, Li H, Patel DP, Spivak AM, Alukal JP, Zhang X, Xiong C, Li PS, Hotaling JM (2020) No evidence of severe acute respiratory syndrome-coronavirus 2 in semen of males recovering from coronavirus disease 2019. Fertil Steril 113(6):1135–1139. https://doi.org/10.1016/j.fertnstert.2020.04.024
Chen H, Guo J, Wang C, Luo F, Yu X, Zhang W, Li J, Zhao D, Xu D, Gong Q, Liao J, Yang H, Hou W, Zhang Y (2020) Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet (London, England) 395(10226):809–815. https://doi.org/10.1016/S0140-6736(20)30360-3
Liu W, Wang J, Li W, Zhou Z, Liu S, Rong Z (2020) Clinical characteristics of 19 neonates born to mothers with COVID-19. Front Med 14(2):193–198. https://doi.org/10.1007/s11684-020-0772-y
Yu N, Li W, Kang Q, Xiong Z, Wang S, Lin X, Liu Y, Xiao J, Liu H, Deng D, Chen S, Zeng W, Feng L, Wu J (2020) Clinical features and obstetric and neonatal outcomes of pregnant patients with COVID-19 in Wuhan, China: a retrospective, single-centre, descriptive study. Lancet Infect Dis 20(5):559–564. https://doi.org/10.1016/S1473-3099(20)30176-6
Sharps MC, Hayes DJL, Lee S, Zou Z, Brady CA, Almoghrabi Y, Kerby A, Tamber KK, Jones CJ, Adams Waldorf KM, Heazell AEP (2020) A structured review of placental morphology and histopathological lesions associated with SARS-CoV-2 infection. Placenta 101:13–29. https://doi.org/10.1016/j.placenta.2020.08.018
Celik O, Saglam A, Baysal B, Derwig IE, Celik N, Ak M, Aslan SN, Ulas M, Ersahin A, Tayyar AT, Duran B, Aydin S (2020) Factors preventing materno-fetal transmission of SARS-CoV-2. Placenta 97:1–5. https://doi.org/10.1016/j.placenta.2020.05.012
Schwartz DA (2020) An analysis of 38 pregnant women with COVID-19, their newborn infants, and maternal-fetal transmission of SARS-CoV-2: maternal coronavirus infections and pregnancy outcomes. Arch Pathol Lab Med 144(7):799–805. https://doi.org/10.5858/arpa.2020-0901-SA
Baud D, Greub G, Favre G, Gengler C, Jaton K, Dubruc E, Pomar L (2020) Second-trimester miscarriage in a pregnant woman with SARS-CoV-2 infection. JAMA 323(21):2198–2200. https://doi.org/10.1001/jama.2020.7233
Menter T, Mertz KD, Jiang S, Chen H, Monod C, Tzankov A, Waldvogel S, Schulzke SM, Hösli I, Bruder E (2021) Placental pathology findings during and after SARS-CoV-2 infection: features of villitis and malperfusion. Pathobiology 88(1):69–77. https://doi.org/10.1159/000511324
Man J, Hutchinson JC, Heazell AE, Ashworth M, Jeffrey I, Sebire NJ (2016) Stillbirth and intrauterine fetal death: role of routine histopathological placental findings to determine cause of death. Ultrasound Obstet Gynecol 48(5):579–584. https://doi.org/10.1002/uog.16019
Baergen RN, Heller DS (2020) Placental pathology in COVID-19 positive mothers: preliminary findings. Pediatr Dev Pathol 23(3):177–180. https://doi.org/10.1177/1093526620925569
Sharma I, Kumari P, Sharma A, Saha SC (2021) SARS-CoV-2 and the reproductive system: known and the unknown..!! Middle East Fertil Soc J 26(1):1. https://doi.org/10.1186/s43043-020-00046-z
Pereira A, Cruz-Melguizo S, Adrien M, Fuentes L, Marin E, Forti A, Perez-Medina T (2020) Breastfeeding mothers with COVID-19 infection: a case series. Int Breastfeed J 15(1):69. https://doi.org/10.1186/s13006-020-00314-8
Caso F, Costa L, Ruscitti P, Navarini L, Del Puente A, Giacomelli R, Scarpa R (2020) Could Sars-coronavirus-2 trigger autoimmune and/or autoinflammatory mechanisms in genetically predisposed subjects? Autoimmun Rev 19(5):102524. https://doi.org/10.1016/j.autrev.2020.102524
Ehrenfeld M, Tincani A, Andreoli L, Cattalini M, Greenbaum A, Kanduc D, Alijotas-Reig J, Zinserling V, Semenova N, Amital H, Shoenfeld Y (2020) COVID-19 and autoimmunity. Autoimmun Rev 19(8):102597. https://doi.org/10.1016/j.autrev.2020.102597
Sacchi MC, Tamiazzo S, Stobbione P, Agatea L, De Gaspari P, Stecca A, Lauritano EC, Roveta A, Tozzoli R, Guaschino R, Bonometti R (2021) SARS-CoV-2 infection as a trigger of autoimmune response. Clin Transl Sci 14(3):898–907. https://doi.org/10.1111/cts.12953
Wang EY, Mao T, Klein J, Dai Y, Huck JD, Jaycox JR, Liu F, Zhou T, Israelow B, Wong P, Coppi A, Lucas C, Silva J, Oh JE, Song E, Perotti ES, Zheng NS, Fischer S, Campbell M, Fournier JB, Wyllie AL, Vogels CBF, Ott IM, Kalinich CC, Petrone ME, Watkins AE, Yale IMPACT Team, Cruz CD, Farhadian SF, Schulz WL, Ma S, Grubaugh ND, Ko AI, Iwasaki A, Ring AM (2021) Diverse functional autoantibodies in patients with COVID-19. Nature 595(7866):283–288. https://doi.org/10.1038/s41586-021-03631-y
Zhang Y, Xiao M, Zhang S, Xia P, Cao W, Jiang W, Chen H, Ding X, Zhao H, Zhang H, Wang C, Zhao J, Sun X, Tian R, Wu W, Wu D, Ma J, Chen Y, Zhang D, Xie J, Yan X, Zhou X, Liu Z, Wang J, Du B, Qin Y, Gao P, Qin X, Xu Y, Zhang W, Li T, Zhang F, Zhao Y, Li Y, Zhang S (2020) Coagulopathy and antiphospholipid antibodies in patients with COVID-19. N Engl J Med 382(17):e38. https://doi.org/10.1056/NEJMc2007575
Mohammadi S, Abouzaripour M, Hesam Shariati N, Hesam Shariati MB (2020) Ovarian vein thrombosis after coronavirus disease (COVID-19) infection in a pregnant woman: case report. J Thromb Thrombolysis 50(3):604–607. https://doi.org/10.1007/s11239-020-02177-6
Shoenfeld Y, Blank M, Cervera R, Font J, Raschi E, Meroni PL (2006) Infectious origin of the antiphospholipid syndrome. Ann Rheum Dis 65(1):2–6. https://doi.org/10.1136/ard.2005.045443
Revannasiddaiah S, Kumar Devadas S, Palassery R, Kumar Pant N, Maka VV (2020) A potential role for cyclophosphamide in the mitigation of acute respiratory distress syndrome among patients with SARS-CoV-2. Med Hypotheses 144:109850. https://doi.org/10.1016/j.mehy.2020.109850
Gupta R, Deepanjali S, Kumar A, Dadhwal V, Agarwal SK, Pandey RM, Chaturvedi PK (2010) A comparative study of pregnancy outcomes and menstrual irregularities in northern Indian patients with systemic lupus erythematosus and rheumatoid arthritis. Rheumatol Int 30(12):1581–1585. https://doi.org/10.1007/s00296-009-1192-0
Pedersen SF, Ho YC (2020) SARS-CoV-2: a storm is raging. J Clin Investig 130(5):2202–2205. https://doi.org/10.1172/JCI137647
Maiorino MI, Bellastella G, Giugliano D, Esposito K (2018) From inflammation to sexual dysfunctions: a journey through diabetes, obesity, and metabolic syndrome. J Endocrinol Invest 41(11):1249–1258. https://doi.org/10.1007/s40618-018-0872-6
Mehta P, Fajgenbaum DC (2021) Is severe COVID-19 a cytokine storm syndrome: a hyperinflammatory debate. Curr Opin Rheumatol. https://doi.org/10.1097/BOR.0000000000000822 (Advance online publication)
Zhang H, Yin Y, Wang G, Liu Z, Liu L, Sun F (2014) Interleukin-6 disrupts blood-testis barrier through inhibiting protein degradation or activating phosphorylated ERK in sertoli cells. Sci Rep 4:4260. https://doi.org/10.1038/srep042604
Corona G, Baldi E, Isidori AM, Paoli D, Pallotti F, De Santis L, Francavilla F, La Vignera S, Selice R, Caponecchia L, Pivonello R, Ferlin A, Foresta C, Jannini EA, Lenzi A, Maggi M, Lombardo F (2020) SARS-CoV-2 infection, male fertility and sperm cryopreservation: a position statement of the Italian Society of Andrology and Sexual Medicine (SIAMS) (Società Italiana di Andrologia e Medicina della Sessualità). J Endocrinol Invest 43(8):1153–1157. https://doi.org/10.1007/s40618-020-01290-w
Huang C, Ji X, Zhou W, Huang Z, Peng X, Fan L, Lin G, Zhu W (2021) Coronavirus: a possible cause of reduced male fertility. Andrology 9(1):80–87. https://doi.org/10.1111/andr.12907
Leisman DE, Deutschman CS, Legrand M (2020) Facing COVID-19 in the ICU: vascular dysfunction, thrombosis, and dysregulated inflammation. Intensive Care Med 46(6):1105–1108. https://doi.org/10.1007/s00134-020-06059-6
Xu J, Qi L, Chi X, Yang J, Wei X, Gong E, Peh S, Gu J (2006) Orchitis: a complication of severe acute respiratory syndrome (SARS). Biol Reprod 74(2):410–416. https://doi.org/10.1095/biolreprod.105.044776
Itoh M, Hiramine C, Tokunaga Y, Mukasa A, Hojo K (1991) A new murine model of autoimmune orchitis induced by immunization with viable syngeneic testicular germ cells alone. II. Immunohistochemical findings of fully-developed inflammatory lesion. Autoimmunity 10(2):89–97. https://doi.org/10.3109/08916939109004812
Connell NT, Battinelli EM, Connors JM (2020) Coagulopathy of COVID-19 and antiphospholipid antibodies. J Thromb Haemost. https://doi.org/10.1111/jth.14893 (Advance online publication)
Long QX, Liu BZ, Deng HJ, Wu GC, Deng K, Chen YK, Liao P, Qiu JF, Lin Y, Cai XF, Wang DQ, Hu Y, Ren JH, Tang N, Xu YY, Yu LH, Mo Z, Gong F, Zhang XL, Tian WG, Hu L, Zhang XX, Xiang JL, Du HX, Liu HW, Lang CH, Luo XH, Wu SB, Cui XP, Zhou Z, Zhu MM, Wang J, Xue CJ, Li XF, Wang L, Li ZJ, Wang K, Niu CC, Yang QJ, Tang XJ, Zhang Y, Liu XM, Li JJ, Zhang DC, Zhang F, Liu P, Yuan J, Li Q, Hu JL, Chen J, Huang AL (2020) Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat Med 26(6):845–848. https://doi.org/10.1038/s41591-020-0897-1
Chiu WW, Chamley LW (2004) Clinical associations and mechanisms of action of antisperm antibodies. Fertil Steril 82(3):529–535. https://doi.org/10.1016/j.fertnstert.2003.09.084
Mohkhedkar M, Venigalla SSK, Janakiraman V (2021) Untangling COVID-19 and autoimmunity: identification of plausible targets suggests multi organ involvement. Mol Immunol 137:105–113. https://doi.org/10.1016/j.molimm.2021.06.021
Lui DTW, Lee CH, Chow WS, Lee ACH, Tam AR, Pang P, Ho TY, Fong CHY, Law CY, Leung EKH, To KKW, Tan KCB, Woo YC, Lam CW, Hung IFN, Lam KSL (2021) Long COVID in patients with mild to moderate disease: do thyroid function and autoimmunity play a role? Endocr Pract. https://doi.org/10.1016/j.eprac.2021.06.016 (Advance online publication)
Lingel H, Meltendorf S, Billing U, Thurm C, Vogel K, Majer C, Prätsch F, Roggenbuck D, Heuft HG, Hachenberg T, Feist E, Reinhold D, Brunner-Weinzierl MC (2021) Unique autoantibody prevalence in long-term recovered SARS-CoV-2-infected individuals. J Autoimmun 122:102682. https://doi.org/10.1016/j.jaut.2021.102682
Saud A, Naveen R, Aggarwal R, Gupta L (2021) COVID-19 and myositis: what we know so far. Curr Rheumatol Rep 23(8):63. https://doi.org/10.1007/s11926-021-01023-9
Zuo Y, Yalavarthi S, Navaz SA, Hoy CK, Harbaugh A, Gockman K, Zuo M, Madison JA, Shi H, Kanthi Y, Knight JS (2021) Autoantibodies stabilize neutrophil extracellular traps in COVID-19. JCI Insight 6(15):150111. https://doi.org/10.1172/jci.insight.150111
Funding
This study was not funded.
Author information
Authors and Affiliations
Contributions
LG was involved in the conceptualization and methodology. JT was involved in investigation/data curation. JT and LG were involved in writing of the original draft and LG, JT, TC and LA were involved in manuscript review and editing. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
JT, LG, TC and LA report no conflicts of interest/competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
No part of the manuscript was copied or published elsewhere.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tariq, J., Chatterjee, T., Andreoli, L. et al. COVID-19 and fertility—at the crossroads of autoimmunity and thrombosis. Rheumatol Int 41, 1885–1894 (2021). https://doi.org/10.1007/s00296-021-04977-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-021-04977-2