Abstract
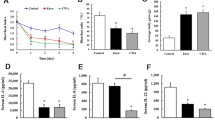
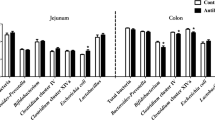
This study was to investigate the effects of chitosan-chelated zinc on ileal microbiota, inflammatory response, and barrier function in weaned piglets challenged with Escherichia coli K88. Piglets of the chitosan-chelated zinc treatment (Cs-Zn; 100 mg zinc + 766 mg chitosan/kg basal diet, from chitosan-chelated zinc) and the chitosan treatment (CS, 766 mg chitosan/kg basal diet) had significantly increased ileal villus height and the ratio of villi height to crypt depth. CS-Zn group piglets had a higher abundance of Lactobacillus in the ileal digesta, while the abundance of Streptococcus, Escherichia shigella, Actinobacillus, and Clostridium sensu stricto 6 was significantly decreased. The concentrations of propionate, butyrate, and lactate in the CS-Zn group piglets were significantly increased, while the pH value was significantly decreased. Furthermore, the concentrations of IL-1β, TNF-α, MPO, and INF-γ in the ileal mucosa of the CS-Zn and the H-ZnO group (pharmacological dose of 1600 mg Zn/kg basal diet, from ZnO) were significantly lower than those of the control group fed with basal diet, and the mRNA expression of TLR4, MyD88, and NF-κB of the CS-Zn group was also reduced. In addition, the mRNA expression of IGF-1 was increased, the protein expression of occludin and claudin-1 was enhanced, while the mRNA expression of caspase 3 and caspase 8 was decreased in the CS-Zn group. These results suggest CS-Zn treatment could help modulate the composition of ileal microbiota, attenuate inflammatory response, and maintain the intestinal function in weaned piglets challenged with Escherichia coli K88.
Graphical abstract

Key points
• Chitosan-chelated zinc significantly modulated ileal microbiota.
• Chitosan-chelated zinc can improve ileal health.
• The ileal microbiota plays an important role in host health.








Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Ahmad S, Elsherbiny NM, Bhatia K, Elsherbini AM, Fulzele S, Liou GI (2014) Inhibition of adenosine kinase attenuates inflammation and neurotoxicity in traumatic optic neuropathy. J Neuroinflammation 277(1–2):96–104. https://doi.org/10.1016/j.jneuroim.2014.10.006
Baregamian N, Song J, Jeschke MG, Evers BM, Chung DH (2006) IGF-1 protects intestinal epithelial cells from oxidative stress-induced apoptosis. J Surg Res 136(1):31–37. https://doi.org/10.1016/j.jss.2006.04.028
Barrow PA, Brooker BE, Fuller R, Newport MJ (1980) The attachment of bacteria to the gastric epithelium of the pig and its importance in the microecology of the intestine. J Appl Bacteriol 48(1):147–154. https://doi.org/10.1111/j.1365-2672.1980.tb05216.x
Bednorz C, Guenther S, Oelgeschlager K, Kinnemann B, Pieper R, Hartmann S, Tedin K, Semmler T, Neumann K, Schierack P (2013) Feeding the probiotic Enterococcus faecium strain NCIMB 10415 to piglets specifically reduces the number of Escherichia coli pathotypes that adhere to the gut mucosa. Appl Environ Microbiol 79(24):7896–7904. https://doi.org/10.1128/AEM.03138-13
Burcelin R, Garidou L, Pomie C (2012) Immuno-microbiota cross and talk: the new paradigm of metabolic diseases. Semin Immunol 24(1):67–74. https://doi.org/10.1016/j.smim.2011.11.011
Caporaso JG, Bittinger K, Bushman FD, DeSantis TZ, Andersen GL, Knight R (2010) PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics 26(2):266–267. https://doi.org/10.1093/bioinformatics/btp636
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6(8):1621–1624. https://doi.org/10.1038/ismej.2012.8
Devriendt B, Stuyven E, Verdonck F, Goddeeris BM, Cox E (2010) Enterotoxigenic Escherichia coli (K88) induce proinflammatory responses in porcine intestinal epithelial cells. Dev Comp Immunol 34(11):1175–1182. https://doi.org/10.1016/j.dci.2010.06.009
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27(16):2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Fan TJ, Han LH, Cong RS, Liang J (2010) Caspase family proteases and apoptosis. Acta Bioch Bioph Sin 37(11):719–727. https://doi.org/10.1111/j.1745-7270.2005.00108.x
Feng D (2020) Effects of chitosan-chelated zinc on intestinal morphology, microbiota structure and inflammatory response in rats challenged with Escherichia coli. Nanjing Agricultural University, Thesis
Feng D, Zhang M, Tian S, Wang J, Zhu W (2020) Chitosan-chelated zinc modulates cecal microbiota and attenuates inflammatory response in weaned rats challenged with Escherichia coli. J Microbiol 58(9):780–792. https://doi.org/10.1007/s12275-020-0056-x
Hammer y, Harper DA, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):1–9. https://doi.org/10.1016/j.palaeo.2009.11.002
Han XY, Ma YF, Lv MY, Wu ZP, Qian LC (2014) Chitosan-zinc chelate improves intestinal structure and mucosal function and decreases apoptosis in ileal mucosal epithelial cells in weaned pigs. Br J Nutr 111(8):1405–1411. https://doi.org/10.1017/S0007114513004042
Hanczakowska E, Agnieszka S, Krzysztof O (2011) Effects of dietary caprylic and capric acids on piglet performance and mucosal epithelium structure of the ileum. J Anim Feed Sci 20(4):556–565. https://doi.org/10.4081/ijas.2011.e41
Hermann-Bank M, Skovgaard K, Stockmarr A, Strube M, Boye M (2015) Characterization of the bacterial gut microbiota of piglets suffering from new neonatal porcine diarrhoea. BMC Vet Res 11:139. https://doi.org/10.1186/s12917-015-0419-4
Hojberg O, Canibe N, Poulsen HD, Hedemann MS, Jensen BB (2005) Influence of dietary zinc oxide and copper sulfate on the gastrointestinal ecosystem in newly weaned piglets. Appl Environ Microbiol 71(5):2267–2277. https://doi.org/10.1128/AEM.71.5.2267-2277.2005
Hu C, Song J, You Z, Luan Z, Li W (2012) Zinc oxide-montmorillonite hybrid influences diarrhea, intestinal mucosal integrity, and digestive enzyme activity in weaned pigs. Biol Trace Elem Res 149(2):190–196. https://doi.org/10.1007/s12011-012-9422-9
Hu J, Nie Y, Chen S, Xie C, Fan Q, Wang Z, Long B, Yan G, Zhong Q, Yan X (2017) Leucine reduces reactive oxygen species levels via an energy metabolism switch by activation of the mTOR-HIF-1α pathway in porcine intestinal epithelial cells. Int J Biochem Cell B 89:42–56. https://doi.org/10.1016/j.biocel.2017.05.026
Hu L, Cheng S, Li Y, Geng S, Ma Y, Han X (2018) Chitosan-Zn chelate downregulates TLR4-NFκB signal pathway of inflammatory response and cell death-associated proteins compared to inorganic zinc. Biol Trace Elem Res 184(1):92–98. https://doi.org/10.1007/s12011-017-1174-0
Hu P, Zhao F, Wang J, Zhu W (2020) Early-life lactoferrin intervention modulates the colonic microbiota, colonic microbial metabolites and intestinal function in suckling piglets. Appl Microbiol Biotechnol 104:6185–6197. https://doi.org/10.1007/s00253-020-10675-z
Huang C, Qiao S, Li D, Piao X, Ren J (2004) Effects of Lactobacilli on the performance, diarrhea incidence, VFA concentration and gastrointestinal microbial flora of weaning pigs. Asian Austral J Anim 17(3):401–409. https://doi.org/10.5713/ajas.2004.401
Katouli M, Melin L, Jensen-Waern M, Wallgren P, Mllby R (2010) The effect of zinc oxide supplementation on the stability of the intestinal flora with special reference to composition of coliforms in weaned pigs. J Appl Microbiol 87(4):564–573. https://doi.org/10.1046/j.1365-2672.1999.00853.x
Kobayashi KS, Chamaillard M, Ogura Y, Henegariu O, Inohara N, Nunez G, Flavell RA (2005) NOD2-dependent regulation of innate and adaptive immunity in the intestinal tract. Science 307(5710):731–734. https://doi.org/10.1126/science.1104911
Kobayashi M, Mikami D, Kimura H, Kamiyama K, Morikawa Y, Yokoi S, Kasuno K, Takahashi N, Taniguchi T, Iwano M (2017) Short-chain fatty acids, GPR41 and GPR43 ligands, inhibit TNF-α-induced MCP-1 expression by modulating p38 and JNK signaling pathways in human renal cortical epithelial cells. Biochem Biophys Res Commun 486(2):499–505. https://doi.org/10.1016/j.bbrc.2017.03.071
Litvak Y, Byndloss MX, Tsolis RM, Baumler AJ (2017) Dysbiotic proteobacteria expansion: a microbial signature of epithelial dysfunction. Curr Opin Microbiol 39:1–6. https://doi.org/10.1016/j.mib.2017.07.003
Liu P, Piao XS, Kim SW, Wang L, Shen YB, Lee HS, Li SY (2008) Effects of chito-oligosaccharide supplementation on the growth performance, nutrient digestibility, intestinal morphology, and fecal shedding of Escherichia coli and Lactobacillus in weaning pigs. J Anim Sci 86(10):2609–2618. https://doi.org/10.2527/jas.2007-0668
Liu P, Pieper R, Tedin L, Martin L, Meyer W, Rieger J, Plendl J, Vahjen W, Zentek J (2014) Effect of dietary zinc oxide on jejunal morphological and immunological characteristics in weaned piglets. J Anim Sci 92(11):5009–5018. https://doi.org/10.2527/jas.2013-6690
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001
Loh G, Eberhard M, Brunner RM, Hennig U, Kuhla S, Kleessen B, Metges CC (2006) Inulin alters the intestinal microbiota and short-chain fatty acid concentrations in growing pigs regardless of their basal diet. J Nutr 136(5):1198–1202. https://doi.org/10.1093/jn/136.5.1198
Nauseef WM (2007) Diagnostic assays for myeloperoxidase deficiency. Methods Mol Biol 412:525–530. https://doi.org/10.1007/978-1-59745-467-4_32
Opree A, Kress M (2000) Involvement of the proinflammatory cytokines tumor necrosis factor-α, IL-1β, and IL-6 but not IL-8 in the development of heat hyperalgesia: effects on heat-evoked calcitonin gene-related peptide release from rat skin. J Neurosci 20(16):6289–6293. https://doi.org/10.1016/S0006-3223(00)00914-8
Petra L, Harry JF (2017) Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 19(1):29–41. https://doi.org/10.1111/1462-2920.13589
Piccolo BD, Mercer KE, Bhattacharyya S, Bowlin AK, Saraf MK, Pack L, Chintapalli SV, Shankar K, Adams SH, Badger TM, Yeruva L (2017) Early postnatal diets affect the bioregional small intestine microbiome and ileal metabolome in neonatal pigs. J Nutr 147(8):1499–1509. https://doi.org/10.3945/jn.117.252767
Pieper R, Vahjen W, Neumann K, Van Kessel AG, Zentek J (2012) Dose-dependent effects of dietary zinc oxide on bacterial communities and metabolic profiles in the ileum of weaned pigs. J Anim Physiol Anim Nutr 96(5):825–833. https://doi.org/10.1111/j.1439-0396.2011.01231.x
Rastall RA, Gibson GR, Gill HS, Guarner F, Klaenhammer TR, Pot B, Reid G, Rowland IR, Sanders ME (2005) Modulation of the microbial ecology of the human colon by probiotics, prebiotics and synbiotics to enhance human health: an overview of enabling science and potential applications. Fems Microbiol Ecol 52(2):145–152. https://doi.org/10.1016/j.femsec.2005.01.003
Roller S, Covill N (1999) The antifungal properties of chitosan in laboratory media and apple juice. Int J Food Microbiol 47(1–2):67–77. https://doi.org/10.1016/s0168-1605(99)00006-9
Roselli M, Finamore A, Garaguso I, Britti M, Serena ME (2003) Zinc oxide protects cultured enterocytes from the damage induced by Escherichia coli. J Nutr 133(12):4077–4082. https://doi.org/10.1046/j.1365-277X.2003.00485.x
Sales J (2013) Effects of pharmacological concentrations of dietary zinc oxide on growth of post-weaning pigs: a meta-analysis. Biol Trace Elem Res 152(3):343–349. https://doi.org/10.1007/s12011-013-9638-3
Seil JT, Webster TJ (2012) Antibacterial effect of zinc oxide nanoparticles combined with ultrasound. Nanotechnology 23(49):495101. https://doi.org/10.1088/0957-4484/23/49/495101
Song ZH, Ke YL, Xiao K, Jiao LF, Hong QH, Hu CH (2015a) Diosmectite-zinc oxide composite improves intestinal barrier restoration and modulates TGF-β1, ERK1/2, and Akt in piglets after acetic acid challenge. J Anim Sci 93(4):1599–1607. https://doi.org/10.2527/jas.2014-8580
Song ZH, Xiao K, Ke YL, le Jiao F, Hu CH (2015b) Zinc oxide influences mitogen-activated protein kinase and TGF-β1 signaling pathways, and enhances intestinal barrier integrity in weaned pigs. Innate Immun 21(4):341–348. https://doi.org/10.1177/1753425914536450
Swiatkiewicz S, Swiatkiewicz M, Arczewska-Wlosek A, Jozefiak D (2015) Chitosan and its oligosaccharide derivatives (chito-oligosaccharides) as feed supplements in poultry and swine nutrition. J Anim Physiol Anim Nutr 99(1):1–12. https://doi.org/10.1111/jpn.12222
Tedelind S, Westberg F, Kjerrulf M, Vidal A (2007) Anti-inflammatory properties of the short-chain fatty acids acetate and propionate: a study with relevance to inflammatory bowel disease. World J Gastroenterol 13(20):2826–2832. https://doi.org/10.3748/wjg.v13.i20.2826
Tian S, Shi Q, Zhu Y, Yang H, Wang J, Zhu W (2020) Significant changes in caecal microbial composition and metabolites of weaned piglets after protein restriction and succedent realimentation. J Anim Physiol Anim Nutr 104(4):1126–1133. https://doi.org/10.1111/jpn.13268
Toth S, Jonecova Z, Kruzliak P, Ciccocioppo R, Nemcova R (2015) Influence of dietary supplementation with flaxseed and Lactobacilli on the cells of local innate immunity response in the jejunal mucosa in piglets after weaning. Acta Histochem 117(2):188–195. https://doi.org/10.1016/j.acthis.2014.12.005
Tremaroli V, Bäckhed F (2012) Functional interactions between the gut microbiota and host metabolism. Nature 489(7415):242–249. https://doi.org/10.1038/nature11552
Van Baarlen P, Wells JM, Kleerebezem M (2013) Regulation of intestinal homeostasis and immunity with probiotic Lactobacilli. Trends Immunol 34(5):208–215. https://doi.org/10.1016/j.it.2013.01.005
Wang X, Du Y, Liu H (2004) Preparation, characterization and antimicrobial activity of chitosan–Zn complex. Carbohydr Polym 56(1):21–26. https://doi.org/10.1016/j.carbpol.2003.11.007
Wang J, Chen L, Li P, Li X, Zhou H, Wang F, Li D, Yin Y, Wu G (2008) Gene expression is altered in piglet small intestine by weaning and dietary glutamine supplementation. J Nutr 138(6):1025–1032. https://doi.org/10.1089/jmf.2007.650
Wang MQ, Du YJ, Wang C, Tao WJ, He YD, Li H (2012) Effects of copper-loaded chitosan nanoparticles on intestinal microflora and morphology in weaned piglets. Biol Trace Elem Res 149(2):184–189. https://doi.org/10.1007/s12011-012-9410-0
Wang X, Wang W, Wang L, Yu C, Zhang G, Zhu H, Wang C, Zhao S, Hu CA, Liu Y (2019) Lentinan modulates intestinal microbiota and enhances barrier integrity in a piglet model challenged with lipopolysaccharide. Food Funct 10(1):479–489. https://doi.org/10.1039/c8fo02438c
Xia T, Lai W, Han M, Han M, Ma X, Zhang L (2017) Dietary ZnO nanoparticles alters intestinal microbiota and inflammation response in weaned piglets. Oncotarget 8(39):64878–64891. https://doi.org/10.18632/oncotarget.17612
Xiao D, Tang Z, Yin Y, Zhang B, Hu X, Feng Z, Wang J (2013) Effects of dietary administering chitosan on growth performance, jejunal morphology, jejunal mucosal sIgA, occludin, claudin-1 and TLR4 expression in weaned piglets challenged by enterotoxigenic Escherichia coli. Int J Immunopharmacol 17(3):670–676. https://doi.org/10.1016/j.intimp.2013.07.023
Xiong X, Yang HS, Wang XC, Hu Q, Liu CX, Wu X, Deng D, Hou YQ, Nyachoti CM, Xiao DF, Yin YL (2015) Effect of low dosage of chito-oligosaccharide supplementation on intestinal morphology, immune response, antioxidant capacity, and barrier function in weaned piglets. J Anim Sci 93(3):1089–1097. https://doi.org/10.2527/jas.2014-7851
Yang CM, Ferket PR, Hong QH, Zhou J, Cao GT, Zhou L, Chen AG (2012) Effect of chito-oligosaccharide on growth performance, intestinal barrier function, intestinal morphology and cecal microflora in weaned pigs. J Anim Sci 90(8):2671–2676. https://doi.org/10.2527/jas.2011-4699
Zhang B, Guo Y (2009) Supplemental zinc reduced intestinal permeability by enhancing occludin and zonula occludens protein-1 (ZO-1) expression in weaning piglets. Br J Nutr 102(05):687–693. https://doi.org/10.1017/S0007114509289033
Zhang C, Yu M, Yang Y, Mu C, Su Y, Zhu W (2016) Effect of early antibiotic administration on cecal bacterial communities and their metabolic profiles in pigs fed diets with different protein levels. Anaerobe 42:188–196. https://doi.org/10.1016/j.anaerobe.2016.10.016
Zhang Y, Zhou J, Dong Z, Li G, Yin Y (2019) Effect of dietary copper on intestinal microbiota and antimicrobial resistance profiles of Escherichia coli in weaned piglets. Front Microbiol 10:2808. https://doi.org/10.3389/fmicb.2019.02808
Zhang M, Hou G, Hu P, Feng D, Wang J, Zhu W (2021) Nano chitosan-zinc complex improves the growth performance and antioxidant capacity of the small intestine in weaned piglets. Br J Nutr 126(6):801–812. https://doi.org/10.1017/s0007114520004766
Zhou L, Fang L, Sun Y, Su Y, Zhu W (2016) Effects of the dietary protein level on the microbial composition and metabolomic profile in the hindgut of the pig. Anaerobe 38:61–69. https://doi.org/10.1016/j.anaerobe.2015.12.009
Zhu LH, Zhao KL, Chen XL, Xu JX (2012) Impact of weaning and an antioxidant blend on intestinal barrier function and antioxidant status in pigs. J Anim Sci 90(8):2581–2589. https://doi.org/10.2527/jas.2012-4444
Zhu H, Liu Y, Xie X, Huang J, Hou Y (2013) Effect of L-arginine on intestinal mucosal immune barrier function in weaned pigs after Escherichia coli LPS challenge. Innate Immun 19(3):242–252. https://doi.org/10.1177/1753425912456223
Zoetendal EG, Akkermans AD, De Vos WM (1998) Temperature gradient gel electrophoresis analysis of 16S rRNA from human fecal samples reveals stable and host-specific communities of active bacteria. Appl Environ Microbiol 64(10):3854–3859. https://doi.org/10.1128/AEM.64.10.3854-3859.1998
Acknowledgements
The authors thank the National Center for International Research on Animal Gut Nutrition for the financial support.
Funding
This study was supported by the National Key R&D Program of China 2017YFD0500505.
Author information
Authors and Affiliations
Contributions
GJ H and MY Z led the design and performance of the experiments, the analysis of the data, and the writing of the paper. J W and WY Z participated in designing the experiments and editing the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The experimental protocol was approved by the Animal Care and Use Committee of Nanjing Agricultural University (Nanjing, Jiangsu Province, China). All procedures in the present study were conducted in accordance with the approved guidelines and regulations.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hou, G., Zhang, M., Wang, J. et al. Chitosan-chelated zinc modulates ileal microbiota, ileal microbial metabolites, and intestinal function in weaned piglets challenged with Escherichia coli K88. Appl Microbiol Biotechnol 105, 7529–7544 (2021). https://doi.org/10.1007/s00253-021-11496-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11496-4