Abstract
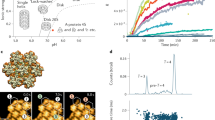
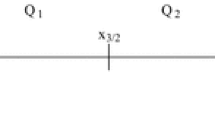
The formation of a viral capsid can be seen as a time-dependent process generated by the arrival of aggregates of various sizes at a nucleation site. Based on a model where aggregates arrive at a Poissonian rate to form a capsid particle, we develop kinetics assembly equations, that account for a finite size cluster and thus for rejection of too large aggregate. The model is derived under the assumption that the aggregate distribution has reached an exponential steady state. To account for the stochastic nature of the aggregates arrival, we also derive a stochastic equation to compute the mean time for a cluster to be formed. We find that this time has a minimum for a unique aggregate distribution. We obtain asymptotic expression for this time that we compare with numerically simulations. Finally, we find the mean size of the largest aggregate forming a viral capsid.






Similar content being viewed by others
References
Becker R, Döring W (1935) Kinetische Behandlung der Keimbildung in übersättigten Dämpfern. Ann Phys (Leipzig) 24:719–752
Chandrasekar S (1943) Stochastic problems in physics and astrophysics. Rev Mod Phys 15:1–89
Ding Y, Chuan YP, He L, Middelberg AP (2010) Modeling the competition between aggregation and self-assembly during virus-like particle processing. Biotechnol Bioeng 107(3):550–560
Doering CR, Ben-Avraham D (1989) Diffusion-limited coagulation in the presence of particle input: Exact results in one dimension. Phys Rev Lett 62:2563
D’Orsogna MR, Lakatos G, Chou T (2012) Stochastic nucleation of incommensurate clusters. J Chem Phys 136:084110
Endres D, Zlotnick A (2002) Model-based analysis of assembly kinetics for virus capsids or other spherical polymers. Biophys J 83(2):1217–1230
Gillespie DT (1977) Exact stochastic simulation of coupled chemical reactions. J Phys Chem 81(25):23402361
Gunzenhäuser J, Olivier N, Pengo T, Manley S (2012) Quantitative Super-resolution imaging reveals protein stoichiometry and nanoscale morphology of assembling HIV-Gag virions. Nano Lett 12(9):4705–4710
Hagan MF (2014) Modeling viral capsid assembly. Adv Chem Phys 155:1–42
Hoze N, Holcman D (2012) Coagulation-fragmentation for a finite number of particles and application to telomere clustering in the yeast nucleus. Phys Lett A 376(6–7):845–849
Hoze N, Holcman D (2013) Modeling capsid kinetics assembly from the steady state distribution of multi-sizes aggregates. Phys Lett A 376(6—-7):789–892
Ivanchenko S et al. (2009) Dynamics of HIV-1 assembly and release. PLoS Pathog 5(11):e1000652
Knobler CM, Gelbart WM (2009) Physical chemistry of DNA viruses. Annu Rev Phys Chem 60:367–383
Kumar MS, Schwartz R (2010) A parameter estimation technique for stochastic self-assembly systems and its application to human papillomavirus self-assembly. Phys Biol 7(4):045005
Ono A, Demirov D, Freed EO (2000) Relationship between human immunodeficiency virus type 1 Gag multimerization and membrane binding. J Virol 74:51425150
Schuss Z (2010) Diffusion and stochastic processes: an analytical approach. Springer, New York
von Smoluchowski M (1916) Versuch einer mathematischen Theorie der Koagulationskinetik kolloider. Lösungen Phys Z 17:557
Tritel M, Resh MD (2000) Kinetic analysis of human immunodeficiency virus type 1 assembly reveals the presence of sequential intermediates. J Virol 74:58455855
Twarock R (2006) Mathematical virology: a novel approach to the structure and assembly of viruses. Philos Transact A Math Phys Eng Sci 364(1849):3357–3373
Wattis JAD (2006) An introduction to mathematical models of coagulation fragmentation processes: a discrete deterministic mean-field approach. Phys D : Nonlinear Phenomena 222(1):120
Yvenic R, D’Orsogna MR, Chou T (2012) First passage times in stochastic self-assembly. J Chem Phys 137(24):244107
Zandi R, Reguera D, Bruinsma RF, Gelbart WM, Rudnick J (2004) Origin of icosahedral symmetry in viruses. Proc Natl Acad Sci USA 101(44):15556–15560
Acknowledgments
N. Hoze is supported by a Labex MemoLife fellowship. D. H. research is supported by an ERC-starting-Grant
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoze, N., Holcman, D. Kinetics of aggregation with a finite number of particles and application to viral capsid assembly. J. Math. Biol. 70, 1685–1705 (2015). https://doi.org/10.1007/s00285-014-0819-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-014-0819-2
Keywords
- Kinetics
- Model
- First passage time
- Takas equation
- Cluster formation
- Asymptotics
- Viral capsid assembly
- Aggregation
- Stochastic assembly
- Becker–Doering equation