Abstract
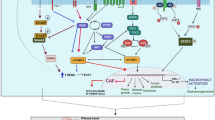
Hematopoietic stem cell transplantation (HSCT) recipients have been reported to have an increased risk of chronic graft versus host disease (cGVHD) and hematological and solid cancers. Oral manifestations are the first signs of cGVHD observed in the majority of patients, and oropharyngeal cancer is the most frequent secondary malignancy occurred after HSCT. In this study, we have evaluated the inflammatory infiltrate cell content and correlated with the vascular density in patients affected by primary oral squamous cell carcinoma (OSCC) from previous healthy controls and OSCC after cGVHD. Results have demonstrated that patients with OSCC after GVHD show a more consistent inflammatory infiltrate as compared with the OSCC ones. In detail, the inflammatory background composed of CD3-positive T cells, tryptase-positive mast cells, CD31-positive endothelial cells, and CD68-positive macrophages may be more pronounced in the setting of GVHD + OSCC than in the control group. By contrast, CD20-positive B cells and CD1a-positive dendritic cells were more abundant in the latter population. Finally, a positive correlation was found as between vascular density and inflammatory cell infiltration in both GVHD + OSCC and OSCC groups. Overall, these results confirm the role played by immune cells in enhancing tumor progression and angiogenesis and suggest a potential therapeutic strategy involving inhibition of recruitment of immune cells to the tumor microenvironment and blockade of pro-tumoral effects and pro-angiogenic functions.




Similar content being viewed by others
References
Danylesko I, Shimoni A (2018) Second malignancies after hematopoietic stem cell transplantation. Curr Treat Options in Oncol 19(2):9. https://doi.org/10.1007/s11864-018-0528-y
Qian L, Dima D, Berce C, Liu Y, Rus I, Raduly LZ, Liu Y, Petrushev B, Berindan-Neagoe I, Irimie A, Tanase A, Jurj A, Shen J, Tomuleasa C (2018) Protein dysregulation in graft versus host disease. Oncotarget 9(1):1483–1491. https://doi.org/10.18632/oncotarget.23276
Dander E, Balduzzi A, Zappa G, Lucchini G, Perseghin P, Andre V et al (2009) Interleukin-17-producing T-helper cells as new potential player mediating graft-versus-host disease in patients undergoing allogeneic stem-cell transplantation. Transplantation 88(11):1261–1272. https://doi.org/10.1097/TP.0b013e3181bc267e
Zeiser R, Sarantopoulos S, Blazar BR (2018) B-cell targeting in chronic graft-versus-host disease. Blood 131(13):1399–1405. https://doi.org/10.1182/blood-2017-11-784017
Majhail NS, Brazauskas R, Rizzo JD, Sobecks RM, Wang Z, Horowitz MM, Bolwell B, Wingard JR, Socie G (2011) Secondary solid cancers after allogeneic hematopoietic cell transplantation using busulfan-cyclophosphamide conditioning. Blood 117(1):316–322. https://doi.org/10.1182/blood-2010-07-294629
Inamoto Y, Shah NN, Savani BN, Shaw BE, Abraham AA, Ahmed IA, Akpek G, Atsuta Y, Baker KS, Basak GW, Bitan M, DeFilipp Z, Gregory TK, Greinix HT, Hamadani M, Hamilton BK, Hayashi RJ, Jacobsohn DA, Kamble RT, Kasow KA, Khera N, Lazarus HM, Malone AK, Lupo-Stanghellini MT, Margossian SP, Muffly LS, Norkin M, Ramanathan M, Salooja N, Schoemans H, Wingard JR, Wirk B, Wood WA, Yong A, Duncan CN, Flowers MED, Majhail NS (2015) Secondary solid cancer screening following hematopoietic cell transplantation. Bone Marrow Transplant 50(8):1013–1023. https://doi.org/10.1038/bmt.2015.63
Atsuta Y, Suzuki R, Yamashita T, Fukuda T, Miyamura K, Taniguchi S, Iida H, Uchida T, Ikegame K, Takahashi S, Kato K, Kawa K, Nagamura-Inoue T, Morishima Y, Sakamaki H, Kodera Y, Japan Society for Hematopoietic Cell Transplantation (2014) Continuing increased risk of oral/esophageal cancer after allogeneic hematopoietic stem cell transplantation in adults in association with chronic graft-versus-host disease. Ann Oncol 25(2):435–441. https://doi.org/10.1093/annonc/mdt558
Demarosi F, Lodi G, Carrassi A, Soligo D, Sardella A (2005) Oral malignancies following HSCT: graft versus host disease and other risk factors. Oral Oncol 41(9):865–877. https://doi.org/10.1016/j.oraloncology.2005.02.001
Adhikari J, Sharma P, Bhatt VR (2015) Risk of secondary solid malignancies after allogeneic hematopoietic stem cell transplantation and preventive strategies. Future Oncol 11(23):3175–3185. https://doi.org/10.2217/fon.15.252
Gallagher G, Forrest DL (2007) Second solid cancers after allogeneic hematopoietic stem cell transplantation. Cancer 109(1):84–92. https://doi.org/10.1002/cncr.22375
Leisenring W, Friedman DL, Flowers ME, Schwartz JL, Deeg HJ (2006) Nonmelanoma skin and mucosal cancers after hematopoietic cell transplantation. J Clin Oncol 24(7):1119–1126. https://doi.org/10.1200/JCO.2005.02.7052
Rizzo JD, Curtis RE, Socie G, Sobocinski KA, Gilbert E, Landgren O, Travis LB, Travis WD, Flowers MED, Friedman DL, Horowitz MM, Wingard JR, Deeg HJ (2009) Solid cancers after allogeneic hematopoietic cell transplantation. Blood 113(5):1175–1183. https://doi.org/10.1182/blood-2008-05-158782
Noce CW, Gomes A, Copello A, Barbosa RD, Sant’anna S, Moreira MC et al (2011) Oral involvement of chronic graft-versus-host disease in hematopoietic stem cell transplant recipients. Gen Dent 59(6):458–462 quiz 463–454
Imanguli MM, Pavletic SZ, Guadagnini JP, Brahim JS, Atkinson JC (2006) Early and late features of chronic graft-versus-host disease of oral mucosa: review of available therapies. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 101(2):175–183. https://doi.org/10.1016/j.tripleo.2005.08.028
Gomes AO, Torres SR, Maiolino A, Santos CW, Junior AS, Correa ME et al (2014) Not available. Rev Bras Hematol Hemoter 36(1):43–49. https://doi.org/10.5581/1516-8484.20140012
Weng X, Xing Y, Cheng B (2017) Multiple and recurrent squamous cell carcinoma of the oral cavity after graft-versus-host disease. J Oral Maxillofac Surg 75(9):1899–1905. https://doi.org/10.1016/j.joms.2017.02.012
de Araujo RL, Lyko Kde F, Funke VA, Torres-Pereira CC (2014) Oral cancer after prolonged immunosuppression for multiorgan chronic graft-versus-host disease. Rev Bras Hematol Hemoter 36(1):65–68. https://doi.org/10.5581/1516-8484.20140016
Hanahan D, Folkman J (1996) Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86(3):353–364
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285(21):1182–1186. https://doi.org/10.1056/NEJM197111182852108
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C et al (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964. https://doi.org/10.1126/science.1129139
Tamma R, Guidolin D, Annese T, Tortorella C, Ruggieri S, Rega S, Zito FA, Nico B, Ribatti D (2017) Spatial distribution of mast cells and macrophages around tumor glands in human breast ductal carcinoma. Exp Cell Res 359(1):179–184. https://doi.org/10.1016/j.yexcr.2017.07.033
Penack O, Socie G, van den Brink MR (2011) The importance of neovascularization and its inhibition for allogeneic hematopoietic stem cell transplantation. Blood 117(16):4181–4189. https://doi.org/10.1182/blood-2010-10-312934
Laishram D, Rao K, Devi HSU, Priya NS, Smitha T, Sheethal HS (2017) Mast cells and angiogenesis in malignant and premalignant oral lesions: an immunohistochemical study. J Oral Maxillofac Pathol 21(2):229–238. https://doi.org/10.4103/jomfp.JOMFP_111_15
Stasikowska-Kanicka O, Wagrowska-Danilewicz M, Danilewicz M (2017) Association of infiltrating cells with microvessel density in oral squamous cell carcinoma. Pol J Pathol 68(1):40–48. https://doi.org/10.5114/pjp.2017.67614
Ringden O, Brazauskas R, Wang Z, Ahmed I, Atsuta Y, Buchbinder D et al (2014) Second solid cancers after allogeneic hematopoietic cell transplantation using reduced-intensity conditioning. Biol Blood Marrow Transplant 20(11):1777–1784. https://doi.org/10.1016/j.bbmt.2014.07.009
Sloand EM, Pfannes L, Ling C, Feng X, Jasek M, Calado R, Tucker ZCG, Hematti P, Maciejewski J, Dunbar C, Barrett J, Young N (2010) Graft-versus-host disease: role of inflammation in the development of chromosomal abnormalities of keratinocytes. Biol Blood Marrow Transplant 16(12):1665–1673. https://doi.org/10.1016/j.bbmt.2010.07.014
Beachy PA, Karhadkar SS, Berman DM (2004) Tissue repair and stem cell renewal in carcinogenesis. Nature 432(7015):324–331. https://doi.org/10.1038/nature03100
Mashhadiabbas F, Fayazi-Boroujeni M (2017) Correlation of vascularization and inflammation with severity of oral leukoplakia. Iran J Pathol 12(3):225–230
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420(6917):860–867. https://doi.org/10.1038/nature01322
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140(6):883–899. https://doi.org/10.1016/j.cell.2010.01.025
Ribatti D, Crivellato E (2009) Immune cells and angiogenesis. J Cell Mol Med 13(9A):2822–2833. https://doi.org/10.1111/j.1582-4934.2009.00810.x
Ribatti D, Vacca A (2008) Overview of angiogenesis during tumor growth. In: Figg WD, Folkman J (eds) Angiogenesis. Springer, Boston
Nelson BH (2010) CD20+ B cells: the other tumor-infiltrating lymphocytes. J Immunol 185(9):4977–4982. https://doi.org/10.4049/jimmunol.1001323
Shen M, Sun Q, Wang J, Pan W, Ren X (2016) Positive and negative functions of B lymphocytes in tumors. Oncotarget 7(34):55828–55839. https://doi.org/10.18632/oncotarget.10094
Yang C, Lee H, Pal S, Jove V, Deng J, Zhang W, Hoon DSB, Wakabayashi M, Forman S, Yu H (2013) B cells promote tumor progression via STAT3 regulated-angiogenesis. PLoS One 8(5):e64159. https://doi.org/10.1371/journal.pone.0064159
Mor F, Quintana FJ, Cohen IR (2004) Angiogenesis-inflammation cross-talk: vascular endothelial growth factor is secreted by activated T cells and induces Th1 polarization. J Immunol 172(7):4618–4623
Carvalho MI, Pires I, Dias M, Prada J, Gregorio H, Lobo L et al (2015) Intratumoral CD3+ T-lymphocytes immunoexpression and its association with c-Kit, angiogenesis, and overall survival in malignant canine mammary tumors. Anal Cell Pathol (Amst) 2015:920409. https://doi.org/10.1155/2015/920409
Yu JL, Rak JW (2003) Host microenvironment in breast cancer development: inflammatory and immune cells in tumour angiogenesis and arteriogenesis. Breast Cancer Res 5(2):83–88
Bosisio D, Ronca R, Salvi V, Presta M, Sozzani S (2018) Dendritic cells in inflammatory angiogenesis and lymphangiogenesis. Curr Opin Immunol 53:180–186. https://doi.org/10.1016/j.coi.2018.05.011
Blois SM, Piccioni F, Freitag N, Tirado-Gonzalez I, Moschansky P, Lloyd R et al (2014) Dendritic cells regulate angiogenesis associated with liver fibrogenesis. Angiogenesis 17(1):119–128. https://doi.org/10.1007/s10456-013-9382-5
Qu Z, Liebler JM, Powers MR, Galey T, Ahmadi P, Huang XN, Ansel JC, Butterfield JH, Planck SR, Rosenbaum JT (1995) Mast cells are a major source of basic fibroblast growth factor in chronic inflammation and cutaneous hemangioma. Am J Pathol 147(3):564–573
Lee HS, Myers A, Kim J (2009) Vascular endothelial growth factor drives autocrine epithelial cell proliferation and survival in chronic rhinosinusitis with nasal polyposis. Am J Respir Crit Care Med 180(11):1056–1067. https://doi.org/10.1164/rccm.200905-0740OC
Cimpean AM, Tamma R, Ruggieri S, Nico B, Toma A, Ribatti D (2017) Mast cells in breast cancer angiogenesis. Crit Rev Oncol Hematol 115:23–26. https://doi.org/10.1016/j.critrevonc.2017.04.009
Funding
This work was supported in part by Fellowship FIRC-AIRC 1-year fellowship “Laura Bassi” id. 20879 to TA.
Author information
Authors and Affiliations
Contributions
All authors have seen and approved the manuscript being submitted.
Corresponding author
Ethics declarations
Full ethical approval and signed informed consent from individual patients were obtained to conduct the study. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Tamma, R., Limongelli, L., Maiorano, E. et al. Vascular density and inflammatory infiltrate in primary oral squamous cell carcinoma and after allogeneic hematopoietic stem cell transplantation. Ann Hematol 98, 979–986 (2019). https://doi.org/10.1007/s00277-018-3575-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-018-3575-3