Abstract
Purpose
The programmed death-1 (PD-1)/B7-H1 (also called PD-L1) pathway negatively regulates T cell activation and has been suggested to play an important role in regulating antitumor host immunity. To investigate the clinical significance of B7-H1 expression to the tumor grade and postoperative prognosis of patients with urothelial cancer, we analyzed the relationship between B7-H1 expression and various clinicopathological features and postoperative prognosis.
Experimental design
Sixty-five urothelial cancer cases were examined. B7-H1 expression in tumors and the numbers and phenotypes of tumor-infiltrating lymphocytes were evaluated by immunohistochemistry and flow cytometry.
Results
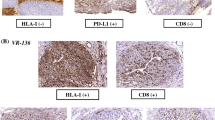
A substantial expression of B7-H1 was observed in all urothelial cancers investigated. Tumor specimens from patients with higher WHO grade or primary tumor classifications showed significantly higher percentages of tumor-associated B7-H1. Tumor-associated B7-H1 expression was significantly associated with a high frequency of postoperative recurrence and poor survival rate. Furthermore, multivariate analysis indicated that tumor-associated B7-H1 was more significant prognostic factor than WHO grade.
Conclusions
Our results demonstrate that the aberrant expression of B7-H1 in urothelial cancer is associated with aggressive tumors, suggesting a regulatory role of tumor-associated B7-H1 in antitumor immunity. Therefore, the manipulation of tumor-associated B7-H1 may become a beneficial target for immunotherapy in human urothelial cancer.




Similar content being viewed by others
References
Tsujihashi H, Matsuda H, Uejima S, Akiyama T, Kurita T (1988) Immunocompetence of tissue infiltrating lymphocytes in bladder tumors. J Urol 140:890–894
Tsujihashi H, Matsuda H, Uejima S, Akiyama T, Kurita T (1989) Immunoresponse of tissue infiltrating lymphocytes in bladder tumors. J Urol 141:1467–1470
Lipponen PK, Eskelinen MJ, Jauhiainen K, Harju E, Terho R (1993) Tumor-infiltrating lymphocytes as an independent prognostic factor in transitional cell bladder cancer. Eur J Cancer 29A:69–75
Patard JJ, Saint F, Velotti F, Abbou CC, Chopin DK (1998) Immune response following intravesical Bacillus Calmette-Guerin instillations in superficial bladder cancer: a review. Urol Res 26:155–159
Dong H, Chen L (2003) B7-H1 pathway and its role in the evasion of tumor immunity. J Mol Med 81:281–287
Blank C, Gajewski TF, Mackensen A (2005) Interaction of PD-L1 on tumor cells with PD-1 on tumor-specific T cells as a mechanism of immune evasion: implications for tumor immunotherapy. Cancer Immunol Immunother 54:307–314
Okazaki T, Iwai Y, Honjo T (2002) New regulatory co-receptors: inducible co-stimulator and PD-1. Curr Opin Immunol 14:779–782
Agata Y, Kawasaki A, Nishimura H, Ishida Y, Tsubata T, Yagita H, Honjo T (1996) Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int Immunol 8:765–772
Nishimura H, Nose M, Hirai H, Minato N, Honjo T (1999) Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 11:141–151
Nishimura H, Minato N, Nakano T, Honjo T (1998) Immunological studies on PD-1 deficient mice: implication of PD-1 as a negative regulator for B cell responses. Int Immunol 10:1563–1572
Nishimura H, Okazaki T, Tanaka Y, Nakatani K, Hara M, Matsunori A, Sasayama S, Mizoguchi A, Hiai H, Minato N, Honjo T (2001) Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science 291:319–322
Dong H, Zhu G, Tamada K, Chen L (1999) B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med 5:1365–1369
Latchman Y, Wood CR, Chernova T, Chaudhary D, Borde M, Chernova I, Iwai Y, Long AJ, Brown JA, Nunes R, Greenfield EA, Bourque K, Boussiotis VA, Carter LL, Carreno BM, Malenkovich N, Nishimura H, Okazaki T, Honjo T, Sharpe AH, Freeman GJ (2001) PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol 2:261–268
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC, Horton HF, Fouser L, Carter L, Ling V, Bowman MR, Carreno BM, Collins M, Wood CR, Honjo T (2000) Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192:1027–1034
Tamura H, Dong H, Zhu G, Sica GL, Flies DB, Tamada K, Chen L (2001) B7-H1 costimulation preferentially enhances CD28-independent T-help cell function. Blood 97:1809–1816
Youngnak-Piboonratanakit P, Tsushima F, Otsuki N, Igarashi H, Machida U, Iwai H, Takahashi Y, Omura K, Yokozeki H, Azuma M (2004) The expression of B7-H1 on keratinocytes in chronic inflammatory mucocutaneous disease and its regulatory role. Immunol Lett 94:215–222
Blank C, Brown I, Peterson AC, Spiotto M, Iwai Y, Honjo T, Gajewski TF (2004) PD-L1/B7H-1 inhibits the effector phase of tumor rejection by T cell receptor (TCR) transgenic CD8+ T cells. Cancer Res 64:1140–1145
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, Lennon VA, Celis E, Chen L (2002) Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8:793–800
Wintterle S, Screiner B, Mitsdoerffer M, Schneider D, Chen L, Meyermann R, Weller M, Wiendl H (2003) Expression of the B7-related molecule B7-H1 by glioma cells: a potential mechanism of immune paralysis. Cancer Res 63:7462–7467
Tsuda M, Matsumoto K, Inoue H, Matsumura M, Nakano T, Mori A, Azuma M, Nakanishi Y (2005) Expression of B7-H1 and B7-DC on the airway epithelium is enhanced by double-stranded RNA. Biochem Biophys Res Commun 330:263–270
Brown JA, Dorfman DM, Ma FR, Sullivan EL, Munoz O, Wood CR, Greenfield EA, Freeman GJ (2003) Blockade of programmed death-1 ligands on dendritic cells enhances T cell activation and cytokine production. J Immunol 170:1257–1266
Curiel TJ, Wei S, Dong H, Alvarez X, Cheng P, Mottram P, Krzysiek R, Knutson KL, Daniel B, Zimmermann MC, David O, Burow M, Gordon A, Dhurandhar N, Myers L, Berggern R, Hemminiki A, Alvarez RD, Emilie D, Curiel DT, Chen L, Zou W (2003) Blockade of B7-H1 improves myeloid dendritic cell-mediated antitumor immunity. Nat Med 9:562–567
Iwai Y, Ishida M, Tanaka Y, Okazaki T, Honjo T, Minato N (2002) Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci USA 99:12293–12297
Konishi J, Yamazaki K, Azuma M, Kinoshita I, Dosaka-Akita H, Nishimura M (2004) B7-H1 expression on non-small cell lung cancer cells and its relationship with tumor-infiltrating lymphocytes and their PD-1 expression. Clin Cancer Res 10:5094–5100
Ohigashi Y, Sho M, Yamada Y, Tsurui Y, Hamada K, Ikeba N, Mizuno T, Yoriki R, Kashizuka H, Yane K, Tsushima F, Otsuki N, Yagita H, Azuma M, Nakajima Y (2005) Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer. Clin Cancer Res 11:2947–2953
Thompson RH, Webster WS, Cheville JC, Lohse CM, Dong H, Leibovich BC, Kuntz SM, Sengupta S, Kwon ED, Blute ML (2005) B7-H1 glycoprotein blockade: a novel strategy to enhance immunotherapy in patients with renal cell carcinoma. Urology 66:10–14
Thompson RH, Gillett MD, Cheville JC, Lohse CM, Dong H, Webster WS, Krejci KG, Lobo JR, Sengupta S, Chen L, Zincke H, Blute ML, Strome SE, Leibovich BC, Kwon ED (2004) Costimulatory B7-H1 in renal cell carcinoma patients: Indicator of tumor aggressiveness and potential therapeutic target. Proc Natl Acad Sci USA 101:17174–17179
Thompson RH, Kuntz SM, Leibovich BC, Dong H, Lohse CM, Webster WS, Sengupta S, Frank I, Parker AS, Zincke H, Blute ML, Sebo TJ, Cheville JC, Kwon ED (2006) Tumor B7-H1 is associated with poor prognosis in renal cell carcinoma patients with long-term follow-up. Cancer Res 66:3381–3385
Carter L, Fouser LA, Jussif J, Fitz L, Deng B, Wood CR, Collins M, Honjo T, Freeman GJ, Carreno BM (2002) PD-1: PD-L inhibitory pathway affects both CD4 (+) and CD8 (+) T cells and is overcome by IL-2. Eur J Immunol 32:634–643
Youngnak P, Kozono Y, Kozono H, Iwai H, Otsuki N, Jin H, Omura K, Yagita H, Pardoll DM, Chen L, Azuma M (2003) Differential binding properties of B7-H1 and B7-DC to programmed death-1. Biochem Biophys Res Commun 307:672–677
Tsushima F, Tanaka K, Otsuki N, Youngnak P, Iwai H, Omura K, Azuma M (2006) Predominant expression of B7-H1 and its immunoregulatory roles in oral squamous cell carcinoma. Oral Oncol 42:268–274
Mehal WZ, Azzaroli F, Crispe IN (2001) Antigen presentation by liver cells controls intrahepatic T cell trapping, whereas bone marrow-derived cells preferentially promote intrahepatic T cell apoptosis. J Immunol 167:667–673
Strome SE, Dong H, Tamura H, Voss SG, Flies DB, Tamada K, Salomao D, Cheville J, Hirano F, Lin W, Kasperbauer JL, Ballman KV, Chen L (2003) B7-H1 blockade augments adoptive T-cell immunotherapy for squamous cell carcinoma. Cancer Res 63:6501–6505
Schmitt WE, Stassar MJ, Schmitt W, Little M, Cochlovius B (2001) In vitro induction of a bladder cancer-specific T-cell response by mRNA-transfected dendritic cells. J Cancer Res Clin Oncol 127:203–206
Nishiyama T, Tachibana M, Horiguchi Y, Nakamura K, Ikebe Y, Takesako K, Murai M (2001) Immunotherapy of bladder cancer using autologous dendritic cells pulsed with human lymphocyte antigen-A24-specific MAGE-3 peptide. Clin Cancer Res 7:23–31
Hsu FJ, Komarovskaya M (2002) CTLA4 blockade maximizes antitumor T-cell activation by dendritic cells presenting idiotype protein or opsonized anti-CD20 antibody-coated lymphoma cells. J Immunother 25:455–268
Nakano O, Sato M, Naito Y, Suzuki K, Orikasa S, Aizawa M, Suzuki Y, Shintaku I, Nagura H, Ohtani H (2001) Proliferative activity of intratumoral CD8(+) T-lymphocytes as a prognostic factor in human renal cell carcinoma: clinicopathologic demonstration of antitumor immunity. Cancer Res 61:5132–5136
Zisman A, Pantuck AJ, Dorey F, Said JW, Shvarts O, Quintana D, Gitlitz BJ, deKernion JB, Figlin RA, Belldegrun AS (2001) Improved prognostication of renal cell carcinoma using an integrated staging system. J Clin Oncol 19:1649–1657
Motzer RJ, Bander NH, Nanus DM (1996) Renal-cell carcinoma. N Engl J Med 335:865–875
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakanishi, J., Wada, Y., Matsumoto, K. et al. Overexpression of B7-H1 (PD-L1) significantly associates with tumor grade and postoperative prognosis in human urothelial cancers. Cancer Immunol Immunother 56, 1173–1182 (2007). https://doi.org/10.1007/s00262-006-0266-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-006-0266-z