Abstract
Background
Indocyanine green (ICG) is a near-infrared fluorescent contrast agent, which preferentially accumulates in cancer tissue. The aim of our study was to investigate the role of fluorescence imaging (FI) with ICG (ICG-FI) for detecting peritoneal carcinomatosis (PC) from colorectal cancer (CRC).
Methods
Four CRC patients with PC scheduled for cytoreductive surgery + hyperthermic intraperitoneal chemotherapy were enrolled in this prospective study. At a median time of 50 min after 0.25 mg/kg ICG injected intravenously, intraoperative ICG-FI using Fluobeam® was performed in vivo and ex vivo on all specimens. The Peritoneal Cancer Index was used to estimate the likelihood of complete cytoreduction.
Results
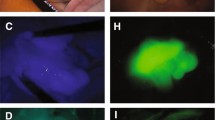
No severe complications were recorded. ICG-FI took a median of 20 min (range 10–30, IQR 15–25). Sixty-nine nodules were harvested. Fifty-two nodules had been diagnosed preoperatively by conventional imaging (n = 30; 43%) or intraoperatively by visual inspection/palpation (n = 22; 32%). With ICG-FI, 47 (90%) nodules were hyperfluorescent, and five hypofluorescent. Intraoperative ICG-FI identified 17 additional hyperfluorescent nodules. On histopathology, 16 were metastatic nodules. Sensitivity increased from 76.9%, with the conventional diagnostic procedures, to 96.9% with ICG-FI. The positive predictive value of ICG-FI was 98.4%, and test accuracy was 95.6%. Diagnostic performance of ICG-FI was significantly better than preoperative (p = 0.027) and intraoperative conventional procedures (p = 0.042). The median PCI score increased from 7 to 10 after ICG-FI (p < 0.001).
Conclusions
Our results suggest that intraoperative ICG-FI can improve outcomes in patients undergoing CS for PC from CRC. Further studies are needed to determine the role of ICG-FI in this patient population.



Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:359–386
Chu DZ, Lung NP, Thompson C et al (1989) Peritoneal carcinomatosis in non-gynecologic malignancies: a prospective study of prognostic factors. Cancer 63:364–367
Sadeghi B, Arvieux C, Glehen O et al (2000) Peritoneal carcinomatosis in non-gynecologic malignancies: results of EVOCAPE 1 multicentric prospective study. Cancer 88:358–363
Jessup MJ, Goldberg RM, Asara EA et al (2017) Colon and rectum. In: American joint committee on cancer. AJCC Cancer Staging Manual, 8 edn. Springer, Berlin, pp 251–274
Elias D, Faron M, Juga BS et al (2015) Prognostic similarities and differences in optimally resected liver metastases and peritoneal metastases from colorectal cancers. Ann Surg 261:157–163
Glehen O, Kwiatkowski F, Sugarbaker PH et al (2004) Cytoreductive surgery combined with preoperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: a multi-institutional study. J Clin Oncol 22:3284–3292
Da Silva RG, Sugarbaker PH (2006) Analysis of prognostic factors in seventy patients having complete cytoreduction plus perioperative intraperitoneal chemotherapy for carcinomatosis from colorectal cancer. J Am Coll Surg 203:878–886
Elias D, Raynard B, Farkhondeh F et al (2006) Long-term survival of peritoneal carcinomatosis of colorectal origin. Gastroenterol Clin Biol 30:1200–1204
Quere P, Facy O, Manfredi S et al (2015) Epidemiology, management, and survival of peritoneal carcinomatosis from colorectal cancer: a population-based study. Dis Colon Rectum 58:743–752
Blackham AU, Russell GB, Stewert JH 4th et al (2014) Metastatic colorectal cancer: survival comparison of hepatic resection versus cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann Surg Oncol 21:2667–2674
Begossi G, Gonzales-Moreno S, Ortega Perez G et al (2002) Cytoreduction and intraperitoneal chemotherapy for the management of peritoneal carcinomatosis, sarcomatosis and mesothelioma. Eur J Surg Oncol 28:80–87
Dromain C, Leboulleux S, Auperin A et al (2008) Staging of peritoneal carcinomatosis: enhanced CT vs. PET/CT. Abdom Imaging 33:87–89
Sugarbaker PH (1998) Intraperitoneal chemotherapy and cytoreductive surgery for the prevention and treatment of peritoneal carcinomatosis and sarcomatosis. Semin Surg Oncol 14:254–261
Liberale G, Vankerckhove S, Caldon MG et al (2016) Fluorescence imaging after indocyanine green injection for detection of peritoneal metastases in patients undergoing cytoreductive surgery for peritoneal carcinomatosis from colorectal cancer: a pilot study. Ann Surg 264:1110–1115
Sugarbaker PH (1999) Successful management of microscopic residual disease in large bowel cancer. Cancer Chemother Pharmacol 43:15–25
Gomez Portilla A, Sugarbaker PH, Chang D (1999) Second look surgery after cytoreductive and intraperitoneal chemotherapy for peritoneal carcinomatosis from colorectal cancer: analysis of prognostic features. World J Surg 23:23–29
Berthet B, Sugarbaker TA, Chang D et al (1999) Quantitative methodologies for selection of patients with recurrent abdominopelvic sarcoma for treatment. Eur J Cancer 3:413–419
Kim S, Lim YT, Soltesz EG et al (2004) Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat Biotechnol 22:93–97
Schaafsma BE, Mieog JSD, Hutteman M et al (2011) The clinical use of indocyanine green as a near-infrared fluorescent contrast agent for image-guided oncologic surgery. J Surg Oncol 104:323–332
Polom K, Murawa D, Rho Y et al (2011) Current trends and emerging future of indocyanine green usage in surgery and oncology: a literature review. Cancer 117:4817–4822
Fox IJ, Wood EH (1960) Indocyanine green: physical and physiologic properties. Proc Staff Meet Mayo Clin 35:732–744
Barabino G, Klein JP, Porcheron J et al (2016) Intraoperative near-infrared fluorescence imaging using indocyanine green in colorectal carcinomatosis surgery: proof of concept. Eur J Oncol Surg 42:1931–1937
Maeda H, Wu J, Sawa T et al (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release 65:271–284
Bekheit M, Vibert E (2015) Fluorescent-guided liver surgery: Paul Brousse experience and perspectives. In: Dip FD (ed) Fluorescence imaging for surgeons: concepts and applications, vol 11. Springer, Switzerland, pp 117–126
Frangioni J (2003) In vivo near-infrared fluorescence imaging. Curr Opin Chem Biol 7:626–634
Miyashiro I, Miyoshi N, Hiratsuka M et al (2008) Detection of sentinel node in gastric cancer surgery by indocyanine green fluorescence imaging: comparison with infrared imaging. Ann Surg Oncol 15:1640–1643
Takahashi H, Zaidi N, Berber E (2016) An initial report on the intraoperative use of indocyanine green fluorescence imaging in the surgical management of liver tumors. J Surg Oncol 114:625–629
Faybik P, Hetz H (2006) Plasma disappearance rate of indocyanine green in liver dysfunction. Transplant Proc 38:801–802
Honoré C, Goéré D, Souadka A et al (2013) Definition of patients presenting a high risk of developing peritoneal carcinomatosis after curative surgery for colorectal cancer: a systematic review. Ann Surg Oncol 20:183–192
Cortes-Guiral D, Elias D, Cascales-Campos PA et al (2017) Second-look surgery plus hyperthermic intraperitoneal chemotherapy for patients with colorectal cancer at high risk of peritoneal carcinomatosis: does it really save lives? World J Gastroenterol 23:377–381
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1.
Receiver operating characteristic curves of ICG-FI guided surgery, and pre- and intraoperative conventional technology (AUC= area under the curve) (TIFF 252 kb)
Rights and permissions
About this article
Cite this article
Lieto, E., Auricchio, A., Cardella, F. et al. Fluorescence-Guided Surgery in the Combined Treatment of Peritoneal Carcinomatosis from Colorectal Cancer: Preliminary Results and Considerations. World J Surg 42, 1154–1160 (2018). https://doi.org/10.1007/s00268-017-4237-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-4237-7