Abstract
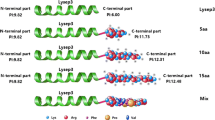
The present paper describes the generation of derivatives from the hybrid peptide called Ent35-MccV, active against Gram-positive and Gram-negative bacteria. This peptide has a triple glycine hinge region between enterocin CRL35 and microcin V. In order to obtain variants of Ent35-MccV with greater biotechnological potential, a saturation mutagenesis was carried out in the hinge region. As a result, we obtained a bank of E. coli strains expressing different mutated hybrid bacteriocins in the central position of the hinge region. From all these variants, we found that the one bearing a tyrosine in the central region of the hinge (Ent35-GYG-MccV) is 2-fold more active against E. coli and 4-fold more active against Listeria than the original peptide Ent35-MccV. This derivative was purified and characterized. The development and evaluation of alternative hinges for Ent35-MccV represents a step forward in the bioengineering of antimicrobial peptides. This approach fosters the rational design of peptides with enhanced antimicrobial activity.





Similar content being viewed by others
References
Acuña L, Corbalan NS, Fernandez-No IC, Morero RD, Barros-Velazquez J, Bellomio A (2015) Inhibitory effect of the hybrid Bacteriocin Ent35-MccV on the growth of Escherichia coli and Listeria monocytogenes in model and food systems. Food Bioprocess Technol 8:1063–1075. https://doi.org/10.1007/s11947-015-1469-0
Acuña L, Morero RD, Bellomio A (2011) Development of wide-Spectrum hybrid Bacteriocins for food biopreservation. Food Bioprocess Technol 4:1029–1049. https://doi.org/10.1007/s11947-010-0465-7
Acuña L, Picariello G, Sesma F, Morero RD, Bellomio A (2012) A new hybrid bacteriocin, Ent35-MccV, displays antimicrobial activity against pathogenic gram-positive and gram-negative bacteria. FEBS Open Bio 2:12–19. https://doi.org/10.1016/j.fob.2012.01.002
Alvarez-Sieiro P, Montalbán-López M, Mu D, Kuipers OP (2016) Bacteriocins of lactic acid bacteria: extending the family. Appl Microbiol Biotechnol 100:2939–2951. https://doi.org/10.1007/s00253-016-7343-9
Arbulu S, Jiménez JJ, Gútiez L, Feito J, Cintas LM, Herranz C, Hernández PE (2019) Cloning and expression of synthetic genes encoding native, hybrid- and bacteriocin-derived chimeras from mature class IIa bacteriocins, by Pichia pastoris (syn. Komagataella spp.). Food research international. https://doi.org/10.1016/j.foodres.2019.01.015
Azpiroz MF, Laviña M (2007) Modular structure of microcin H47 and colicin V. Antimicrob Agents Chemother 51:2412–2419. https://doi.org/10.1128/AAC.01606-06
Barraza DE, Ríos Colombo NS, Galván AE, Acuña L, Minahk CJ, Bellomio A, Chalón MC (2017) New insights into enterocin CRL35; mechanism of action and immunity revealed by heterologous expression in Escherichia coli. Mol Microbiol. https://doi.org/10.1111/mmi.13746
Bédard F, Hammami R, Zirah S, Rebuffat S, Fliss I, Biron E (2018) Synthesis, antimicrobial activity and conformational analysis of the class IIa bacteriocin pediocin PA-1 and analogs thereof. Sci Rep 8:9029–9013. https://doi.org/10.1038/s41598-018-27225-3
Bellomio, A, Minahk, C.J, Dupuy, F (2015) Broad-Spectrum hybrid and engineered Bacteriocins for food biopreservation: what will be the future of Bacteriocins? In: | microbial food safety and preservation techniques |. CRC Press, Estados Unidos
Buchan DWA, Jones DT (2019) The PSIPRED protein analysis workbench: 20 years on. Nucleic Acids Res 47:W402–W407. https://doi.org/10.1093/nar/gkz297
Chikindas ML, Weeks R, Drider D, Chistyakov VA, Dicks LM (2018) Functions and emerging applications of bacteriocins. Curr Opin Biotechnol 49:23–28. https://doi.org/10.1016/j.copbio.2017.07.011
Cui Y, Zhang C, Wang Y, Shi J, Zhang L, Ding Z, Qu X, Cui H (2012) Class IIa bacteriocins: diversity and new developments. Int J Mol Sci 13:16668–16707. https://doi.org/10.3390/ijms131216668
Farías ME, Farías RN, de Ruiz Holgado AP, Sesma F (1996) Purification and N-terminal amino acid sequence of Enterocin CRL 35, a “pediocin-like” bacteriocin produced by Enterococcus faecium CRL 35. Lett Appl Microbiol 22:417–419
Field D, Connor PMO, Cotter PD, Hill C, Ross RP (2008) The generation of nisin variants with enhanced activity against specific gram-positive pathogens. Mol Microbiol 69:218–230. https://doi.org/10.1111/j.1365-2958.2008.06279.x
Field D, Quigley L, O’Connor PM, Rea MC, Daly K, Cotter PD, Hill C, Ross RP (2010) Studies with bioengineered Nisin peptides highlight the broad-spectrum potency of Nisin V. Microb Biotechnol 3:473–486. https://doi.org/10.1111/j.1751-7915.2010.00184.x
Fimland G, Blingsmo OR, Sletten K, Jung G, Nes IF, Nissen-Meyer J (1996) New biologically active hybrid bacteriocins constructed by combining regions from various pediocin-like bacteriocins: the C-terminal region is important for determining specificity. Appl Environ Microbiol 62:3313–3318
Fimland G, Eijsink VGH, Nissen-Meyer J (2002) Comparative studies of immunity proteins of pediocin-like bacteriocins. Microbiology (Reading, Engl) 148:3661–3670. https://doi.org/10.1099/00221287-148-11-3661
Fink J, Boman A, Boman HG, Merrifield RB (1989) Design, synthesis and antibacterial activity of cecropin-like model peptides. Int J Pept Protein Res 33:412–421
Fregeau Gallagher NL, Sailer M, Niemczura WP, Nakashima TT, Stiles ME, Vederas JC (1997) Three-dimensional structure of leucocin a in trifluoroethanol and dodecylphosphocholine micelles: spatial location of residues critical for biological activity in type IIa bacteriocins from lactic acid bacteria. Biochemistry 36:15062–15072. https://doi.org/10.1021/bi971263h
Gasteiger E, Gattiker A, Hoogland C, Ivanyi I, Appel RD, Bairoch A (2003) ExPASy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 31:3784–3788. https://doi.org/10.1093/nar/gkg563
Gérard F, Pradel N, Wu L-F (2005) Bactericidal activity of colicin V is mediated by an inner membrane protein, SdaC, of Escherichia coli. J Bacteriol 187:1945–1950. https://doi.org/10.1128/JB.187.6.1945-1950.2005
Gratia (1925) Sur un remarquable example d’antagonisme entre deux souches de colibacille. Compt Rend Soc Biol 93:1040–1042
Holak TA, Engström A, Kraulis PJ, Lindeberg G, Bennich H, Jones TA, Gronenborn AM, Clore GM (1988) The solution conformation of the antibacterial peptide cecropin a: a nuclear magnetic resonance and dynamical simulated annealing study. Biochemistry 27:7620–7629
Hosseini H (1969) Bacterial sensitivity to antibiotics, 1964-1968. Curr Ther Res Clin Exp 11:397–405
Johnsen L, Fimland G, Eijsink V, Nissen-Meyer J (2000) Engineering increased stability in the antimicrobial peptide pediocin PA-1. Appl Environ Microbiol 66:4798–4802
Jones DT (1999) Protein secondary structure prediction based on position-specific scoring matrices. J Mol Biol 292:195–202. https://doi.org/10.1006/jmbi.1999.3091
Krokhin OV, Antonovici M, Ens W, Wilkins JA, Standing KG (2006) Deamidation of -Asn-Gly- sequences during sample preparation for proteomics: consequences for MALDI and HPLC-MALDI analysis. Anal Chem 78:6645–6650. https://doi.org/10.1021/ac061017o
Kyte J, Doolittle RF (1982) A simple method for displaying the hydropathic character of a protein. J Mol Biol 157:105–132. https://doi.org/10.1016/0022-2836(82)90515-0
Li Q, Montalban-Lopez M, Kuipers OP (2018) Increasing the Antimicrobial Activity of Nisin-Based Lantibiotics against Gram-Negative Pathogens Appl Environ Microbiol:84. https://doi.org/10.1128/AEM.00052-18
Pomares MF, Salomón RA, Pavlova O, Severinov K, Farías R, Vincent PA (2009) Potential applicability of chymotrypsin-susceptible microcin J25 derivatives to food preservation. Appl Environ Microbiol 75:5734–5738. https://doi.org/10.1128/AEM.01070-09
Rihakova J, Petit VW, Demnerova K, Prévost H, Rebuffat S, Drider D (2009) Insights into structure-activity relationships in the C-terminal region of divercin V41, a class IIa bacteriocin with high-level antilisterial activity. Appl Environ Microbiol 75:1811–1819. https://doi.org/10.1128/AEM.02266-08
Ríos Colombo NS, Chalón MC, Dupuy FG, Gonzalez CF, Bellomio A (2019) The case for class II bacteriocins: a biophysical approach using “suicide probes” in receptor-free hosts to study their mechanism of action. Biochimie 165:183–195. https://doi.org/10.1016/j.biochi.2019.07.024
Ríos Colombo NS, Chalón MC, Navarro SA, Bellomio A (2018) Pediocin-like bacteriocins: new perspectives on mechanism of action and immunity. Curr Genet 64:345–351. https://doi.org/10.1007/s00294-017-0757-9
Saavedra L, Minahk C, de Ruiz Holgado AP, Sesma F (2004) Enhancement of the enterocin CRL35 activity by a synthetic peptide derived from the NH2-terminal sequence. Antimicrob Agents Chemother 48:2778–2781. https://doi.org/10.1128/AAC.48.7.2778-2781.2004
Salvucci E, Saavedra L, Sesma F (2007) Short peptides derived from the NH2-terminus of subclass IIa bacteriocin enterocin CRL35 show antimicrobial activity. J Antimicrob Chemother 59:1102–1108. https://doi.org/10.1093/jac/dkm096
Schägger H (2006) Tricine-SDS-PAGE. Nat Protoc 1:16–22. https://doi.org/10.1038/nprot.2006.4
Shin SY, Kang JH, Jang SY, Kim Y, Kim KL, Hahm KS (2000) Effects of the hinge region of cecropin a(1-8)-magainin 2(1-12), a synthetic antimicrobial peptide, on liposomes, bacterial and tumor cells. Biochim Biophys Acta 1463:209–218
Steffens DL, Williams JGK (2007) Efficient site-directed saturation mutagenesis using degenerate oligonucleotides. J Biomol Tech 18:147–149
Tiwari BK, Valdramidis VP, O’ Donnell CP, Muthukumarappan K, Bourke P, Cullen PJ (2009) Application of natural antimicrobials for food preservation. J Agric Food Chem 57:5987–6000 . doi: https://doi.org/10.1021/jf900668n
Uteng M, Hauge HH, Markwick PRL, Fimland G, Mantzilas D, Nissen-Meyer J, Muhle-Goll C (2003) Three-dimensional structure in lipid micelles of the pediocin-like antimicrobial peptide sakacin P and a sakacin P variant that is structurally stabilized by an inserted C-terminal disulfide bridge. Biochemistry 42:11417–11426. https://doi.org/10.1021/bi034572i
van der Spoel D (1998) The solution conformations of amino acids from molecular dynamics simulations of Gly-X-Gly peptides: comparison with NMR parameters. Biochem Cell Biol 76:164–170
Yuan J, Zhang Z-Z, Chen X-Z, Yang W, Huan L-D (2004) Site-directed mutagenesis of the hinge region of nisinZ and properties of nisinZ mutants. Appl Microbiol Biotechnol 64:806–815. https://doi.org/10.1007/s00253-004-1599-1
Zaschke-Kriesche J, Reiners J, Lagedroste M, Smits SHJ (2019) Influence of nisin hinge-region variants on lantibiotic immunity and resistance proteins. Bioorg Med Chem 27:3947–3953. https://doi.org/10.1016/j.bmc.2019.07.014
Zhou L, van Heel AJ, Kuipers OP (2015) The length of a lantibiotic hinge region has profound influence on antimicrobial activity and host specificity. Front Microbiol 6. https://doi.org/10.3389/fmicb.2015.00011
Funding
This study was funded by grants PICT 4610 from the Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT), PIP 06906 CO from the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) and PIUNT D641/1 from the Secretaría de Ciencia, Arte e Innovación Tecnológica (SCAIT) from Universidad Nacional de Tucumán (UNT). S.A.N, L.L, N.S.R.C, M.F.U, are recipients of CONICET fellowship. S.A.N has received an ANPCyT fellowship. L.A, A.B and M.C.CH are career investigators of CONICET. B.S.P is CPA form CONICET.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 377 kb)
Rights and permissions
About this article
Cite this article
Navarro, S.A., Lanza, L., Colombo, N.S.R. et al. Obtaining an Ent35-MccV derivative with mutated hinge region that exhibits increased activity against Listeria monocytogenes and Escherichia coli. Appl Microbiol Biotechnol 103, 9607–9618 (2019). https://doi.org/10.1007/s00253-019-10187-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10187-5