Abstract
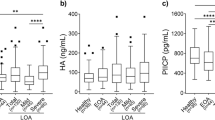
Biochemical markers reflecting joint remodeling in osteoarthritis (OA) are a promising diagnostic tool. The aim of this study was to investigate serum levels of candidate biomarkers in subjects with and without knee OA and assess their correlation with clinical parameters and knee structural damage. 56 patients with primary knee OA and 31 healthy controls participated in this study. Patients were separated into two groups: isolated knee OA and generalized OA. Clinical parameters were obtained by validated self-reported questionnaires and a visual analogue scale. Serum levels of cartilage oligomeric protein (COMP), matrix metalloproteinase-3 (MMP-3), and Coll2-1 were quantified by enzyme-linked immunosorbent assay. Knee structural damage was determined by plain X-ray and 1.5 T magnetic resonance imaging (MRI), using Kellgren–Lawrence (KL) grading scale and Whole-Organ Magnetic Resonance Imaging Score (WORMS), respectively. Compared to controls, patients had significantly higher median serum COMP (985 vs. 625 ng/ml; p < 0.001) and MMP-3 (36.85 vs. 22.10 ng/ml; p = 0.003) levels. Patients with radiographic evidence of KLII/III knee OA had greater median COMP levels than KLI patients (1095 vs. 720 ng/ml; p = 0.001). In the generalized OA group, mean MMP-3 levels were higher than in the isolated knee OA group (30.40 vs. 55.13 ng/ml; p < 0.001). COMP correlated positively with WORMS (r s = 0.454, p < 0.001) and MMP-3 (r s = 0.337, p = 0.003). Cut-off values for serum COMP and MMP-3 were determined. We observed higher serum COMP and MMP-3 levels in knee OA patients compared to controls. COMP may reflect knee structural damage, while MMP-3—OA “generalization”.



Similar content being viewed by others
References
Altman R, Asch E, Bloch D, Bole G, Borenstein D, Brandt K, Christy W, Cooke TD, Greenwald R, Hochberg M, Howell D, Kaplan D, Koopman W, Longley S III, Mankin H, McShane DJ, Medsger T Jr, Meenan R, Mikkelsen W, Moskowitz R, Murphy W, Rothschild B, Segal M, Sokoloff L, Wolfe F (1986) Development of criteria for the classification and reporting of osteoarthritis: classification of osteoarthritis of the knee. Arthritis Rheumatol 29(8):1039–1049
Hart DJ, Spector TD, Brown P, Wilson P, Doyle DV, Silman AJ (1991) Clinical signs of early osteoarthritis: reproducibility and relation to X-ray changes in 541 women in the general population. Ann Rheum Dis 50(7):467–470
Biomarkers Definitions Working Group (2001) Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin Pharmacol Ther 69: 89–95
Eckstein F, Burstein D, Link TM (2006) Quantitative MRI of cartilage and bone: degenerative changes in osteoarthritis. NMR Biomed 19(7):822–854
Pelletier JP, Cooper C, Peterfy C, Reginster JY, Brandi ML, Bruyère O, Chapurlat R et al (2013) What is the predictive value of MRI for the occurrence of knee replacement surgery in knee osteoarthritis? Ann Rheum Dis 72(10):1594–1604
Kraus VB, Burnett B, Coindreau J et al (2011) Application of biomarkers in the development of drugs intended for the treatment of osteoarthritis. Osteoarthr Cartil 19(5):515–542
Lotz M, Martel-Pelletier J, Christiansen C, Brandi ML, Bruyère O, Chapurlat R, Collette J et al (2013) Value of biomarkers in osteoarthritis: current status and perspectives. Ann Rheum Dis 72(11):1756–1763
Georgiev TA, Stoilov R, Ivanova M (2013) Biomarkers in osteoarthritis. Revmatologiia 21(4):21–25
Smith RKW, Heinegård D (2000) Cartilage oligomeric matrix protein (COMP) levels in digital sheath synovial fluid and serum with tendon injury. Equine Vet J 32(1):52–58
Bauer DC, Hunter DJ, Abramson SB et al (2006) Classification of osteoarthritis biomarkers: a proposed approach. Osteoarthr Cartil 14(8):723–727
Rousseau JC, Garnero P (2012) Biological markers in osteoarthritis. Bone 51(2):265–277
Bruyere O, Collette J, Kothari M et al (2006) Osteoarthritis, magnetic resonance imaging, and biochemical markers: a one year prospective study. Ann Rheum Dis 65:1050–1054
Vilım V, Olejarova M, Macháček S, Gatterova J, Kraus VB, Pavelka K (2002) Serum levels of cartilage oligomeric matrix protein (COMP) correlate with radiographic progression of knee osteoarthritis. Osteoarthr Cartil 10(9):707–713
Singh S, Shahi U, Kumar D, Shahi NT (2014) Serum Cartilage Oligomeric Matrix Protein: Tool for early diagnosis and grading of severity of primary knee osteoarthritis. Int J Osteol Orthop 1:1–7
Awadallah AM, Gehan HS, Tarek MK (2010) Serum level of cartilage oligomeric matrix protein as a screening modality for osteoarthritis among knee joint pain patients. J Am Sci 6(12)
Clark AG, Jordan JM, Vilim V, Renner JB, Dragomir AD, Luta G, Kraus VB (1999) Serum cartilage oligomeric matrix protein reflects osteoarthritis presence and severity: the Johnston County Osteoarthritis Project. Arthritis Rheumatol 42(11):2356–2364
Ribbens CY, Porras MM, Franchimont N et al (2002) Increased matrix metalloproteinase-3 serum levels in rheumatic diseases: relationship with synovitis and steroid treatment. Ann Rheum Dis 61(2):161–166
Kopchev A, Monov S, Kyurkchiev D, Ivanova I, Georgiev T (2017) Vascular endothelial growth factor (VEGF), cartilage oligomeric protein (COMP) and matrix metalloproteinase 3 (MMP-3) as serum biomarkers in psoriatic arthritis. Int J Pharm Sci Inven 6(7):08–12
Connelly AE, Tucker AJ, Kott LS, Duncan AM, Wright AJ (2014) Serum biochemical markers of joint metabolism and inflammation in relation to clinical symptoms and physical function in adults with symptomatic knee osteoarthritis. Osteoarthr Cartil 22:S66
Li W, Du C, Wang H, Zhang C (2014) Increased serum ADAMTS-4 in knee osteoarthritis: a potential indicator for the diagnosis of osteoarthritis in early stages. Genet Mol Res 13(4):9642–9649
Pelletier J, Raynauld J, Caron J, Mineau F, Abram F, Dorais M et al (2010) Decrease in serum level of matrix metalloproteinases is predictive of the disease-modifying effect of osteoarthritis drugs assessed by quantitative MRI in patients with knee osteoarthritis. Ann Rheum Dis 69(12):2095–2101
Deberg M, Labasse A, Christgau S, Cloos P et al (2005) New serum biochemical markers (Coll 2-1 and Coll 2-1 NO 2) for studying oxidative-related type II collagen network degradation in patients with osteoarthritis and rheumatoid arthritis. Osteoarthr Cartil 13(3):258–265
Henrotin Y, Conrozier T, Deberg M, Walliser-Lohse A, Richette P, Mulleman D et al (2013) Early decrease of serum biomarkers of type II collagen degradation (Coll2-1) and joint inflammation (Coll2-1 NO2) by hyaluronic acid intra-articular injections in patients with knee osteoarthritis: A research study part of the Biovisco study. J Orthop Res 31(6):901–907
Deberg MA, Labasse AH, Collette J, Seidel L, Reginster JY, Henrotin YE (2005) One-year increase of Coll 2-1, a new marker of type II collagen degradation, in urine is highly predictive of radiological OA progression. Osteoarthr Cartil 13:1059–1065
Lequesne MG, Mery C, Samson M, Gerard P (1987) Indexes of severity for osteoarthritis of the hip and knee. Validation–value in comparison with other assessment tests. Scand J Rheumatol Suppl 65:85–89
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt LW (1988) Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol 15:1833–1840
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Peterfy CG, Guermazi A, Zaim S et al (2004) Whole-organ magnetic resonance imaging score (WORMS) of the knee in osteoarthritis. Osteoarthr Cartil 12(3):177–190
Schiphof D, Boers M, Bierma-Zeinstra SM (2008) Differences in descriptions of Kellgren and Lawrence grades of knee osteoarthritis. Ann Rheum Dis 67(7):1034–1036
Verma P, Dalal K (2013) Serum cartilage oligomeric matrix protein (COMP) in knee osteoarthritis: a novel diagnostic and prognostic biomarker. J Orthop Res 31(7):999–1006
Naito К, Takahashi М, Kushida К, Suzuki М, Ohishi Т, Miura М, Inoue Т, Nagano А (1999) Measurement of matrix metalloproteinases (MMPs) and tissue inhibitor of metalloproteinases-1 (TIMP-1) in patients with knee osteoarthritis: comparison with generalized osteoarthritis. Rheumatology 38(6):510–515
Recht MP, Kramer J, Marcelis S, Pathria MN et al (1993) Abnormalities of articular cartilage in the knee: analysis of available MR techniques. Radiology 187:473–478
Rifai N, Paul MR (2001) High-sensitivity C-reactive protein: a novel and promising marker of coronary heart disease. Clin Chem 47(3):403–411
Danesh J, Wheeler JG, Hirschfield GM, Eda S et al (2004) C-reactive protein and other circulating markers of inflammation in the prediction of coronary heart disease. N Engl J Med 350(14):1387–1397
Pearson TA, Mensah GA, Alexander RW et al (2003) Markers of inflammation and cardiovascular disease. Circulation 107(3):499–511
Pearle AD, Scanzello CR, George S et al (2007) Elevated high-sensitivity C-reactive protein levels are associated with local inflammatory findings in patients with osteoarthritis. Osteoarthr Cartil 15(5):516–523
Miller ME, Rejeski WJ, Messier SP et al (2001) Modifiers of change in physical functioning in older adults with knee pain: the Observational Arthritis Study in Seniors (OASIS). Arthritis Rheum 45:331–339
Bedson J, Croft PR (2008) The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord 9:116
Stoilov R, Ivanova M, Garbeva K, Marinova N, Manolova I, Kalitchin Z (2011) Evaluation of lubrication and friction in knee joint attacked by osteoarthritis treated with ostenil, ostenil plus, yaral forte and durolane. J Balk Tribol Assoc 17(3):456–461
Felson DT, Naimark A, Anderson J, Kazis L, Castelli W, Meenan RF (1987) The prevalence of knee osteoarthritis in the elderly. Fram Osteoarthr Study Arthritis Rheumatol 30(8):914–918
Anderson АS, Richard FL (2010) Why is osteoarthritis an age-related disease? Best Pract Res Clin Rheumatol 24(1):15–26
Author information
Authors and Affiliations
Contributions
TG participated in constructing the idea behind this manuscript, executing the experiments, data management, analysis and reporting, logical interpretation, and presentation of the results. MI and AK took part in statistical analysis, logical interpretation and presentation of the results and in planning the methodology to reach the conclusion. AM and MP did an excellent work at designing and collecting the necessary data concerning the imaging modalities. Patient data collection and processing of relevant information was PK’s main task. TV, EK and KY carried out the immunoassays for detection of biomarkers. RR and RS organized and supervised the course of the work, which ended with reviewing the article before submission for its intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Funding
This work was supported by the Medical University of Sofia under project “Young Investigator” no 353/2015, contract no 22-D/2015.
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Rights and permissions
About this article
Cite this article
Georgiev, T., Ivanova, M., Kopchev, A. et al. Cartilage oligomeric protein, matrix metalloproteinase-3, and Coll2-1 as serum biomarkers in knee osteoarthritis: a cross-sectional study. Rheumatol Int 38, 821–830 (2018). https://doi.org/10.1007/s00296-017-3887-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-017-3887-y