Abstract
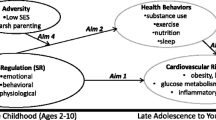
The pace-of-life syndrome (POLS) hypothesis posits that life-history characteristics, among individual differences in behavior, and physiological traits have coevolved in response to environmental conditions. This hypothesis has generated much research interest because it provides testable predictions concerning the association between the slow-fast life-history continuum and behavioral and physiological traits. Although humans are among the most well-studied species and similar concepts exist in the human literature, the POLS hypothesis has not yet been directly applied to humans. Therefore, we aimed to (i) test predicted relationships between life history, physiology, and behavior in a human population and (ii) better integrate the POLS hypothesis with other similar concepts. Using data of a representative sample of German adolescents, we extracted maturation status for girls (menarche, n = 791) and boys (voice break, n = 486), and a set of health-related risk-taking behaviors and cardiovascular parameters. Maturation status and health-related risk behavior as well as maturation status and cardiovascular physiology covaried in boys and girls. Fast maturing boys and girls had higher blood pressure and expressed more risk-taking behavior than same-aged slow maturing boys and girls, supporting general predictions of the POLS hypothesis. Only some physiological and behavioral traits were positively correlated, suggesting that behavioral and physiological traits might mediate life-history trade-offs differently. Moreover, some aspects of POLS were sex-specific. Overall, the POLS hypothesis shares many similarities with other conceptual frameworks from the human literature and these concepts should be united more thoroughly to stimulate the study of POLS in humans and other animals.
Significance statement
The pace-of-life syndrome (POLS) hypothesis suggests that life history, behavioral and physiological traits have coevolved in response to environmental conditions. Here, we tested this link in a representative sample of German adolescents, using data from a large health survey (the KIGGs study) containing information on individual age and state of maturity for girls and boys, and a set of health-related risk-taking behaviors and cardiovascular parameters. We found that fast maturing girls and boys had overall higher blood pressure and expressed more risk-taking behavior than same-aged slow maturing girls and boys. Only some behavioral and physiological traits were positively correlated, suggesting that behavioral and physiological traits might mediate life-history trade-offs differently and not necessarily form a syndrome. Our results demonstrate a general link between life history, physiological and behavioral traits in humans, while simultaneously highlighting a more complex and rich set of relationships, since not all relationships followed predictions by the POLS hypothesis.



Similar content being viewed by others
References
Alvergne A, Jokela M, Lummaa V (2010) Personality and reproductive success in a high-fertility human population. Proc Natl Acad Sci USA 107:11745–11750
Belsky J (2012) The development of human reproductive strategies: progress and prospects. Curr Dir Psychol Sci 21:310–316
Belsky J, Steinberg L, Draper P (1991) Childhood experience, interpersonal development, and reproductive strategy: an evolutionary theory of socialization. Child Dev 62:647–670
Bogin B (1999) Patterns of human growth. Cambridge University Press, Cambridge
Brener ND, Kann L, McManus T, Kinchen SA, Sundberg EC, Ross JG (2002) Reliability of the 1999 youth risk behavior survey questionnaire. J Adolesc Health 31:336–342
Brumbach BH, Figueredo AJ, Ellis BJ (2009) Effects of harsh and unpredictable environments in adolescence on development of life history strategies. Hum Nat 20:25–51
Careau V, Garland T (2012) Performance, personality, and energetics: correlation, causation, and mechanism. Physiol Biochem Zool 85:543–571
Careau V, Thomas D, Humphries M, Réale D (2008) Energy metabolism and animal personality. Oikos 117:641–653
Chen X, Wang Y, Mi J (2007) Tracking of blood pressure from childhood to adulthood: a systematic review and meta-analysis. FASEB J 21:A1363
Cho SD, Mueller WH, Meininger JC, Liehr P, Chan W (2001) Blood pressure and sexual maturity in adolescents: the heartfelt study. Am J Hum Biol 13:227–234
Croll J, Neumark-Sztainer D, Story M, Ireland M (2002) Prevalence and risk and protective factors related to disordered eating behaviors among adolescents: relationship to gender and ethnicity. J Adolescent Health 31:166–175
Dammhahn M, Dingemanse NJ, Niemelä P, Réale D (2018) Pace-of-life syndromes: a framework for the adaptive integration of behaviour, physiology and life-history. Behav Ecol Sociobiol. (in press)
Del Giudice M, Belsky J (2011) The development of life history strategies: toward a multi-stage theory. In: Buss DM, Hawley PH (eds) The evolution of personality and individual differences. Oxford University Press, Oxford, pp 154–176
Del Giudice M, Ellis BJ, Shirtcliff EA (2011) The adaptive calibration model of stress responsivity. Neurosci Biobehav Rev 35:1562–1592
Development Core Team R (2014) R: a language and environment for statistical computing. The R Foundation for Statistical Computing. Austria, Vienna http://www.R-project.org
Dingemanse NJ, Bouwman KM, van de Pol M, van Overveld T, Patrick SC, Matthysen E, Quinn JL (2012) Variation in personality and behavioural plasticity across four populations of the great tit Parus major. J Anim Ecol 81:116–126
Dreyfus J, Jacobs DR, Mueller N, Schreiner PJ, Moran A, Carnethon MR, Demerath EW (2015) Age at menarche and cardiometabolic risk in adulthood: the coronary artery risk development in young adults study. J Pediatr 167:344–352
Ellis L (1988) Criminal behavior and r/K selection: an extension of gene-based evolutionary theory. Pers Individ Differ 9:697–708
Ellis BJ, Jackson JJ, Boyce WT (2006) The stress response systems: universality and adaptive individual differences. Dev Rev 26:175–212
Ellis BJ, Figueredo AJ, Brumbach BH, Schlomer GL (2009) Fundamental dimensions of environmental risk. Hum Nat 20:204–268
Ellis BJ, Shirtcliff EA, Boyce WT, Deardorff J, Essex MJ (2011) Quality of early family relationships and the timing and tempo of puberty: effects depend on biological sensitivity to context. Dev Psychopathol 23:85–99
Ellis BJ, Del Giudice M, Dishion TJ et al (2012) The evolutionary basis of risky adolescent behavior: implications for science, policy, and practice. Dev Psychol 48:598–623
Ellis BJ, Del Giudice M, Shirtcliff EA (2017) The adaptive calibration model of stress responsitivity: concepts, findings, and implications for developmental psychopathology. In: Beauchaine TP, Hinshaw SP (eds) Child and adolescent psychopathology, 3rd edn. Wiley & Sons, New York, pp 237–276
Eveleth PB, Tanner JM (1990) Worldwide variation in human growth. Cambridge University Press, Cambridge
Fabrigar LR, Wegener DT, MacCallum RC, Strahan EJ (1999) Evaluating the use of exploratory factor analysis in psychological research. Psychol Methods 4:272–299
Fan Y, Chen J, Shirkey G, John R, Wu SR, Park H, Shao C (2016) Applications of structural equation modeling (SEM) in ecological studies: an updated review. Ecol Process 5:19–31
Figueredo AJ, Rushton JP (2009) Evidence for shared genetic dominance between the general factor of personality, mental and physical health, and life history traits. Twin Res Hum Genet 12:555–563
Figueredo AJ, Vasquez G, Brumbach BH, Schneider SM (2004) The heritability of life history strategy: the k-factor, covitality, and personality. Soc Biol 51:121–143
Figueredo AJ, Sefcek JA, Vasquez G, Brumbach BH, King JE, Jacobs WJ (2005) Evolutionary personality psychology. In: Buss DM (ed) The handbook of evolutionary psychology. John Wiley, Hoboken, pp 851–877
Figueredo AJ, Vásquez G, Brumbach BH, Stephanie M, Schneider R (2007) The k-factor, covitality, and personality. Hum Nat 18:47–73
Figueredo AJ, de Baca TC, Woodley MA (2013) The measurement of human life history strategy. Pers Individ Differ 55:251–255
Gosling SD (2001) From mice to men: what can we learn about personality from animal research? Psychol Bull 127:45–86
Graziano P, Derefinko K (2013) Cardiac vagal control and children's adaptive functioning: a meta-analysis. Biol Psychol 94:22–37
Griskevicius V, Tybur JM, Delton AW, Robertson TE (2011) The influence of mortality and socioeconomic status on risk and delayed rewards: a life history theory approach. J Pers Soc Psychol 100:1015–1026
Hämäläinen A, Immonen E, Tarka M, Schütt W (2018) Evolution of sex-specific pace-of-life syndromes: causes and consequences. Behav Ecol Sociobiol. https://doi.org/10.1007/s00265-018-2466-x
Harries M, Walker JM, Williams DM, Hawkins S, Hughes I (1997) Changes in the male voice at puberty. Arch Dis Child 77:445–447
Hill K, Kaplan H (1999) Life history traits in humans: theory and empirical studies. Annu Rev Anthropol 28:397–430
Hölling H, Kamtsiuris P, Lange M, Thierfelder W, Thamm M, Schlack R (2007) The German health interview and examination survey for children and adolescents (KiGGS): study management and conduct of fieldwork. Bundesgesundheitsbla 50:557–566
Hölling H, Schlack R, Kamtsiuris P, Butschalowsky H, Schlaud M, Kurth BM (2012) The KiGGS study. Nationwide representative longitudinal and cross-sectional study on the health of children and adolescents within the framework of health monitoring at the Robert Koch Institute. Bundesgesundheitsbla 55:836–842
Höpker T, Lampert T, Spallek J (2014) Identification and characterisation of health behaviours in 11- to 17-year-old adolescents: a cluster analysis based on the German health interview and examination survey for children and adolescents. Gesundheitswesen 76:453–461
Hoyle RH (1995) Structural equation modeling: concepts, issues, and applications. Sage, Thousand Oaks
Huisman H, Schutte A, Van Rooyen J, Malan N, Malan L, Schutte R, Kruger A (2006) The influence of testosterone on blood pressure and risk factors for cardiovascular disease in a black South African population. Ethnic Dis 16:693–698
Immonen E, Hämäläinen A, Schütt W, Tarka M (2018) Evolution of sex-specific pace-of-life syndromes: genetic architecture and physiological mechanisms. Behav Ecol Sociobiol. https://doi.org/10.1007/s00265-018-2462-1
Jelenkovic A, Rebato E (2016) Association of maternal menarcheal age with anthropometric dimensions and blood pressure in children from Greater Bilbao. Ann Hum Biol 43:430–437
Jokela M, Kivimäki M, Elovainio M, Keltikangas-Järvinen L (2009) Personality and having children: a two-way relationship. J Pers Soc Psychol 96:218–230
Jokela M, Alvergne A, Pollet TV, Lummaa V (2011) Reproductive behavior and personality traits of the five factor model. Eur J Pers 25:487–500
Juul A, Magnusdottir S, Scheike T, Prytz S, Skakkebæk NE (2007) Age at voice break in Danish boys: effects of pre-pubertal body mass index and secular trend. Int J Androl 30:537–542
Kamtsiuris P, Lange M, Rosario AS (2007) Der Kinder- und Jugendgesundheitssurvey (KiGGS): Stichprobendesign, Response und Nonresponse-Analyse. Bundesgesundheitsbla 50:547–556
Kann L, McManus T, Harris WA et al (2016) Youth risk behavior surveillance—United States, 2015. MMWR Surveill Summ 65:1–174
Kaplan H, Hill K, Lancaster J, Hurtado AM (2000) A theory of human life history evolution: diet, intelligence, and longevity. Evol Anthropol 9:156–185
Kline RB (2011) Principles and practice of structural equation modeling. The Guilford Press, New York
Koelsch S, Enge J, Jentschke S (2012) Cardiac signatures of personality. PLoS One 7:e31441
Koolhaas J, Korte S, de Boer S, van der Vegt B, van Reenen C, Hopster H, de Jong I, Ruis M, Blokhuis H (1999) Coping styles in animals: current status in behavior and stress-physiology. Neurosci Biobehav Rev 23:925–935
Koolhaas J, de Boer S, Coppens C, Buwalda B (2010) Neuroendocrinology of coping styles: towards understanding the biology of individual variation. Front Neuroendocrinol 31:307–321
Korkmaz S, Goksuluk D, Zararsiz G (2014) MVN: an R package for assessing multivariate normality. R J 6:151–162
Kurth B-M, Kamtsiuris P, Hölling H, Schlaud M, Dölle R, Ellert U, Kahl H, Knopf H, Lange M, Mensink GB (2008) The challenge of comprehensively mapping children’s health in a nation-wide health survey: design of the German KiGGS-study. BMC Public Health 8:196
Linting M, van der Kooij A (2012) Nonlinear principal components analysis with CATPCA: a tutorial. J Pers Assess 94:12–25
Linting M, Meulman JJ, Groenen PJ, van der Koojj AJ (2007) Nonlinear principal components analysis: introduction and application. Psychol Methods 12:336–358
MacArthur RH (1962) Some generalized theorems of natural selection. Proc Natl Acad Sci USA 48:1893–1897
MacArthur RH, Wilson EO (2015) Theory of island biogeography (MPB-1), vol. 1. Princeton University Press, Princeton
Mardia KV (1970) Measures of multivariate skewnees and kurtosis with applications. Biometrika 57:519–530
Marshall WA, Tanner JM (1969) Variations in pattern of pubertal changes in girls. Arch Dis Child 44:291–303
Mathot KJ, Dingemanse NJ (2015) Energetics and behavior: unrequited needs and new directions. Trends Ecol Evol 30:199–206
Mathot KJ, Frankenhuis WE (2018) Models of pace-of-life syndromes (POLS): a systematic review. Behav Ecol Sociobiol. https://doi.org/10.1007/s00265-018-2459-9
Montiglio P-O, Dammhahn M, Dubuc-Messier G, Réale D (2018) The pace-of-life syndrome revisited: the role of ecological conditions and natural history on the slow-fast continuum. Behav Ecol Sociobiol. (in press)
Najman JM, Hayatbakhsh MR, McGee TR, Bor W, O'Callaghan MJ, Williams GM (2009) The impact of puberty on aggression/delinquency: adolescence to young adulthood. Aust N Z J Criminol 42:369–386
National Research Council (2011) The science of adolescent risk-taking: workshop report. National Academies Press, Washington, DC
Nettle D (2005) An evolutionary approach to the extraversion continuum. Evol Hum Behav 26:363–373
Nettle D (2006) The evolution of personality variation in humans and other animals. Am Psychol 61:622–631
Nettle D, Penke L (2010) Personality: bridging the literatures from human psychology and behavioural ecology. Philos Trans R Soc B 365:4043–4050
Neuhauser H, Thamm M (2007) Blutdruckmessung im Kinder- und Jugendgesundheitssurvey (KiGGS). Bundesgesundheitsbla 50:728–735
Neuhauser HK, Thamm M, Ellert U, Hense HW, Rosario AS (2011) Blood pressure percentiles by age and height from nonoverweight children and adolescents in Germany. Pediatrics 127:e978-e988
Oli MK (2004) The fast–slow continuum and mammalian life-history patterns: an empirical evaluation. Basic Appl Ecol 5:449–463
Patel N, Walker N (2016) Clinical assessment of hypertension in children. Clin Hypertens 22:15–18
Pettay JE, Helle S, Jokela J, Lummaa V (2007) Natural selection on female life-history traits in relation to socio-economic class in pre-industrial human populations. PLoS One 2:e606
Pianka ER (1970) On r-and K-selection. Am Nat 104:592–597
Réale D, Dingemanse NJ (2010) Personality and individual social specialisation. In: Szekely T, Moore AJ, Komdeur J (eds) Social behaviour: genes, ecology and evolution. Cambridge University Press, New York, pp 417–441
Réale D, Garant D, Humphries MM, Bergeron P, Careau V, Montiglio PO (2010) Personality and the emergence of the pace-of-life syndrome concept at the population level. Philos Trans R Soc B 365:4051–4063
Rosario AS, Kurth B-M, Stolzenberg H, Ellert U, Neuhauser H (2010) Body mass index percentiles for children and adolescents in Germany based on a nationally representative sample (KiGGS 2003–2006). Eur J Clin Nutr 64:341–349
Rosseel Y (2012) lavaan: an R package for structural equation modeling. J Stat Softw 48:1–36
Royauté R, Berdal MA, Garrison CR, Dochtermann NA (2018) Paceless life? A meta-analysis of the pace-of- life syndrome hypothesis. Behav Ecol Sociobiol. https://doi.org/10.1007/s00265-018-2472-z
Rushton JP (1985) Differential K theory: the sociobiology of individual and group differences. Pers Indiv Differ 6:441–452
Simonetti GD, Schwertz R, Klett M, Hoffmann GF, Schaefer F, Wühl E (2011) Determinants of blood pressure in preschool children. The role of parental smoking. Circulation 123:292–298
Smith BH (1992) Life history and the evolution of human maturation. Evol Anthropol 1:134–142
Snodgrass JJ, Leonard WR, Sorensen MV, Tarskaia LA, Mosher MJ (2008) The influence of basal metabolic rate on blood pressure among indigenous Siberians. Am J Phys Anthropol 137:145–155
Stearns S (1982) The role of development in the evolution of life histories. In: Bonner JT (ed) Evolution and development. Dahlem workshop reports (life sciences research report), vol. 22. Springer, Berlin, pp 237–258
Stearns SC (1989) Trade-offs in life-history evolution. Funct Ecol 3:259–268
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
Steinberg L (2008) A social neuroscience perspective on adolescent risk-taking. Dev Rev 28:78–106
Susanne C, Bodzsar E (1998) Patterns of secular change of growth and development. In: Bodzsar EB, Susanne C (eds) Secular growth changes in Europe, 1st edn. Eötvös University Press, Budapest, pp 5–26
Swanson SA, Crow SJ, Le Grange D, Swendsen J, Merikangas KR (2011) Prevalence and correlates of eating disorders in adolescents: results from the national comorbidity survey replication adolescent supplement. Arch Gen Psychiat 68:714–723
Tanner J (1962) Growth at adolescence. Blackwell Scientific Publications, Oxford
Thayer JF, Åhs F, Fredrikson M, Sollers JJ, Wager TD (2012) A meta-analysis of heart rate variability and neuroimaging studies: implications for heart rate variability as a marker of stress and health. Neurosci Biobehav Rev 36:747–756
Uher J (2011) Personality in nonhuman primates: what can we learn from human personality psychology? In: Weiss A, King J, Murray L (eds) Personality and temperament in nonhuman primates. Springer, New York, pp 41–76
Ullman JB (2006) Structural equation modeling: reviewing the basics and moving forward. J Pers Assess 87:35–50
van der Steen M, Hokken-Koelega AC (2016) Growth and metabolism in children born small for gestational age. Endocrin Metab Clin 45:283–294
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor package. J Stat Softw 36:1–48
Walker R, Gurven M, Hill K, Migliano A, Chagnon N, De Souza R, Djurovic G, Hames R, Hurtado AM, Kaplan H (2006) Growth rates and life histories in twenty-two small-scale societies. Am J Hum Biol 18:295–311
Worthman CM (1999) Evolutionary perspectives on the onset of puberty. Evol Med:135–163
Acknowledgments
We thank all participants of the two workshops Towards a general theory of the pace-of-life syndrome, held in Hannover in 2015 and 2016, for inspiring discussions as well as the Volkswagen Stiftung (Az. 89905) for funding these workshops. We thank Marco Del Giudice, Denis Réale, Willem Frankenhuis and one anonymous reviewer as well as members of the Animal Ecology group at the University of Potsdam for providing constructive comments on earlier versions of the manuscript.
Funding
This study and two workshops Towards a general theory of the pace of life syndrome were funded by Volkswagen Stiftung (Az. 89905).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. The KiGGS survey was reviewed and approved by the responsible ethics committee at the University Hospital of the Charité of the Humboldt University in Berlin.
Additional information
Communicated by D. Réale
This article is a contribution to the Topical Collection Pace-of-life syndromes: a framework for the adaptive integration of behavior, physiology and life-history - Guest Editors: Melanie Dammhahn, Niels J. Dingemanse, Petri T. Niemelä, Denis Réale.
Rights and permissions
About this article
Cite this article
Lehmann, A., Eccard, J.A., Scheffler, C. et al. Under pressure: human adolescents express a pace-of-life syndrome. Behav Ecol Sociobiol 72, 57 (2018). https://doi.org/10.1007/s00265-018-2465-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-018-2465-y