Abstract
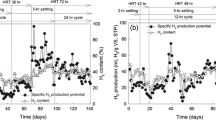
In this study, the biohydrogen production ability of isolated strains with sago industrial effluent in anaerobic batch fermentation was investigated. The bacteria responsible for the biohydrogen were isolated and identified as Clostridium sartagoforme NASGE 01 and Enterobacter cloacae NASGE 02. The volume of biohydrogen gas generated from the effluent was determined by gas chromatography (GC) and the organic acids formed during the biohydrogen production were determined by GC equipped with a flame ionization detector (GC—FID). In batch fermentation, C. sartagoforme NASGE 01 produced high amount of biogas (232 ± 11.02 mL/L) and biohydrogen (41.5%) followed by E. cloacae NASGE 02 produced 212.8 ± 8 mL/L biogas containing 31.5% of biohydrogen. Moreover, the hydrogen production potential (P), production rate (Rm) and lag time (λ) were analyzed from Gompertz non-linear curve fit model. The peak hydrogen yield was obtained with C. sartagoforme NASGE 01 was 158.7 mL/g glucose (1.26 mol H2/mol glucose) with the substrate degradation of 56.7%. Butyric acid was the major organic acid formed while hydrogen production with Clostridium sartagoforme NASGE 01 (176.4 mg/L) and Enterobacter cloacae NASGE 02 (285.1 mg/L). These experimental data demonstrated the feasibility of biohydrogen production using pure culture of anaerobic bacteria with sago industrial waste water as substrate.




Similar content being viewed by others
References
Sloan ED (2003) Fundamental principles and applications of natural gas hydrates. Nature 426:353–363. https://doi.org/10.1038/nature02135
Evans RL (2007) Fueling our future—an introduction to sustainable energy. Cambridge University Press, Cambridge, p 180
Wang J, Wan W (2009) Factors influencing fermentative hydrogen production: a review. Int J Hydrogen Energy 34:799–811. https://doi.org/10.1016/j.ijhydene.2008.11.015
Kapdan IK, Kargi F (2006) Bio-hydrogen production from waste materials. Enzym Microb Technol 38:569–582. https://doi.org/10.1016/j.enzmictec.2005.09.015
Nizzy AM, Kannan S (2014) Physicochemical properties of sago industrial effluents and their effects on seed germination. Int J Recent Sci Res 5(1):266–268
Nizzy AM, Kannan S, Anand SB (2019) Identification and characterization of bacteria isolated from sago industrial effluent. Indian J Exp Biol 57:427–434
Nasr M, Tawfik A, Ookowara S, Suzuki M (2013) Biological hydrogen production from starch waste water using a Novel Up-flow Anaerobic staged reactor. BioResources 8(4):4951–4968
Banu JR, Kaliappan S, Beck D (2006) High rate anaerobic treatment of Sago wastewater using HUASB with PUF as carrier. Int J Environ Sci technol 3:69–77
Muthukumaran VR (2011) Isolation and characterization of thiocyanate degrading bacteria from sago effluent contaminated site. Thesis submitted to Bharathidasan University, Tiruchirappalli, India, 84
Mycin TR, Lenin M (2012) Morphometrical and biochemical changes on sunflower (Helianthus annuus L.) under sago factory effluent. Int J Environ Biol 2:115–120
Rajivgandhi MMC, Singaravelu M, Kiruthika R (2013) Biogas generation potential from tapioca processing industry wastes in Tamil Nadu. Int J Eng Res Technol 2:1981–1983
Su H, Cheng J, Zhou J, Song W, Chen K (2009) Improving hydrogen production from cassava starch by combination of dark and photo fermentation. Int J Hydrogen Energy 34:1780–1786. https://doi.org/10.1016/j.ijhydene.2008.12.045
Cappelletti BM, Reginatto V, Amante ER, Antonio RV (2011) Fermentative production of hydrogen from cassava processing wastewater by Clostridium acetobutylicum. Renew Energy 36:3367–3372. https://doi.org/10.1016/j.renene.2011.05.015
Shorgani NKN, Tibin EM, Ali E, Hamid AA, Yusoff WMW, Kalil MS (2013) Biohydrogen production from agroindustrial wastes via Clostridium saccharoperbutylacetonicum N1–4 (ATCC 13564). Clean Technol Environ Policy 16(1):11–21. https://doi.org/10.1007/s10098-013-0586-6
Zhang JN, Li YH, Zheng HQ, Fan YT, Hou HW (2015) Direct degradation of cellulosic biomass to bio-hydrogen from a newly isolated strain Clostridium sartagoforme FZ11. Bioresour Technol 192:60–67. https://doi.org/10.1016/j.biortech.2015.05.034
Hamilton C, Calusinska M, Baptiste S, Masset J, Beckers L, Thonart P, Hiligsmann S (2018) Effect of the nitrogen source on the hydrogen production metabolism and hydrogenases of Clostridium butyricum CWBI1009. Int J Hydrogen Energy 43(11):5451–5462. https://doi.org/10.1016/j.ijhydene.2017.12.162
Turhal S, Turanbaev M, Argun H (2019) Hydrogen production from melon and watermelon mixture by dark fermentation. Int J Hydrogen Energy 44(34):18811–18817. https://doi.org/10.1016/j.ijhydene.2018.10.011
Mishra P, Das D (2014) Biohydrogen production from Enterobacter cloacae IIT-BT 08 using distillery effluent. Int J Hydrogen Energy 39(14):7496–7507. https://doi.org/10.1016/j.ijhydene.2013.08.100
Sun L, Huang A, Gu W, Ma Y, Zhu D, Wang G (2015) Hydrogen production by Enterobacter cloacae isolated from sugar refinery sludge. Int J Hydrogen Energy 40(3):1402–1407. https://doi.org/10.1016/j.ijhydene.2014.11.121
Mohanraj S, Anbalagan K, Rajaguru P, Pugalenthi V (2016) Effects of phytogenic copper nanoparticles on fermentative hydrogen production by Enterobacter cloacae and Clostridium acetobutylicum. Int J Hydrogen Energy 41:10639–10645. https://doi.org/10.1016/j.ijhydene.2016.04.197
Batista AP, Gouveia L, Marques P (2018) Fermentative hydrogen production from microalgal biomass by a single strain of bacterium Enterobacter Aerogenes—effect of operational conditions and fermentation kinetics. Renew Energy 119:203–209. https://doi.org/10.1016/j.renene.2017.12.017
Abd-Alla MH, Gabra FA, Danial AW, Abdel-Wahab AM (2019) Enhancement of biohydrogen production from sustainable orange peel wastes using Enterobacter species isolated from domestic wastewater. Int J Energy Res 43:391–404. https://doi.org/10.1002/er.4273
Skonieczny MT (2008) Biological hydrogen production from industrial wastewater with Clostridium beijerinckii. Thesis submitted to McGill University, Montreal. 5–6
Pattil UK, Muskan K (2009) Essentials of biotechnology. I.K. International Publishing House Pvt. Ltd., New Delhi, p 244
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Kim M, Oh S, Rakwal R, Liu C, Zhang Z (2013) 2nd international conference on environment, energy and biotechnology, IPCBEE, IACSIT Press, Singapore 51:87–93
Abdul PM, Jahim JM, Harun S, Markom M, Hassan O, Mohammad AW, Asis AJ (2013) Biohydrogen production from pentose-rich oil palm empty fruit bunch molasses: a first trial. Int J Hydrogen Energy 38:15693–15699. https://doi.org/10.1016/j.ijhydene.2013.05.050
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Calorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Xiao B, Liu J (2009) Biological hydrogen production from sterilized sewage sludge by anaerobic self-fermentation. J Hazard Mater 168:163–167. https://doi.org/10.1016/j.jhazmat.2009.02.008
Bellucci M, Botticella G, Francavilla M, Beneduce L (2016) Inoculum pre-treatment affects the fermentative activity of hydrogen-producing communities in the presence of 5-hydroxymethylfurfural. Appl Microbiol Biotechnol 100:493–504. https://doi.org/10.1007/s00253-015-7002-6
Patel SKS, Lee JK, Kalia VC (2017) Dark-fermentative biological hydrogen production from mixed biowastes using defined mixed cultures. Indian J Microbiol 57:171–176. https://doi.org/10.1007/s12088-017-0643-7
Luo G, Karakashev D, Xie L, Zhou Q, Angelidaki I (2011) Long-term effect of inoculum pretreatment on fermentative hydrogen production by repeated batch cultivations: homoacetogenesis and methanogenesis as competitors to hydrogen production. Biotechnol Bioeng 108:1816–1827. https://doi.org/10.1002/bit.23122
Thauer RK, Jungermann K, Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev 41:100–180
Kumar N, Das D (2000) Enhancement of hydrogen production by Enterobacter cloacae IIT-BT 08. Process Biochem 35:589–593. https://doi.org/10.1016/S0032-9592(99)00109-0
Khanna N, Kotay SM, Gilbert JJ, Das D (2011) Improvement of biohydrogen production by Enterobacter cloacae IIT-BT 08 under regulated pH. J Biotechnol 152:9–15. https://doi.org/10.1016/j.jbiotec.2010.12.014
Mezzatesta ML, Gona F, Stefani S (2012) Enterobacter cloacae complex: clinical impact and emerging antibiotic resistance. Future Microbiol 7:887–902. https://doi.org/10.2217/fmb.12.61
Izdebski R, Baraniak A, Herda M, Fiett J, Bonten MJ, Carmeli Y (2014) MLST reveals potentially high-risk international clones of Enterobacter cloacae. J Antimicrob Chemother 70:48–56. https://doi.org/10.1093/jac/dku359
Levin DB, Pitt L, Love M (2004) Biohydrogen production prospects and limitations to practical application. Int J Hydrogen Energy 29:173–185. https://doi.org/10.1016/S0360-3199(03)00094-6
Yin Y, Wang J (2016) Optimization of hydrogen production by response surface methodology using γ-irradiated sludge as inoculum. Energy Fuels 30:4096–4103. https://doi.org/10.1021/acs.energyfuels.6b00262
Shin HS, Youn JH, Kim SH (2004) Hydrogen production from food waste in anaerobic mesophilic and thermophilicacidogenesis. Int J Hydrogen Energy 29:1355–1363. https://doi.org/10.1016/j.ijhydene.2003.09.011
Amorim ELC, Amorim NCS, Alves I, Martins JS (2014) Biohydrogen production from Cassava wastewater in an anaerobic fluidized bed reactor. Braz J Chem Eng 31:603–612. https://doi.org/10.1590/0104-6632.20140313s00002458
Acknowledgements
This work was supported by DST—INSPIRE, Department of Science and Technology, New Delhi, India (Grant No. IF120738).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nizzy, A.M., Kannan, S. & Anand, S.B. Identification of Hydrogen Gas Producing Anaerobic Bacteria Isolated from Sago Industrial Effluent. Curr Microbiol 77, 2544–2553 (2020). https://doi.org/10.1007/s00284-020-02092-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-020-02092-2