Abstract
Purpose
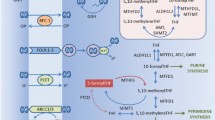
Oral leucovorin (LV) is used with uracil/tegafur (UFT) in the treatment of colorectal cancer (CRC). In order to find the factors related to the efficacy of LV in enhancing the antitumour effect of UFT, we investigated the relationships between the reduced folate levels in the CRC tissue after LV administration and the gene-expression levels of folate-metabolizing enzymes and folate transporters.
Methods
The subjects were 60 CRC patients, scheduled to undergo surgery. The control group (n = 30) did not receive LV. Three groups (n = 10 for each) received a single dose of oral LV at 25 mg, 4, 12 or 18 h before surgery (LV 4 h, LV 12 h or LV 18 h groups, respectively). The reduced folate levels in plasma and tissues were measured by high-performance liquid chromatography (HPLC) or a thymidylate synthase-FdUMP binding assay, respectively. The intratumoral expression levels of 34 genes were quantitatively evaluated with a real-time polymerase chain reaction (RT-PCR) assay.
Results
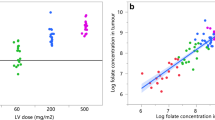
The reduced folate levels persisted for a longer period of time in the CRC tissue than in the plasma after LV administration. A multivariate logistic regression analysis revealed that high folylpolyglutamate synthase (FPGS) gene expression, low γ-glutamyl hydrolase (GGH) gene expression and low ATP-binding cassette sub-family C, number 1 (ABCC1) gene expression in CRC tissues were predictive factors for a high reduced folate level after LV administration.
Conclusions
The expression level of FPGS, GGH and ABCC1 in CRC tissues could predict the reduced folate level after LV administration, and these factors may determine the efficacy of LV treatment.


Similar content being viewed by others
References
Carmichael J, Popiela T, Radstone D, Falk S, Borner M, Oza A, Skovsgaard T, Munier S, Martin C (2002) Randomized comparative study of tegafur/uracil and oral leucovorin versus parenteral fluorouracil and leucovorin in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 20:3617–3627
Douillard JY, Hoff PM, Skillings JR, Eisenberg P, Davidson N, Harper P, Vincent MD, Lembersky BC, Thompson S, Maniero A, Benner SE (2002) Multicenter phase III study of uracil/tegafur and oral leucovorin versus fluorouracil and leucovorin in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 20:3605–3616
Lembersky BC, Wieand HS, Petrelli NJ, O’Connell MJ, Colangelo LH, Smith RE, Seay TE, Giguere JK, Marshall ME, Jacobs AD, Colman LK, Soran A, Yothers G, Wolmark N (2006) Oral uracil and tegafur plus leucovorin compared with intravenous fluorouracil and leucovorin in stage II and III carcinoma of the colon: results from National Surgical Adjuvant Breast and Bowel Project Protocol C-06. J Clin Oncol 24:2059–2064
Longley DB, Harkin DP, Johnston PG (2003) 5-Fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer 3:330–338
Priest DG, Schmitz JC, Bunni MA, Stuart RK (1991) Pharmacokinetics of leucovorin metabolites in human plasma as a function of dose administered orally and intravenously. J Natl Cancer Inst 83:1806–1812
Houghton JA, Williams LG, Cheshire PJ, Wainer IW, Jadaud P, Houghton PJ (1990) Influence of dose of [6RS]leucovorin on reduced folate pools and 5-fluorouracil-mediated thymidylate synthase inhibition in human colon adenocarcinoma xenografts. Cancer Res 50:3940–3946
Houghton JA, Williams LG, de Graaf SS, Cheshire PJ, Rodman JH, Maneval DC, Wainer IW, Jadaud P, Houghton PJ (1990) Relationship between dose rate of [6RS]leucovorin administration, plasma concentrations of reduced folates, and pools of 5, 10-methylenetetrahydrofolates and tetrahydrofolates in human colon adenocarcinoma xenografts. Cancer Res 50:3493–3502
Raghunathan K, Schmitz JC, Priest DG (1997) Impact of schedule on leucovorin potentiation of fluorouracil antitumor activity in dietary folic acid deplete mice. Biochem Pharmacol 53:1197–1202
Damle B, Ravandi F, Kaul S, Sonnichsen D, Ferreira I, Brooks D, Stewart D, Alberts D, Pazdur R (2001) Effect of food on the oral bioavailability of UFT and leucovorin in cancer patients. Clin Cancer Res 7:517–523
Shirao K, Hoff PM, Ohtsu A, Loehrer PJ, Hyodo I, Wadler S, Wadleigh RG, O’Dwyer PJ, Muro K, Yamada Y, Boku N, Nagashima F, Abbruzzese JL (2004) Comparison of the efficacy, toxicity, and pharmacokinetics of a uracil/tegafur (UFT) plus oral leucovorin (LV) regimen between Japanese and American patients with advanced colorectal cancer: joint United States and Japan study of UFT/LV. J Clin Oncol 22:3466–3474
Schlemmer M, Kuehl M, Schalhorn A, Rauch J, Jauch KW, Hentrich M (2008) Tissue levels of reduced folates in patients with colorectal carcinoma after infusion of folinic Acid at various dose levels. Clin Cancer Res 14:7930–7934
Tsukioka S, Uchida J, Tsujimoto H, Nakagawa F, Sugimoto Y, Oka T, Kiniwa M (2009) Oral fluoropyrimidine S-1 combined with leucovorin is a promising therapy for colorectal cancer: evidence from a xenograft model of folate-depleted mice. Mol Med Rep 2:393–398
Etienne MC, Speziale N, Milano G (1993) HPLC of folinic acid diastereoisomers and 5-methyltetrahydrofolate in plasma. Clin Chem 39:82–86
Belz S, Frickel C, Wolfrom C, Nau H, Henze G (1994) High-performance liquid chromatographic determination of methotrexate, 7-hydroxymethotrexate, 5-methyltetrahydrofolic acid and folinic acid in serum and cerebrospinal fluid. J Chromatogr B Biomed Appl 661:109–118
Etienne-Grimaldi MC, Francois E, Cardot JM, Renee N, Douillard JY, Gamelin E, Bennouna J, Chateau Y, Milano G (2007) A clinical pharmacokinetic analysis of tegafur-uracil (UFT) plus leucovorin given in a new twice-daily oral administration schedule. Clin Pharmacokinet 46:953–963
Suh JR, Herbig AK, Stover PJ (2001) New perspectives on folate catabolism. Annu Rev Nutr 21:255–282
Schneider E, Ryan TJ (2006) Gamma-glutamyl hydrolase and drug resistance. Clin Chim Acta 374:25–32
Sakamoto E, Tsukioka S, Oie S, Kobunai T, Tsujimoto H, Sakamoto K, Okayama Y, Sugimoto Y, Oka T, Fukushima M, Oka T (2008) Folylpolyglutamate synthase and gamma-glutamyl hydrolase regulate leucovorin-enhanced 5-fluorouracil anticancer activity. Biochem Biophys Res Commun 365:801–807
Kawakami K, Ooyama A, Ruszkiewicz A, Jin M, Watanabe G, Moore J, Oka T, Iacopetta B, Minamoto T (2008) Low expression of gamma-glutamyl hydrolase mRNA in primary colorectal cancer with the CpG island methylator phenotype. Br J Cancer 98:1555–1561
Assaraf YG (2006) The role of multidrug resistance efflux transporters in antifolate resistance and folate homeostasis. Drug Resist Updat 9:227–246
Rayl EA, Moroson BA, Beardsley GP (1996) The human purH gene product, 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase/IMP cyclohydrolase. Cloning, sequencing, expression, purification, kinetic analysis, and domain mapping. J Biol Chem 271:2225–2233
Galivan J, Ryan TJ, Chave K, Rhee M, Yao R, Yin D (2000) Glutamyl hydrolase pharmacological role and enzymatic characterization. Pharmacol Ther 85:207–215
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadahiro, S., Suzuki, T., Maeda, Y. et al. Molecular determinants of folate levels after leucovorin administration in colorectal cancer. Cancer Chemother Pharmacol 65, 735–742 (2010). https://doi.org/10.1007/s00280-009-1079-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-1079-5