Abstract
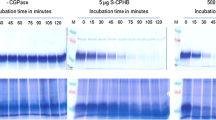
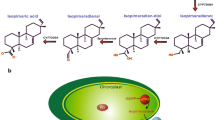
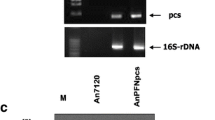
One of the major constraints in pig and poultry farming is the supply of protein-rich forage, containing sufficient amounts of key amino acids such as arginine (Ufaz and Galili 2008). Since these are underrepresented in plant proteins, the usage of plants as feed is limited. The heterologous production of the cyanobacterial storage polymer cyanophycin granule polypeptide (CGP) in plastids increases the amount of arginine substantially (Huhns et al. 2008; Huhns et al. 2009; Nausch et al. 2016a). CGP degradation releases arginine-aspartate dipeptides. CGP is stable in plants because its degradation is exclusively restricted to bacterial cyanophycinases (CGPases; Law et al. 2009). Since animals are also unable to digest CGP, CGPases need to be co-delivered with CGP-containing plant feed in order to release the dipeptides in the gastrointestinal tract of animals during digestion. Therefore, an extracellular CGPase, CphE from Pseudomonas alcaligenes DIP-1, was targeted to the cytosol, ER, and apoplasm of Nicotiana benthamiana. Translocation to the chloroplast was not successful. Although CphE accumulated in high amounts in the cytosol, only moderate levels were present in the ER, while the enzyme was nearly undetectable in the apoplasm. This correlates with the higher instability of post-translationally modified CphE in crude plant extracts. In addition, the production in the ER led to an increased number and size of necroses compared with cytosolic expression and might therefore interfere with the endogenous metabolism in the ER. Due to the high and robust enzyme activity, even moderate CphE concentrations were sufficient to degrade CGP in plant extracts.








Similar content being viewed by others
References
Arai Y, Shikanai T, Doi Y, Yoshida S, Yamaguchi I, Nakashita H (2004) Production of Polyhydroxybutyrate by polycistronic expression of bacterial genes in tobacco plastid. Plant Cell Physiol 45(9):1176–1184. doi:10.1093/pcp/pch139
Badri MA, Rivard D, Coenen K, Michaud D (2009) Unintended molecular interactions in transgenic plants expressing clinically useful proteins: the case of bovine aprotinin traveling the potato leaf cell secretory pathway. Proteomics 9(3):746–756. doi:10.1002/pmic.200700234
Barahimipour R, Strenkert D, Neupert J, Schroda M, Merchant SS, Bock R (2015) Dissecting the contributions of GC content and codon usage to gene expression in the model alga Chlamydomonas reinhardtii. Plant J 84(4):704–717. doi:10.1111/tpj.13033
Benchabane M, Goulet C, Rivard D, Faye L, Gomord V, Michaud D (2008) Preventing unintended proteolysis in plant protein biofactories. Plant Biotechnol J 6(7):633–648. doi:10.1111/j.1467-7652.2008.00344.x
Bendandi M, Marillonnet S, Kandzia R, Thieme F, Nickstadt A, Herz S, Fröde R, Inogés S, Lòpez-Dìaz de Cerio A, Soria E, Villanueva H, Vancanneyt G, McCormick A, Tusé D, Lenz J, Butler-Ransohoff J-E, Klimyuk V, Gleba Y (2010) Rapid, high-yield production in plants of individualized idiotype vaccines for non-Hodgkin’s lymphoma. Ann Oncol 21(12):2420–2427. doi:10.1093/annonc/mdq256
Broer S (2008) Amino acid transport across mammalian intestinal and renal epithelia. Physiol Rev 88(1):249–286. doi:10.1152/physrev.00018.2006
Chen H, Wong EA, Webb KE (1999) Tissue distribution of a peptide transporter mRNA in sheep, dairy cows, pigs, and chickens. J Anim Sci 77(5):1277–1283
Delannoy M, Alves G, Vertommen D, Ma J, Boutry M, Navarre C (2008) Identification of peptidases in Nicotiana tabacum leaf intercellular fluid. Proteomics 8(11):2285–2298. doi:10.1002/pmic.200700507
Duwadi K, Chen L, Menassa R, Dhaubhadel S (2015) Identification, characterization and down-regulation of cysteine protease genes in tobacco for use in recombinant protein production. PLoS One 10(7). doi:10.1371/journal.pone.0130556
Fan P, Wang X, Kuang T, Li Y (2009) An efficient method for the extraction of chloroplast proteins compatible for 2-DE and MS analysis. Electrophoresis 30(17):3024–3033. doi:10.1002/elps.200900172
FAO (2014) pigs and nutrition and feed
Frommeyer M, Wiefel L, Steinbüchel A (2014) Features of the biotechnologically relevant polyamide family “cyanophycins” and their biosynthesis in prokaryotes and eukaryotes. Crit Rev Biotechnol 0(0):1–12. doi:10.3109/07388551.2014.946467
Gómez G, Pallás V (2010) Noncoding RNA mediated traffic of foreign mRNA into chloroplasts reveals a novel signaling mechanism in plants. PLoS One 5(8):e12269. doi:10.1371/journal.pone.0012269
Goulet C, Khalf M, Sainsbury F, D’Aoust M-A, Michaud D (2012) A protease activity-depleted environment for heterologous proteins migrating towards the leaf cell apoplast. Plant Biotechnol J 10(1):83–94. doi:10.1111/j.1467-7652.2011.00643.x
Güttler S (2008) Forschung und Entwicklung in der Aquakultur - Ein Überblick über Arbeitsgebiete und offene Fragen. Innovation & Information Working Paper; Institut für Agrarökonomie; Christian-Albrechts-Universität Kiel doi:http://www.agric-econ.uni-kiel.de/Abteilungen/II/wp.shtml
Hamorsky KT, Kouokam JC, Bennett LJ, Baldauf KJ, Kajiura H, Fujiyama K, Matoba N (2013) Rapid and scalable plant-based production of a cholera toxin B subunit variant to aid in mass vaccination against cholera outbreaks. PLoS Negl Trop Dis 7(3):e2046. doi:10.1371/journal.pntd.0002046
Hamorsky KT, Kouokam JC, Jurkiewicz JM, Nelson B, Moore LJ, Husk AS, Kajiura H, Fujiyama K, Matoba N (2015) N-glycosylation of cholera toxin B subunit in Nicotiana benthamiana: impacts on host stress response, production yield and vaccine potential. Scientific Reports 5. doi:10.1038/srep08003
He J, Peng L, Lai H, Hurtado J, Stahnke J (2014) Chen Q (2014) a plant-produced antigen elicits potent immune responses against west nile virus in mice. Biomed Res Int. doi:10.1155/2014/952865
Huang Z, Santi L, LePore K, Kilbourne J, Arntzen CJ, Mason HS (2006) Rapid, high-level production of hepatitis B core antigen in plant leaf and its immunogenicity in mice. Vaccine 24(14):2506–2513. doi:10.1016/j.vaccine.2005.12.024
Huhns M, Neumann K, Hausmann T, Klemke F, Lockau W, Kahmann U, Kopertekh L, Staiger D, Pistorius EK, Reuther J, Waldvogel E, Wohlleben W, Effmert M, Junghans H, Neubauer K, Kragl U, Schmidt K, Schmidtke J, Broer I (2009) Tuber-specific cphA expression to enhance cyanophycin production in potatoes. Plant Biotechnol J 7(9):883–898. doi:10.1111/j.1467-7652.2009.00451.x
Huhns M, Neumann K, Hausmann T, Ziegler K, Klemke F, Kahmann U, Staiger D, Lockau W, Pistorius EK, Broer I (2008) Plastid targeting strategies for cyanophycin synthetase to achieve high-level polymer accumulation in Nicotiana tabacum. Plant Biotechnol J 6(4):321–336. doi:10.1111/j.1467-7652.2007.00320.x
Huhns M, Neumann K, Lockau W, Ziegler K, Pistorius EK, Broer I (2004) Bioplastic in transgenic plants: cyanophycin as a suitable resource for polyaspartate. Paper presented at the Agricultural Biotechnology International Conference, Cologne, Germany
Hussain H, Maldonado-Agurto R, Dickson AJ (2014) The endoplasmic reticulum and unfolded protein response in the control of mammalian recombinant protein production. Biotechnol Lett 36(8):1581–1593. doi:10.1007/s10529-014-1537-y
Kalthoff D, Giritch A, Geisler K, Bettmann U, Klimyuk V, Hehnen HR, Gleba Y, Beer M (2010) Immunization with plant-expressed hemagglutinin protects chickens from lethal highly pathogenic avian influenza virus H5N1 challenge infection. J Virol 84(22):12002–12010. doi:10.1128/JVI.00940-10
Klang JE, Burnworth LA, Pan YX, Webb KE Jr, Wong EA (2005) Functional characterization of a cloned pig intestinal peptide transporter (pPepT1). J Anim Sci 83(1):172–181
Klemke F, Nürnberg DJ, Ziegler K, Beyer G, Kahmann U, Lockau W, Volkmer T (2016) CphA2 is a novel type of cyanophycin synthetase in N2-fixing cyanobacteria. Microbiology (United Kingdom) 162(3):526–536. doi:10.1099/mic.0.000241
Lallemand J, Bouché F, Desiron C, Stautemas J, de Lemos EF, Périlleux C, Tocquin P (2015) Extracellular peptidase hunting for improvement of protein production in plant cells and roots. Front Plant Sci 6. doi:10.3389/fpls.2015.00037
Law AM, Lai SWS, Tavares J, Kimber MS (2009) The structural basis of β-peptide-specific cleavage by the serine protease cyanophycinase. J Mol Biol 392(2):393–404. doi:10.1016/j.jmb.2009.07.001
Liu J-Z, Blancaflor EB, Nelson RS (2005) The tobacco mosaic virus 126-kilodalton protein, a constituent of the virus replication complex, alone or within the complex aligns with and traffics along microfilaments. Plant Physiol 138(4):1853–1865. doi:10.1104/pp.105.065722
Lu H, Klaassen C (2006) Tissue distribution and thyroid hormone regulation of Pept1 and Pept2 mRNA in rodents. Peptides 27(4):850–857. doi:10.1016/j.peptides.2005.08.012
Marillonnet S, Giritch A, Gils M, Kandzia R, Klimyuk V, Gleba Y (2004) In planta engineering of viral RNA replicons: efficient assembly by recombination of DNA modules delivered by Agrobacterium. Proc Natl Acad Sci U S A 101(18):6852–6857. doi:10.1073/pnas.0400149101
Nakamura Y, Kaneko T, Sato S, Ikeuchi M, Katoh H, Sasamoto S, Watanabe A, Iriguchi M, Kawashima K, Kimura T, Kishida Y, Kiyokawa C, Kohara M, Matsumoto M, Matsuno A, Nakazaki N, Shimpo S, Sugimoto M, Takeuchi C, Yamada M, Tabata S (2002) Complete genome structure of the thermophilic cyanobacterium Thermosynechococcus elongatus BP-1. DNA Res 9(4):123–130. doi:10.1093/dnares/9.4.123
Nausch H, Hausmann T, Ponndorf D, Hühns M, Hoedtke S, Wolf P, Zeyner A, Broer I (2016a) Tobacco as platform for a commercial production of cyanophycin. New Biotechnol 33(6):842–851. doi:10.1016/j.nbt.2016.08.001
Nausch H, Huckauf J, Broer I (2016b) Peculiarities and impacts of expression of bacterial cyanophycin synthetases in plants. Appl Microbiol Biotechnol 100(4):1559–1565. doi:10.1007/s00253-015-7212-y
Nausch H, Mischofsky H, Koslowski R, Meyer U, Broer I, Huckauf J (2012) Expression and subcellular targeting of human complement factor C5a in Nicotiana species. PLoS One 7(12). doi:10.1371/journal.pone.0053023
Pillay P, Schlüter U, van Wyk S, Kunert KJ, Vorster BJ (2014) Proteolysis of recombinant proteins in bioengineered plant cells. Bioengineered 5(1):15–20. doi:10.4161/bioe.25158
Pinkhasov J, Alvarez ML, Rigano MM, Piensook K, Larios D, Pabst M, Grass J, Mukherjee P, Gendler SJ, Walmsley AM, Mason HS (2011) Recombinant plant-expressed tumour-associated MUC1 peptide is immunogenic and capable of breaking tolerance in MUC1.Tg mice. Plant Biotechnol J 9(9):991–1001. doi:10.1111/j.1467-7652.2011.00614.x
Ponndorf D, Ehmke S, Waliser B, Unger C, Görs S, Das G, Metges C, Broer I, Nausch H (2016) Production and stabilization of cyanophycinase in Nicotiana benthamiana and its functionality to hydrolyse cyanophycin in the murine intestine. Plant Biotechnol J (accepted). doi:10.1111/pbi.12658
Robert S, Khalf M, Goulet M-C, D’Aoust M-A, Sainsbury F, Michaud D (2013) Protection of recombinant mammalian antibodies from development-dependent proteolysis in leaves of Nicotiana benthamiana. PLoS One 8(7):e70203. doi:10.1371/journal.pone.0070203
Sainsbury F, Varennes-Jutras P, Goulet M-C, D’Aoust M-A, Michaud D (2013) Tomato cystatin SlCYS8 as a stabilizing fusion partner for human serpin expression in plants. Plant Biotechnol J 11(9):1058–1068. doi:10.1111/pbi.12098
Sallam A, Kalkandzhiev D, Steinbüchel A (2011) Production optimization of cyanophycinase ChpEal from Pseudomonas alcaligenes DIP1. AMB Express 1(1):1–9. doi:10.1186/2191-0855-1-38
Sallam A, Steinbuchel A (2010) Dipeptides in nutrition and therapy: cyanophycin-derived dipeptides as natural alternatives and their biotechnological production. Appl Microbiol Biotechnol 87(3):815–828. doi:10.1007/s00253-010-2641-0
Sallam A, Steinbüchel A (2009a) Process for the preparation of dipeptides from cyanophycin employing the isolated Pseudomonas alcaligenes DIP1 CGPase CphEal. Google Patents
Sallam A, Steinbuchel A (2009b) Cyanophycin-degrading bacteria in digestive tracts of mammals, birds and fish and consequences for possible applications of cyanophycin and its dipeptides in nutrition and therapy. J Appl Microbiol 107(2):474–484. doi:10.1111/j.1365-2672.2009.04221.x
Santi L, Batchelor L, Huang Z, Hjelm B, Kilbourne J, Arntzen CJ, Chen Q, Mason HS (2008) An efficient plant viral expression system generating orally immunogenic Norwalk virus-like particles. Vaccine 26(15):1846–1854. doi:10.1016/j.vaccine.2008.01.053
Santi L, Giritch A, Roy CJ, Marillonnet S, Klimyuk V, Gleba Y, Webb R, Arntzen CJ, Mason HS (2006) Protection conferred by recombinant Yersinia pestis antigens produced by a rapid and highly scalable plant expression system. Proc Natl Acad Sci U S A 103(4):861–866. doi:10.1073/pnas.0510014103
Santos S, Torcato I, Castanho MA (2012) Biomedical applications of dipeptides and tripeptides. Biopolymers 98(4):288–293
Solá RJ, Griebenow KAI (2009) Effects of glycosylation on the stability of protein pharmaceuticals. J Pharm Sci 98(4):1223–1245. doi:10.1002/jps.21504
Stevens LH, Stoopen GM, Elbers IJW, Molthoff JW, Bakker HAC, Lommen A, Bosch D, Jordi W (2000) Effect of climate conditions and plant developmental stage on the stability of antibodies expressed in transgenic tobacco. Plant Physiol 124(1):173–182. doi:10.1104/pp.124.1.173
Teh YHA, Kavanagh TA (2010) High-level expression of camelid nanobodies in Nicotiana benthamiana. Transgenic Res 19(4):575–586. doi:10.1007/s11248-009-9338-0
Terova G, Robaina L, Izquierdo M, Cattaneo A, Molinari S, Bernardini G, Saroglia M (2013) PepT1 mRNA expression levels in sea bream (Sparus aurata) fed different plant protein sources. SpringerPlus 2(1):17. doi:10.1186/2193-1801-2-17
Thomas DR, Walmsley AM (2014) The effect of the unfolded protein response on the production of recombinant proteins in plants. Plant Cell Rep 34(2):179–187. doi:10.1007/s00299-014-1680-x
Ufaz S, Galili G (2008) Improving the content of essential amino acids in crop plants: goals and opportunities. Plant Physiol 147(3):954–961. doi:10.1104/pp.108.118091
Webster DE, Wang L, Mulcair M, Ma C, Santi L, Mason HS, Wesselingh SL, Coppel RL (2009) Production and characterization of an orally immunogenic Plasmodium antigen in plants using a virus-based expression system. Plant Biotechnol J 7(9):846–855. doi:10.1111/j.1467-7652.2009.00447.x
Acknowledgements
We thank Prof. Dr. Steinbüchel (Institut für Molekulare Mikrobiologie und Biotechnologie, Westfälische Wilhelms-Universität, Münster, Germany) and Dr. Krehenbrink (Cysal GmbH, Münster, Germany) for providing the CphE encoding plasmid. Likewise, we thank Dr. Gleba and Dr. Giritch (Nomad BioScience; Halle/Saale, Germany) for the MagnICON expression vectors and for their constructive discussions. In addition, we would like to express the warmest of thanks to Alex Rajewski for thoroughly proof-reading the manuscript and the helpful suggestions and comments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors did not receive any funding.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 598 kb)
Rights and permissions
About this article
Cite this article
Nausch, H., Broer, I. Cyanophycinase CphE from P. alcaligenes produced in different compartments of N. benthamiana degrades high amounts of cyanophycin in plant extracts. Appl Microbiol Biotechnol 101, 2397–2413 (2017). https://doi.org/10.1007/s00253-016-8020-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-8020-8