Abstract
Background
Therapeutic approach for patients with metastatic breast cancer (MBC) is still controversial. This study was conducted to assess the efficacy and safety of bevacizumab in combination with docetaxel plus capecitabine as first-line treatment for MBC. The feasibility of bevacizumab maintenance therapy in this setting was also evaluated.
Patients and methods
In this single-arm, multicenter phase II study, patients received bevacizumab 15 mg/kg and docetaxel 60 mg/m2 on day 1, plus capecitabine 900 mg/m2 twice daily on days 1–14 every 21 days. Treatment was administered for up to 6 cycles, then bevacizumab continued until progressive disease. The primary end point was progression-free survival (PFS); secondary end points were tumor response rate, overall survival, and toxicity.
Results
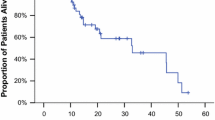
Seventy-nine eligible patients were treated with bevacizumab in combination with docetaxel plus capecitabine. The overall response rate was 61 %, with a complete response rate of 8 % and a median duration of response of 10 months. At a median follow-up of 28 months, the median PFS was 11 months. Fifty-two (65 %) patients received bevacizumab maintenance therapy for a median duration of 7 months (range 1 to 33+). Neutropenia was the most common grade 3–4 toxicity (28.1 % of patients), and two fatal adverse events occurred (septic shock and gastrointestinal perforation).
Conclusions
Bevacizumab in combination with docetaxel and capecitabine demonstrates significant activity and quite acceptable toxicity profile as first-line treatment of MBC. Subsequent maintenance therapy with bevacizumab is feasible for a long period of stable disease. Results deserve confirmation.

Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F et al. GLOBOCAN 2008 (2010) Cancer incidence and mortality worldwide: IARC CancerBase No. 10 [Internet]. International Agency for Research on Cancer. In. Lyon, France
Chia SK, Speers CH, D’Yachkova Y et al (2007) The impact of new chemotherapeutic and hormone agents on survival in a population-based cohort of women with metastatic breast cancer. Cancer 110:973–979
Dawood S, Broglio K, Gonzalez-Angulo AM et al (2008) Trends in survival over the past two decades among white and black patients with newly diagnosed stage IV breast cancer. J Clin Oncol 26:4891–4898
O’Shaughnessy J, Miles D, Vukelja S et al (2002) Superior survival with capecitabine plus docetaxel combination therapy in anthracycline-pretreated patients with advanced breast cancer: phase III trial results. J Clin Oncol 20:2812–2823
Albain KS et al (2008) Gemcitabine plus Paclitaxel versus Paclitaxel monotherapy in patients with metastatic breast cancer and prior anthracycline treatment. J Clin Oncol 26(24):3950–3957
Carrick S, Parker S, Thornton CE, Ghersi D, Simes J, Wilcken N (2009) Single agent versus combination chemotherapy for metastatic breast cancer (ReviewThe). Cochrane Library 2009, Issue 1
Verma S, Clemons M (2007) First-line treatment options for patients with HER-2 negative metastatic breast cancer: the impact of modern adjuvant chemotherapy. Oncologist 12:785–797
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676
Valachis A, Polyzos NP, Patsopoulos NA, Georgoulias V, Mavroudis D, Mauri D (2010) Bevacizumab in metastatic breast cancer: a meta-analysis of randomized controlled trials. Breast Cancer Res Treat 122:1–7
Miller K, Wang M, Gralow J et al (2007) Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med 357:2666–2676
Miles DW, Chan A, Dirix LY et al (2010) Phase III study of bevacizumab plus docetaxel compared with placebo plus docetaxel for the first-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J Clin Oncol 28:3239–3247
Gennari A, Amadori D, De Lena M et al (2006) Lack of benefit of maintenance paclitaxel in first-line chemotherapy in metastatic breast cancer. J Clin Oncol 24:3912–3918
Nooij MA, de Haes JC, Beex LV et al (2003) Continuing chemotherapy or not after the induction treatment in advanced breast cancer patients: clinical outcomes and oncologists’ preferences. Eur J Cancer 39:614–621
Patel JD, Hensing TA, Rademaker A et al (2009) Phase II study of pemetrexed and carboplatin plus bevacizumab with maintenance pemetrexed and bevacizumab as first-line therapy for nonsquamous non-small-cell lung cancer. J Clin Oncol 27:3284–3289
Wolmark N Yothers G O’Connell MJ et al (2009) A phase III trial comparing mFOLFOX6 to mFOLFOX6 plus bevacizumab in stage II or III carcinoma of the colon: results of NSABP protocol C-08. J Clin Oncol 27:LBA4
Miller VA, O’Connor P, Soh C et al (2009) A randomized, double-blind, placebo-controlled, phase IIIb trial (ATLAS) comparing bevacizumab (B) therapy with or without erlotinib (E) after completion of chemotherapy with B for first-line treatment of locally advanced, recurrent, or metastatic non-small cell lung cancer (NSCLC). J Clin Oncol 27:LBA8002
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47:207–214
National Cancer Institute. Common Terminology Criteria for Adverse Events v3.0 (CTCAE). National Cancer Institute 2006
Miles D, Chan A, Romieu G et al (2008) Randomized, double-blind, placebo-controlled, phase III study of bevacizumab with docetaxel or docetaxel with placebo as first-line therapy for patients with locally recurrent or metastatic breast cancer (mBC): AVADO. J Clin Oncol 26:LBA1011
Lueck H, Minckwitz GV, Du Bois A et al (2006) Epirubicin/paclitaxel (EP) versus capecitabine/paclitaxel (XP) in first-line metastatic breast cancer (MBC): a prospective, randomized multicentre phase III study of the AGO breast cancer study group. J Clin Oncol 24:517
Perez EA (2007) Novel enhanced delivery taxanes: an update. Semin Oncol 34(3):suppl 1–5
Lebowitz PF, Eng-Wong J, Swain SM et al (2004) A phase II trial of neoadjuvant docetaxel and capecitabine for locally advanced breast cancer. Clin Cancer Res 10:6764–6769
Cancer Folkman J (2005) Principles and practice of oncology. Lippincott, Williams & Wilkins, Philadelphia
Gray R, Bhattacharya S, Bowden C et al (2009) Independent review of E2100: a phase III trial of bevacizumab plus paclitaxel versus paclitaxel in women with metastatic breast cancer. J Clin Oncol 27:4966–4972
Robert NJ, Dieras V, Glaspy J et al (2011) RIBBON-1: randomized, double-blind, placebo-controlled, phase III trial of chemotherapy with or without bevacizumab for first-line treatment of human epidermal growth factor receptor 2-negative, locally recurrent or metastatic breast cancer. J Clin Oncol 29:1252–1260
Perez EA, Hillman DW, Dentchev T et al (2010) North Central Cancer Treatment Group (NCCTG) N0432: phase II trial of docetaxel with capecitabine and bevacizumab as first-line chemotherapy for patients with metastatic breast cancer. Ann Oncol 21:269–274
Cacheux W, Boisserie T, Staudacher L, Vignaux O, Dousset B, Soubrane O, Terris B, Mateus C, Chaussade S, Goldwasser F (2008) Reversible tumor growth acceleration following bevacizumab interruption in metastatic colorectal cancer patients scheduled for surgery. Ann Oncol 19:1651–1661
Richard M, Zuniga (2010) Rebound tumour progression after the cessation of bevacizumab therapy in patients with recurrent high-grade glioma. J Neurooncol 99:237–242
A Phase III Randomized Study Evaluating the Efficacy and Safety of Continued and Re-induced Bevacizumab in Combination With Chemotherapy for Patients With Locally Recurrent or Metastatic Breast cancer After First-line Chemotherapy and Bevacizumab Treatment. ClinicalTrials.gov identifier: NCT01250379
A Randomized Phase III Study to Determine the Efficacy of a Taxane and Bevacizumab With or Without Capecitabine as First Line Chemotherapy in Patients With Metastatic Breast Cancer. ClinicalTrials.gov identifier: NCT01200212
Acknowledgments
We thank all of the patients and Institutions involved in this study. The authors also thank Silvio Cavuto and Debora Formisano (S. Maria Nuova Hospital, Reggio Emilia) for statistical analysis. Medical writing assistance was provided by Andrea Bothwell in Science Communications, Springer Healthcare.
Conflict of interest
The authors declared that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was conducted on behalf of the Italian Oncology Group for Clinical Research (GOIRC).
Rights and permissions
About this article
Cite this article
Bisagni, G., Musolino, A., Panebianco, M. et al. The Breast Avastin Trial: phase II study of bevacizumab maintenance therapy after induction chemotherapy with docetaxel and capecitabine for the first-line treatment of patients with locally recurrent or metastatic breast cancer. Cancer Chemother Pharmacol 71, 1051–1057 (2013). https://doi.org/10.1007/s00280-013-2100-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-013-2100-6